1 .  的反应机理如下:
的反应机理如下:
反应I:
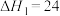 kJ·mol
kJ·mol
反应Ⅱ:
 kJ·mol
kJ·mol
反应中的能量变化如图所示,下列说法不正确 的是

 的反应机理如下:
的反应机理如下:反应I:

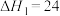 kJ·mol
kJ·mol
反应Ⅱ:

 kJ·mol
kJ·mol
反应中的能量变化如图所示,下列说法

A.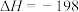 kJ·mol kJ·mol |
B.反应I的 |
C.通入过量空气,可提高 的平衡转化率 的平衡转化率 |
| D.反应速率由反应Ⅱ决定 |
您最近一年使用:0次



