 时,用氨水分别滴定浓度均为
时,用氨水分别滴定浓度均为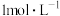 的
的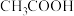 、
、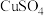 、
、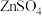 三种溶液,
三种溶液,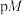 随
随 的变化关系如图所示[
的变化关系如图所示[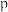 表示负对数,
表示负对数, 表示
表示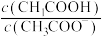 、
、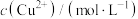 、
、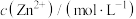 等],已知
等],已知 ,溶液中离子浓度小于
,溶液中离子浓度小于 可以认为已经除去。下列有关分析错误的是
可以认为已经除去。下列有关分析错误的是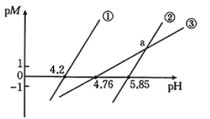
A. 时, 时, |
B. 点对应的 点对应的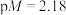 |
C.可通过调节 的方法除尽 的方法除尽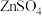 溶液(浓度为 溶液(浓度为 )中含有的 )中含有的 |
D.氨水过量时,均可形成 ( (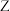 表示 表示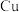 或 或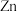 ) ) |
更新时间:2024-05-30 20:12:33
|
相似题推荐
【推荐1】常温时,三种金属硫化物在水中的沉淀溶解平衡曲线如图所示。已知pA=-lgc(A)。下列说法正确的是
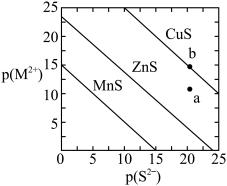

| A.常温下,三种物质中CuS的溶解度最小 |
| B.常温下,Ksp(MnS)=10-30 |
| C.加热可使b点移动到a点 |
| D.若要除去ZnCl2溶液中混有的少量MnCl2,则可加入CuS固体 |
您最近一年使用:0次
【推荐2】向AgBr饱和溶液中滴加Na2S2O3溶液,发生反应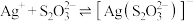 和
和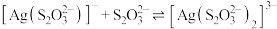 ,
,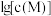 、
、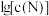 与
与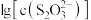 的关系如下图所示(其中M代表Ag+或Br-;N代表
的关系如下图所示(其中M代表Ag+或Br-;N代表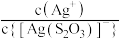 或
或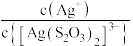 );
);
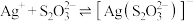 和
和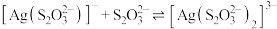 ,
,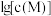 、
、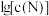 与
与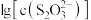 的关系如下图所示(其中M代表Ag+或Br-;N代表
的关系如下图所示(其中M代表Ag+或Br-;N代表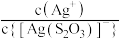 或
或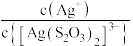 );
);
A.直线L2表示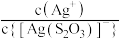 随 随 浓度变化的关系 浓度变化的关系 |
B.AgBr的溶度积常数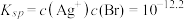 |
C.反应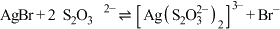 的平衡常数K的数量级为102 的平衡常数K的数量级为102 |
D. 时,溶液中 时,溶液中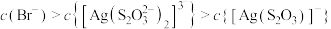 |
您最近一年使用:0次
单选题
|
较难
(0.4)
【推荐1】25℃时部分电解质的电离平衡常数数据如下。
已知 ,
, 。下列说法错误的是
。下列说法错误的是
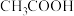 |  | HClO | 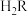 |
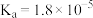 | 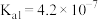 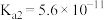 | 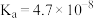 | 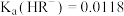 |
 ,
, 。下列说法错误的是
。下列说法错误的是A.等浓度的NaClO和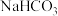 混合溶液中存在 混合溶液中存在 |
B.等浓度溶液的 |
C.向NaClO溶液中通入少量 ,漂白性增强的原因是 ,漂白性增强的原因是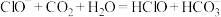 |
D.NaHR溶液中既存在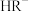 的电离也存在 的电离也存在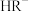 的水解 的水解 |
您最近一年使用:0次
单选题
|
较难
(0.4)
名校
解题方法
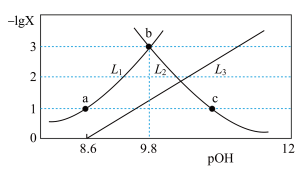
【推荐2】常温下,向一定浓度邻苯二甲酸钠(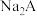 表示)溶液中通入HCl气体,保持溶液体积和温度不变,测得
表示)溶液中通入HCl气体,保持溶液体积和温度不变,测得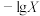 与pOH[X为
与pOH[X为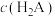 、
、 、
、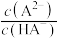 ;
;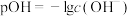 ]的变化关系如图所示。下列说法正确的是
]的变化关系如图所示。下列说法正确的是
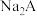 表示)溶液中通入HCl气体,保持溶液体积和温度不变,测得
表示)溶液中通入HCl气体,保持溶液体积和温度不变,测得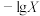 与pOH[X为
与pOH[X为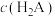 、
、 、
、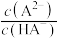 ;
;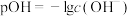 ]的变化关系如图所示。下列说法正确的是
]的变化关系如图所示。下列说法正确的是
A.曲线L₁表示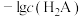 |
B.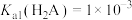 |
| C.水的电离程度:b>a>c |
D.c点溶液中: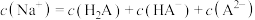 |
您最近一年使用:0次
单选题
|
较难
(0.4)
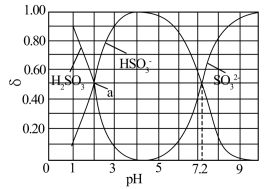
【推荐3】边搅拌边向Na2CO3溶液中通入SO2制备NaHSO3溶液,水溶液中H2SO3、HSO 、SO
、SO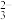 的分布系数δ随pH的变化关系如图所示。[比如H2SO3的分布系数: δ (H2SO3) =
的分布系数δ随pH的变化关系如图所示。[比如H2SO3的分布系数: δ (H2SO3) = ],下列有关叙述错误的是
],下列有关叙述错误的是
 、SO
、SO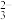 的分布系数δ随pH的变化关系如图所示。[比如H2SO3的分布系数: δ (H2SO3) =
的分布系数δ随pH的变化关系如图所示。[比如H2SO3的分布系数: δ (H2SO3) = ],下列有关叙述错误的是
],下列有关叙述错误的是
| A.当溶液pH范围为4~5时,停止通入SO2 |
| B.根据曲线数据计算可知Ka2(H2SO3)的数量级为10-7 |
C.a点的溶液中c(H+)+c(Na+)=c(OH—)+c( HSO )+2c(SO )+2c(SO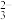 )+c( HCO )+c( HCO ) +2c(CO ) +2c(CO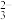 ) ) |
D.pH=3的溶液中,c(Na+)<c(HSO )+c(H2SO3) )+c(H2SO3) |
您最近一年使用:0次

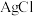 和
和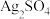 的沉淀溶解平衡曲线如图所示。
的沉淀溶解平衡曲线如图所示。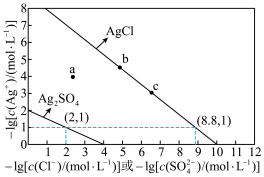
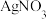 固体可使溶液由
固体可使溶液由 点变到
点变到 点
点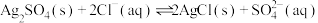 的平衡常数很大,反应趋于完全
的平衡常数很大,反应趋于完全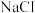 、
、 均为
均为 的混合溶液中滴加
的混合溶液中滴加

