(1)要证明亚硫酸钠具有还原性,除亚硫酸钠外还应选用的试剂有
(2)要证明亚硫酸钠具有氧化性,除亚硫酸钠外还应选用的试剂有
相似题推荐
实验室现有质量分数为36.5%、密度为1.19g•cm﹣3的浓盐酸,若用该浓盐酸配制0.4mol•L﹣1的稀盐酸220mL,试回答下列问题:
(1)配制该稀盐酸时,应选用的容量瓶的容积为
A. 100mL B. 220mL C. 250mL D. 500mL
(2)计算需要
(3)在量取浓盐酸后,进行了下列操作:
①待稀释盐酸的温度与室温一致后,沿玻璃棒注入容量瓶中.
②往容量瓶中加蒸馏水至液面离容量瓶刻度线1~2cm时,改用胶头滴管加蒸馏水,使溶液的液面与瓶颈的刻度标线相切.
③在盛蒸馏水的烧杯中注入盐酸,并用玻璃棒搅动,使其混合均匀.
④用蒸馏水洗涤烧杯和玻璃棒2至3次,并将洗涤液全部注入容量瓶.
上述操作中,正确的顺序是(填序号)
(4)在下述配制过程中,配制的稀盐酸浓度将会偏高的是
①未转移溶液前容量瓶中有少量的蒸馏水
②量取浓盐酸时俯视刻度线
③量取浓盐酸后用蒸馏水洗涤量筒
④若未用蒸馏水洗涤烧杯内壁或未将洗涤液注入容量瓶
⑤定容时俯视刻度线
(5)在标准状况下,用一支收集满HCl气体的试管倒立在水槽中,水倒吸当充满试管时用塞子堵住试管口竖立(假设溶液不泄漏),则该溶液的溶质的物质的量浓度为

(1)除去粗盐中杂质(Mg2+、SO42-、Ca2+),加入的药品顺序正确的是
A.NaOH溶液→Na2CO3溶液→BaCl2溶液→过滤后加盐酸
B.BaCl2溶液→NaOH溶液→Na2CO3溶液→过滤后加盐酸
C.NaOH溶液→BaCl2溶液→Na2CO3溶液→过滤后加盐酸
D.Na2CO3溶液→NaOH溶液→BaCl2溶液→过滤后加盐酸
(2)制备钠单质的方法之一是将熔融NaCl进行电解,写出电解化学方程式:
(3)步骤Ⅰ中已获得Br2,步骤Ⅱ中又将Br2还原为Br-,其目的是
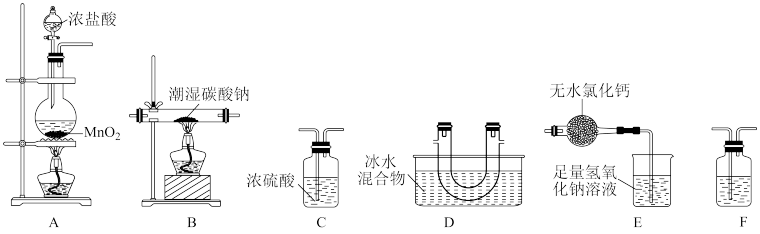
(4)某化学小组的同学为了了解从工业溴中提纯溴的方法,查阅了有关资料,Br2的沸点为59 ℃。微溶于水,有毒性和强腐蚀性。他们参观生产过程后,设计了如下实验装置:

①图中仪器B的名称是
②C中液体的颜色为
③D装置的作用是
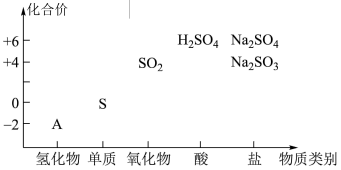
(2)
 与A溶液反应有淡黄色固体生成,该反应的化学方程式为
与A溶液反应有淡黄色固体生成,该反应的化学方程式为(3)
 和氯水都有漂白性,有人为增强漂白效果,将
和氯水都有漂白性,有人为增强漂白效果,将 和
和 混合使用,结果适得其反,几乎没有漂白效果,用离子方程式表示其原因
混合使用,结果适得其反,几乎没有漂白效果,用离子方程式表示其原因(4)
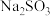 溶液易变质,实验室检验
溶液易变质,实验室检验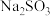 溶液是否变质的方法是
溶液是否变质的方法是(5)大苏打(
 )在照相、电影、纺织、皮革、农药等方面均有重要作用。现欲在实验室制备大苏打,从氧化还原角度分析,下列合理的是 (填字母)。
)在照相、电影、纺织、皮革、农药等方面均有重要作用。现欲在实验室制备大苏打,从氧化还原角度分析,下列合理的是 (填字母)。A. | B.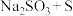 | C. | D.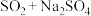 |
(6)利用如下装置探究
 气体与
气体与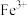 的反应:
的反应: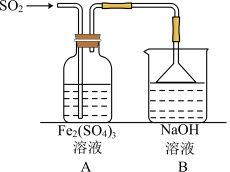
 ,为了验证A中
,为了验证A中 与
与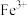 发生了氧化还原反应,取A中的溶液,分成两份,设计如下实验:
发生了氧化还原反应,取A中的溶液,分成两份,设计如下实验:方案①;向第一份溶液中加入少量酸性
 溶液,紫红色褪去。
溶液,紫红色褪去。方案②:向第二份溶液中加入KSCN溶液,不变红,再加入新制的氯水,溶液变红。
上述方案不合理的是
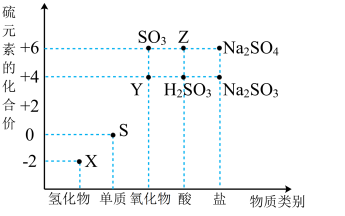
(1)仅从
(2)X与Z在一定条件下可以发生反应:
 (浓)
(浓)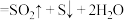 。用双线桥法标出上述反应转移的电子数目和方向
。用双线桥法标出上述反应转移的电子数目和方向(3)请预测X与Y能否共存
(4)写出一个上述含硫物质间相互转化的非氧化还原反应的离子方程式
(5)
 是重要的化工原料。从氧化还原反应的角度分析,下列制备
是重要的化工原料。从氧化还原反应的角度分析,下列制备 的方案理论上可行的是___________(填字母)。
的方案理论上可行的是___________(填字母)。A. | B.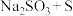 | C.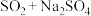 | D. |
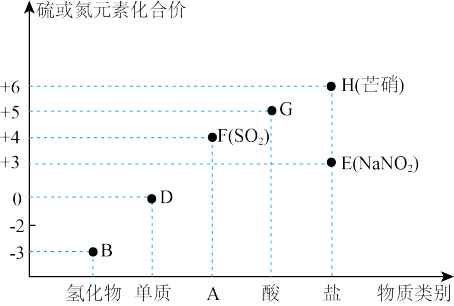
完成下列填空:
(1)
 的物质类别是
的物质类别是(2)D可以为
 或
或 ,已知同温同压下,同体积的
,已知同温同压下,同体积的 与
与 (蒸气)的质量比为7∶64,则
(蒸气)的质量比为7∶64,则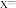
(3)下列物质能与
 发生反应,其中
发生反应,其中 体现还原性的是
体现还原性的是a.氢氧化钠溶液 b.溴水 c.品红溶液
(4)常温下,
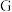 的浓溶液可以用干燥的铁或铝容器盛装,原因是
的浓溶液可以用干燥的铁或铝容器盛装,原因是(5)已知
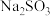 溶液既有氧化性又有还原性,请设计实验证明之。(供选用的试剂是:硫化钠溶液 品红溶液酸性
溶液既有氧化性又有还原性,请设计实验证明之。(供选用的试剂是:硫化钠溶液 品红溶液酸性 溶液 澄清石灰水 稀硫酸)。
溶液 澄清石灰水 稀硫酸)。
 2NaCl+2NaHCO3+Cl2O。
2NaCl+2NaHCO3+Cl2O。