I.称取ag研细的石灰石样品于250mL烧杯中,加过量稀盐酸溶解,水浴加热10分钟;
II.稍冷后逐滴加入氨水至溶液pH≈4,再缓慢加入适量(NH4)2C2O4溶液,继续水浴加热30分钟;
III.冷却至室温后过滤出沉淀,用另外配制的稀(NH4)2C2O4溶液洗涤沉淀三次,再用蒸馏水洗涤至洗涤液中无法检出Cl−;
IV.加入适量热的稀硫酸至沉淀中,获得的溶液用cmol·L−1KMnO4标准溶液滴定至终点。
V.平行测定三次,消耗KMnO4溶液的体积平均为VmL。
已知:H2C2O4是弱酸;CaC2O4是难溶于水的白色沉淀。
(1)I中为了加快反应速率而采取的操作有
(2)II中加入氨水调节溶液pH的作用是
(3)III中洗涤得到干净的沉淀。
①结合平衡移动原理,解释用稀(NH4)2C2O4溶液洗涤沉淀的目的
②验证沉淀是否洗涤干净的操作是
③若沉淀中的Cl−未洗涤干净,则最终测量结果
(4)IV中用KMnO4标准溶液滴定。
①滴定时发生反应的离子方程式为
②滴定至终点的现象为
③样品中以CaO质量分数表示的钙含量为
相似题推荐
(1)NH3还原NO的主反应为4NH3(g)+4NO(g)+O2(g)=4N2(g)+6H2O(g)。
已知:N2(g)+O2(g)=2NO(g)
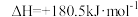 ;4NH3(g)+3O2(g)=2N2(g)+6H2O(g)
;4NH3(g)+3O2(g)=2N2(g)+6H2O(g) 
上述主反应的
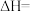
(2)在某钒催化剂中添加一定量Cu2O可加快NO的脱除效率,其可能机理如图所示(*表示物种吸附在催化剂表面,部分物种未画出)。
②烟气中若含有SO2,会生成NH4HSO4堵塞催化剂孔道。生成NH4HSO4的化学方程式为
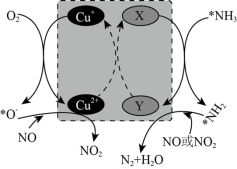
(3)将模拟烟气(一定比例NOx、NH3、O2和N2)以一定流速通过装有Fe/Zr催化剂的反应管,测得NOx转化率随温度变化的曲线如图所示。
②温度高于450℃时,NOx转化率已降低为负值,其可能原因是
(1)该同学认为:由于预先控制了反应的其他条件,那么,两次实验时反应的速率不一样的原因,只有以下五种可能:
原因I:Cl-对反应具有促进作用,而SO42-对反应没有影响;
原因II:
原因III:Cl-对反应具有促进作用,而SO42-对反应具有阻碍作用;
原因IV:Cl-、SO42-均对反应具有促进作用,但Cl-影响更大;
原因V:
(2)该同学设计并进行了两组实验,即得出了正确的结论。他取了两片等质量、外形和组成相同、表面经过砂纸充分打磨的铝片,分别放入到盛有同体积、c(H+)相同的稀硫酸和稀盐酸的试管(两试管的规格相同)中:
①在盛有稀硫酸的试管中加入少量NaCl或KCl固体,观察反应速率是否变化;
②在盛有稀盐酸的试管中加入少量Na2SO4或K2SO4固体,观察反应速率是否变化。若观察到实验①中
(3)为了使实验“定量化”,使结果更精确,可以对实验进行如下改进:
①配制c(H+)相同的稀盐酸和稀硫酸:现有浓度为1mol·L-1的盐酸和密度为1.225g·cm-3、质量分数为20%的硫酸,若要准确量取该硫酸20.00mL,需要用
配制溶液时还需要用到的玻璃仪器有
②比较反应速率:反应时间可以用秒表测定。如果要对上述实验中的反应速率进行比较,可以通过测定哪些物理量来确定?要求回答一种即可。
【推荐3】甲烷是最简单的烃,可用来作为燃料,也是一种重要的化工原料。
(1)A.CH4(g)+2O2(g)=CO2(g)+2H2O(g) ΔH=-Q1kJ/mol
B.CH4(g)+2O2(g)=CO2(g)+2H2O(l) ΔH=-Q2kJ/mol
C.CH4(g)+2O2(g)=CO2(g)+2H2O(l) ΔH=+Q2kJ/mol
D. CH4(g)+O2(g)=
CH4(g)+O2(g)= CO2(g)+H2O(l) ΔH=
CO2(g)+H2O(l) ΔH= Q2kJ/mol
Q2kJ/mol
上述热化学方程式中的反应热能正确表示甲烷燃烧热的是
(2)以甲烷为原料通过以下反应可以合成甲醇:2CH4(g)+O2(g) CH3OH(g) ΔH=-251.0kJ/mol
CH3OH(g) ΔH=-251.0kJ/mol
现将2molCH4(g)和1molO2(g)充入密闭容器中,在不同温度和压强下进行上述反应。实验测得平衡时甲醇的物质的量随温度、压强的变化如图所示:
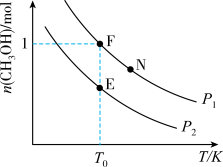
①P1时升高温度,n(CH3OH)
②E、F、N点对应的化学反应速率由大到小的顺序为
③下列能提高CH4平衡转化率的措施是
a.选择高效催化剂b.增大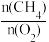 投料比
投料比
c.及时分离产物
④若F点,n(CH3OH)=1mol,总压强为2.5MPa,则T0时F点用分压强代替浓度表示的平衡常数Kp=
⑤在温度为T0,压强为P2时,各物质以下列量进行投料反应,达到平衡时,混合气体各组分的百分含量与E点相同的是
A.4molCH4(g)和2molO2(g)
B.2molCH4(g)、1molO2(g)和2molCH3OH(g)
C.1molCH3OH(g)
D.1molCH4(g)、1molO2(g)和1molCH3OH(g)
E.1molCH4(g)、0.5molO2(g)和1molCH3OH(g)
实验一:焦亚硫酸钠的制取
采用如图装置(实验前已除尽装置内的空气)制取Na2S2O5。装置Ⅱ中有Na2S2O5晶体析出,发生的反应为

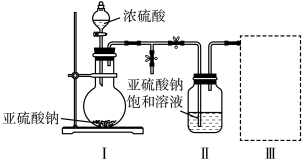
(1)装置Ⅰ中产生气体的化学方程式为
(2)要从装置Ⅱ中获得已析出的晶体,采取的分离方法是
(3)装置Ⅲ中用于处理尾气,可选用的最合理装置(夹持仪器已略去)为
a.
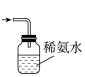 b.
b. 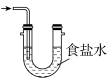 c.
c.  d.
d. 
实验二:焦亚硫酸钠的性质:Na2S2O5溶于水即生成
(4)证明
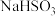 溶液中
溶液中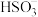 的电离程度大于水解程度,可采用的实验方法是
的电离程度大于水解程度,可采用的实验方法是a.测定溶液的pH b.加入
 溶液 c.加入盐酸
溶液 c.加入盐酸d.加入品红溶液 e.用蓝色石蕊试纸检测
(5)检验Na2S2O5晶体在空气中已被氧化的实验方案是
实验三:葡萄酒中抗氧化剂残留量的测定
(6)葡萄酒常用Na2S2O5作抗氧化剂。测定某葡萄酒中抗氧化剂的残留量(以游离的
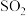 计算)的方案如下:
计算)的方案如下: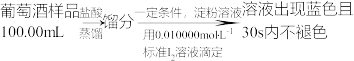
(已知滴定时反应的化学方程式为
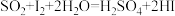 )
)①按上述方案实验,消耗标准
 溶液25.00mL,该次实验测得样品中抗氧化剂的残留量(以游离的
溶液25.00mL,该次实验测得样品中抗氧化剂的残留量(以游离的 计算)为
计算)为②在上述实验过程中,若有部分HI被空气氧化,则测得结果
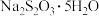 ,
,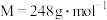 )可用作还原剂。回答下列问题:
)可用作还原剂。回答下列问题:(1)已知:
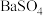 ,
, 都是难溶于水的固体。市售硫代硫酸钠中常含有硫酸根杂质,选用下列试剂设计实验方案进行检验:
都是难溶于水的固体。市售硫代硫酸钠中常含有硫酸根杂质,选用下列试剂设计实验方案进行检验:试剂:稀盐酸、稀
 、
、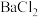 溶液、
溶液、 溶液、
溶液、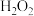 溶液
溶液| 实验步骤 | 现象 |
| ①取少量样品,加入除氧蒸馏水 | ②固体完全溶解得无色澄清溶液 |
| ③取少量溶液溶于盐酸 | ④出现乳黄色浑浊, |
| ⑤静置, | ⑥ |
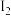 作为标准溶液定量测定硫代硫酸钠的纯度。测定步骤如下:
作为标准溶液定量测定硫代硫酸钠的纯度。测定步骤如下:①溶液配制;称取1.270g纯净的
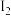 ,在盛有KI溶液的
,在盛有KI溶液的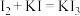 ,
,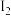 能够充分反应),完全溶解后,全部转移至100 mL的
能够充分反应),完全溶解后,全部转移至100 mL的 溶液的物质的量浓度为
溶液的物质的量浓度为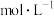 ;再取
;再取 样品配制成溶液,备用。
样品配制成溶液,备用。②滴定:利用发生反应:
 ,对上述配制的
,对上述配制的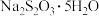 溶液进行滴定,终点时消耗标准溶液20.00 mL,则样品纯度为
溶液进行滴定,终点时消耗标准溶液20.00 mL,则样品纯度为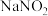 )是一种工业盐,外观与食盐非常相似,毒性较强。
)是一种工业盐,外观与食盐非常相似,毒性较强。I.某化学实验小组用如图所示装置(略去夹持仪器)制备亚硝酸钠。

已知:
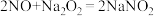 ;酸性条件下,NO、
;酸性条件下,NO、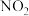 和
和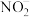 均能与
均能与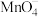 反应生成
反应生成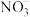 和
和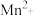 。
。(1)装置A中发生反应的化学方程式为
(2)装置B盛放的液体是
(3)
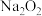 反应完全后,为测定产品纯度,该小组取4.00gD中的产品溶于水配成250mL溶液,取25.00mL溶液于锥形瓶中,用
反应完全后,为测定产品纯度,该小组取4.00gD中的产品溶于水配成250mL溶液,取25.00mL溶液于锥形瓶中,用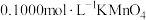 溶液进行滴定,实验所得数据如下表(假设)
溶液进行滴定,实验所得数据如下表(假设) 溶液不与杂质反应):
溶液不与杂质反应):| 滴定次数 | 1 | 2 | 3 | 4 |
消耗 溶液体积/mL 溶液体积/mL | 20.90 | 20.02 | 20.00 | 19.98 |
 溶液应使用
溶液应使用②第一次实验数据出现异常,造成这种异常的原因可能是
a.锥形瓶洗净后未干燥
b.滴定管用蒸馏水洗净后未用标准液润洗
c.滴定终点时俯视读数
d.放标准液的滴定管滴定前有气泡,滴定后气泡消失
③滴定终点的判断依据为
④该产品中亚硝酸钠的质量分数为
Ⅰ.配位-沉淀法制备
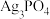 高效光催化剂
高效光催化剂
已知:i.Ag3PO4难溶于水,可溶于硝酸;
ii.Ag3PO4沉淀的生成速率会影响其结构和形貌,从而影响其光催化性能;
iii.银氨溶液中存在:
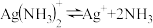
(1)配制银氨溶液时的反应现象是
(2)加入
 溶液时,发生以下反应,请将离子方程式补充完整:
溶液时,发生以下反应,请将离子方程式补充完整:□
 +□ +□ =□NH3+□ +□
+□ +□ =□NH3+□ +□ (3)
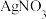 和
和 在溶液中反应也可制得Ag3PO4固体,但制得的Ag3PO4固体光催化性能极差。从速率角度解释其原因:
在溶液中反应也可制得Ag3PO4固体,但制得的Ag3PO4固体光催化性能极差。从速率角度解释其原因:Ⅱ.Ag3PO4光催化剂的使用和再生
已知:Ag3PO4晶体在光照条件下发挥催化作用时,首先引发反应。a.
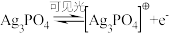
(4)Ag3PO4光催化CO2制备甲醇可实现“碳中和”,a的后续反应:
 ,
,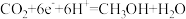 ,则由CO2制备甲醇的总反应的化学方程式为
,则由CO2制备甲醇的总反应的化学方程式为(5)Ag3PO4光催化降解RhB(代表有机污染物),RhB被氧化成CO2和H2O。a的后续反应
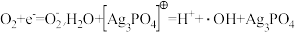
注:Ag3PO4在该催化过程中可能发生光腐蚀,生成单质银,影响其光催化性能。
用Ag3PO4依次降解三份相同的废水,测得3次降解过程中RhB的残留率(
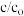 :即时浓度与起始浓度之比)随时间变化的曲线如图。
:即时浓度与起始浓度之比)随时间变化的曲线如图。
①下列说法正确的是
a.
 和
和 是降解RhB的重要氧化剂
是降解RhB的重要氧化剂b.第1次使用后Ag3PO4的光催化性能降低
c.该实验条件下,Ag3PO4使用两次即基本失效
②第1次光降解时,
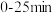 内的反应速率为
内的反应速率为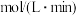 (废水中RhB初始浓度
(废水中RhB初始浓度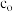 为
为 ,RhB的摩尔质量为
,RhB的摩尔质量为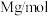 )
)
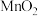 及少量Fe、Al、Si、Ca、Mg等元素的氧化物)为原料制备高纯
及少量Fe、Al、Si、Ca、Mg等元素的氧化物)为原料制备高纯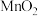 的实验步骤如下:
的实验步骤如下:(1)浸取。实验室浸取软锰矿的装置如图所示:

①为了提高软锰矿的浸出速率和浸取率,上述装置中采取的措施有
②仪器a为
③反应通常在70℃下反应,写出
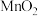 转化为
转化为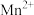 的离子方程式
的离子方程式(2)除杂。向已经除去铁、铝、硅元素的
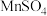 溶液(pH约为5)中加入
溶液(pH约为5)中加入 溶液,溶液中
溶液,溶液中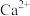 、
、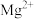 形成氟化物沉淀。但若pH太低,会显著增加NaF的用量,其原因为
形成氟化物沉淀。但若pH太低,会显著增加NaF的用量,其原因为(3)沉锰。向
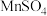 溶液中缓慢滴加
溶液中缓慢滴加 溶液,过滤、洗涤、干燥,得到
溶液,过滤、洗涤、干燥,得到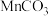 固体。解释生成
固体。解释生成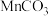 固体所需要的平衡理论有
固体所需要的平衡理论有a、
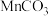 的沉淀溶解平衡 b、
的沉淀溶解平衡 b、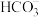 的水解平衡
的水解平衡c、
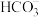 的电离平衡 d、
的电离平衡 d、 的电离平衡
的电离平衡(4)热解。将
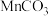 置于热解装置中,通入足量空气,加热到450℃。将固体冷却后研成粉末,边搅拌边加入一定量稀硫酸除去少量的MnO、
置于热解装置中,通入足量空气,加热到450℃。将固体冷却后研成粉末,边搅拌边加入一定量稀硫酸除去少量的MnO、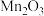 ,加热,充分反应后过滤、洗涤、干燥,即可获得高纯
,加热,充分反应后过滤、洗涤、干燥,即可获得高纯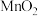 (已知:加热条件下
(已知:加热条件下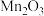 在酸性溶液中转化为
在酸性溶液中转化为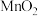 和
和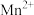 )。检验是否“洗涤”干净的操作方法为
)。检验是否“洗涤”干净的操作方法为Ⅰ. 一种以金属锌和选择性催化材料为电极的新型
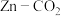 水介质电池的示意图如下:
水介质电池的示意图如下: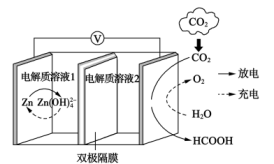
(1)放电时,电池正极发生的电极反应为
(2)充电时,电池负极附近溶液的

(3)充电时,电池总反应式为
(4)放电时,电路中每转移
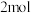 电子,迁移向负极区的水离解产物
电子,迁移向负极区的水离解产物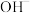 的数量为
的数量为Ⅱ. “湿法炼锌”是现代常用的冶锌方法,由锌浸出、酸锌溶液净化、锌电解沉积三大环节组成。电解环节中,电解液中较高浓度的
 会腐蚀阳极板而增大能耗。可向电解液中同时加入
会腐蚀阳极板而增大能耗。可向电解液中同时加入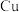 和
和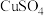 ,生成
,生成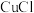 沉淀除去
沉淀除去 。
。
(5)除
 发生的离子反应方程式为
发生的离子反应方程式为(6)
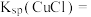
(7)反应
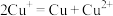 能否完全进行?
能否完全进行?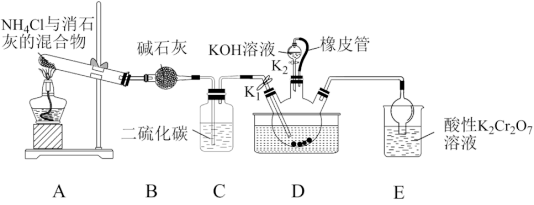
已知:①NH3不溶于CS2,CS2密度比水大且不溶于水;
②三颈烧瓶内盛放:CS2、水和催化剂;
③CS2+3NH3
 NH4SCN+NH4HS,该反应比较缓慢且NH4SCN在高于170℃易分解;
NH4SCN+NH4HS,该反应比较缓慢且NH4SCN在高于170℃易分解;④NH4HS在105℃就会完全分解。
回答下列问题:
(1)装置A中反应的化学方程式是
(2)装置D中橡皮管的作用是
(3)制备硫氰化钾晶体:先滤去三颈烧瓶中的固体催化剂,再减压
(4)测定晶体中KSCN的含量:称取6.0g样品,配成500mL溶液,量取25.00mL溶液于锥形瓶中,加入适量稀硝酸,再加入几滴Fe(NO3)3溶液作指示剂,用0.1000mol/L AgNO3标准溶液滴定,达到滴定终点时消耗AgNO3标准溶液18.00mL。[已知:滴定时发生反应的离子方程式:SCN-+Ag+=AgSCN↓(白色)]
①用AgNO3固体配制500mL 0.1000mol•L-1的AgNO3标准溶液所需的玻璃仪器有烧杯、量筒、玻璃棒、胶头滴管、
②滴定终点溶液颜色的变化
③晶体中KSCN的质量分数为
(1)晒制蓝图时,用K3[Fe(C2O4)3]·3H2O作感光剂,以K3[Fe(CN)6]溶液为显色剂。其光解反应的化学方程式为:2K3[Fe(C2O4)3]
 2FeC2O4+3K2C2O4+2CO2↑;显色反应的化学方程式为
2FeC2O4+3K2C2O4+2CO2↑;显色反应的化学方程式为(2)某小组为探究三草酸合铁酸钾的热分解产物,按下图所示装置进行实验。

①通入氮气的目的是
②实验中观察到装置B、F中澄清石灰水均变浑浊,装置E中固体变为红色,由此判断热分解产物中一定含有
③为防止倒吸,停止实验时应进行的操作是
④样品完全分解后,装置A中的残留物含有FeO和Fe2O3,检验Fe2O3存在的方法是:
(3)测定三草酸合铁酸钾中铁的含量。
①称量m g样品于锥形瓶中,溶解后加稀H2SO4酸化,用c mol·L-1 KMnO4溶液滴定至终点。滴定终点的现象是
②向上述溶液中加入过量锌粉至反应完全后,过滤、洗涤,将滤液及洗涤液全部收集到锥形瓶中。加稀H2SO4酸化,用c mol·L-1 KMnO4溶液滴定至终点,消耗KMnO4溶液V mL。该晶体中铁的质量分数的表达式为
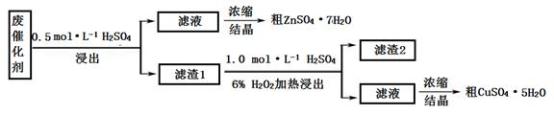
回答下列问题:
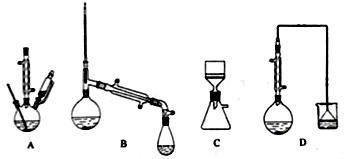
(1)在下列装置中,第一次浸出必须用
(2)第二次浸出时,向盛有滤液1的反应器中加入稀硫酸,后滴入过氧化氢溶液。若顺序相反,会造成
(3)浓缩硫酸锌、硫酸铜溶液使用的器皿名称是
(4)某同学在实验完成之后,得到1.5gCuSO4﹒5H2O,则铜的回收率为



