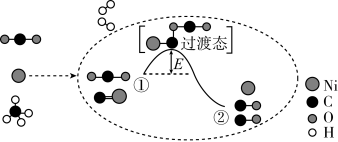
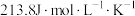已知:
CH3CH2CH2 +HCl
+HCl CH3CH2CH3+Cl
CH3CH2CH3+Cl
 CH(CH3)2+HCl
CH(CH3)2+HCl
CH3CH2CH2 +HBr
+HBr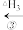 CH3CH2CH3+Br
CH3CH2CH3+Br
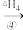 CH(CH3)2+HBr
CH(CH3)2+HBr
(以上微粒均为气体状态)
下列说法正确的是
CH3CH2CH2
 +HCl
+HCl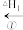 CH3CH2CH3+Cl
CH3CH2CH3+Cl
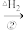 CH(CH3)2+HCl
CH(CH3)2+HClCH3CH2CH2
 +HBr
+HBr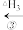 CH3CH2CH3+Br
CH3CH2CH3+Br
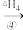 CH(CH3)2+HBr
CH(CH3)2+HBr (以上微粒均为气体状态)
下列说法正确的是
| A.△H1=△H2+△H3-△H4 |
| B.HCl和HBr的键能差可以表示为△H1-△H3 |
| C.△H2、△H4为放热过程,△H2>△H4 |
| D.升高温度,活化能降低,化学反应速率增大 |
更新时间:2020-11-13 10:48:15
|
相似题推荐
单选题
|
适中
(0.65)
【推荐1】相同温度和压强下,有如下热化学方程式,下列叙述正确的是
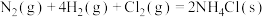

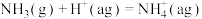

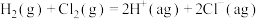
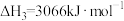

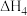

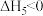
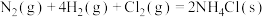

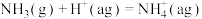

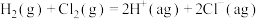
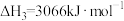

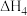

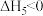
A.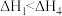 | B.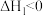 , ,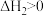 |
C.H-Cl键键能为1533 | D.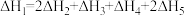 |
您最近一年使用:0次
【推荐2】实验室中利用固体KMnO4进行如图实验,下列说法正确的是
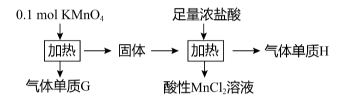

| A.本实验中若用稀盐酸代替浓盐酸会减慢反应速率,但不影响单质H的生成量 |
| B.气体H的量小于0.25mol |
| C.根据盖斯定律,直接用KMnO4和浓盐酸反应的热效应与本实验中的总热效应相等 |
| D.气体H是一种常见的氧化性气体,一般在反应中只表现氧化性 |
您最近一年使用:0次
单选题
|
适中
(0.65)
名校
【推荐1】下列说法中,正确的是
| A.降低温度能使化学反应速率减小,主要原因是降低了反应物分子中活化分子的百分数 |
B. 的反应均是自发进行的反应 的反应均是自发进行的反应 |
C.在恒容密闭容器中发生反应: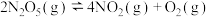 ,充入少量氩气,反应速率减小 ,充入少量氩气,反应速率减小 |
D.氢气燃烧热的热化学方程式为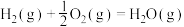 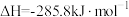 |
您最近一年使用:0次
单选题
|
适中
(0.65)
解题方法
【推荐2】已知反应:2NO(g) + Br2(g) 2NOBr(g) ΔH = -a kJ·mol-1(a﹥0),其反应机理如下:
2NOBr(g) ΔH = -a kJ·mol-1(a﹥0),其反应机理如下:
① NO(g) + Br2(g) NOBr2(g) 快
NOBr2(g) 快
② NO(g) + NOBr2(g) 2NOBr(g) 慢
2NOBr(g) 慢
下列有关该反应的说法不正确 的是
 2NOBr(g) ΔH = -a kJ·mol-1(a﹥0),其反应机理如下:
2NOBr(g) ΔH = -a kJ·mol-1(a﹥0),其反应机理如下:① NO(g) + Br2(g)
 NOBr2(g) 快
NOBr2(g) 快 ② NO(g) + NOBr2(g)
 2NOBr(g) 慢
2NOBr(g) 慢下列有关该反应的说法
| A.该反应的速率主要取决于②的快慢 |
| B.NOBr2是该反应的中间产物 |
| C.正反应的活化能比逆反应的活化能小a kJ·mol-1 |
| D.增大Br2(g)的浓度能增大活化分子百分数,加快反应速率 |
您最近一年使用:0次

 纯物质的规定熵,叫做标准摩尔熵,用符号
纯物质的规定熵,叫做标准摩尔熵,用符号 表示。相关数据如下表,通过数据分析得出的下列结论不正确的是
表示。相关数据如下表,通过数据分析得出的下列结论不正确的是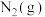
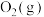

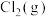

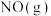
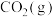
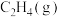
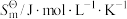
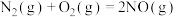 的标准摩尔熵变
的标准摩尔熵变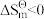
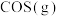 的标准摩尔熵大于
的标准摩尔熵大于