已知在448℃时,反应H2(g)+I2(g) 2HI(g)的平衡常数K1为49,则该温度下反应
2HI(g)的平衡常数K1为49,则该温度下反应 H2(g)+
H2(g)+ I2(g)
I2(g) HI(g)的平衡常数K2为
HI(g)的平衡常数K2为___ 。
 2HI(g)的平衡常数K1为49,则该温度下反应
2HI(g)的平衡常数K1为49,则该温度下反应 H2(g)+
H2(g)+ I2(g)
I2(g) HI(g)的平衡常数K2为
HI(g)的平衡常数K2为
更新时间:2020-12-24 19:03:53
|
【知识点】 化学平衡常数的概念及表达方式解读
相似题推荐
填空题
|
容易
(0.94)
【推荐1】已知:N2(g)+3H2(g) 2NH3(g)的平衡常数为K1,
2NH3(g)的平衡常数为K1, N2(g)+
N2(g)+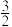 H2(g)
H2(g) NH3(g)的平衡常数为K2,NH3(g)
NH3(g)的平衡常数为K2,NH3(g)
 N2(g)+
N2(g)+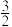 H2(g)的平衡常数为K3。
H2(g)的平衡常数为K3。
(1)写出K1和K2的关系式:_______ ;
(2)写出K2和K3的关系式:_______ ;
(3)写出K1和K3的关系式:_______ 。
 2NH3(g)的平衡常数为K1,
2NH3(g)的平衡常数为K1, N2(g)+
N2(g)+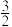 H2(g)
H2(g) NH3(g)的平衡常数为K2,NH3(g)
NH3(g)的平衡常数为K2,NH3(g)
 N2(g)+
N2(g)+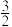 H2(g)的平衡常数为K3。
H2(g)的平衡常数为K3。(1)写出K1和K2的关系式:
(2)写出K2和K3的关系式:
(3)写出K1和K3的关系式:
您最近一年使用:0次
填空题
|
容易
(0.94)
解题方法
【推荐2】在一定温度下,已知以下三个反应的平衡常数:
反应①:CO(g)+CuO(s) CO2(g)+Cu(s) K1
CO2(g)+Cu(s) K1
反应②:H2(g)+CuO(s) Cu(s)+H2O(g) K2
Cu(s)+H2O(g) K2
反应③:CO(g)+H2O(g) CO2(g)+H2(g) K3
CO2(g)+H2(g) K3
(1)反应①的平衡常数表达式为___________ 。
(2)反应③的K3与K1、K2的关系是K3=___________ 。
反应①:CO(g)+CuO(s)
 CO2(g)+Cu(s) K1
CO2(g)+Cu(s) K1反应②:H2(g)+CuO(s)
 Cu(s)+H2O(g) K2
Cu(s)+H2O(g) K2反应③:CO(g)+H2O(g)
 CO2(g)+H2(g) K3
CO2(g)+H2(g) K3(1)反应①的平衡常数表达式为
(2)反应③的K3与K1、K2的关系是K3=
您最近一年使用:0次
填空题
|
容易
(0.94)
【推荐3】电离平衡常数和电离度(电离度是指已电离的电解质分子数占原来总分子数的百分比)均可衡量弱电解质的电离,下表是常温下几种弱酸的电离平衡常数( )和弱碱的电离平衡常数(
)和弱碱的电离平衡常数(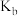 ),回答下列问题∶
),回答下列问题∶
(1) 的水溶液呈
的水溶液呈_______ (填“酸性”、“中性”或“碱性”),理由是_______ 。溶液中各离子浓度大小关系是________ 。
(2) 的电离平衡常数表达式
的电离平衡常数表达式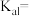
_______ ,现测得溶解了 的某盐酸的pH=4,则该溶液中
的某盐酸的pH=4,则该溶液中 的电离度约为
的电离度约为________ 。
(3)常温下,浓度相同的三种溶液NaF、 、
、 ,pH由大到小的顺序是
,pH由大到小的顺序是___________ 。
(4)将过量 通入
通入 溶液中,反应的离子方程式为
溶液中,反应的离子方程式为___________ 。
 )和弱碱的电离平衡常数(
)和弱碱的电离平衡常数(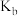 ),回答下列问题∶
),回答下列问题∶| 弱酸或弱碱 | HF | 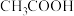 |  |  | 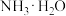 |
| 电离平衡常数 | 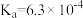 | 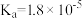 | 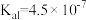 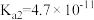 |  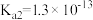 | 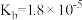 |
(1)
 的水溶液呈
的水溶液呈(2)
 的电离平衡常数表达式
的电离平衡常数表达式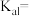
 的某盐酸的pH=4,则该溶液中
的某盐酸的pH=4,则该溶液中 的电离度约为
的电离度约为(3)常温下,浓度相同的三种溶液NaF、
 、
、 ,pH由大到小的顺序是
,pH由大到小的顺序是(4)将过量
 通入
通入 溶液中,反应的离子方程式为
溶液中,反应的离子方程式为
您最近一年使用:0次



