名校
1 . 将 催化转化成CO是实现
催化转化成CO是实现 资源化利用的关键步骤,发生的反应有
资源化利用的关键步骤,发生的反应有
反应Ⅰ:

反应Ⅱ: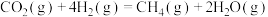

反应Ⅲ: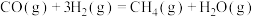
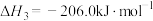 ,0.1MPa,在密闭容器中按
,0.1MPa,在密闭容器中按 投料,平衡时各组分的摩尔分数[物质i的摩尔分数:
投料,平衡时各组分的摩尔分数[物质i的摩尔分数: ,
, 未计入]随温度的变化如图。下列说法
未计入]随温度的变化如图。下列说法不正确 的是
 催化转化成CO是实现
催化转化成CO是实现 资源化利用的关键步骤,发生的反应有
资源化利用的关键步骤,发生的反应有反应Ⅰ:


反应Ⅱ:
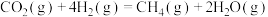

反应Ⅲ:
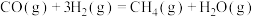
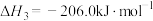 ,0.1MPa,在密闭容器中按
,0.1MPa,在密闭容器中按 投料,平衡时各组分的摩尔分数[物质i的摩尔分数:
投料,平衡时各组分的摩尔分数[物质i的摩尔分数: ,
, 未计入]随温度的变化如图。下列说法
未计入]随温度的变化如图。下列说法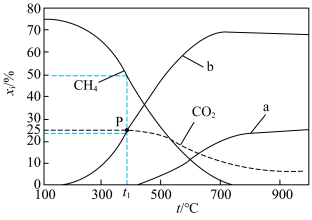
| A.低温下有利于反应Ⅱ正向进行 | B.曲线b为 |
C.P点时反应Ⅰ的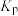 约为 约为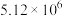 | D.900℃时,适当增大体系压强,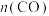 保持不变 保持不变 |
您最近一年使用:0次
2024-04-16更新
|
170次组卷
|
2卷引用:2024届福建省部分地市高三下学期三模化学试题
名校
解题方法
2 . 工业上制备Ti,采用碳氯化法将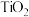 转化成
转化成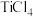 。在1000℃时发生如下:
。在1000℃时发生如下:
①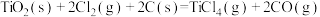
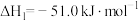
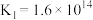
②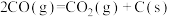
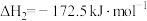
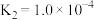
③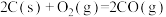
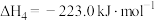
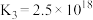 。
。
在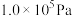 ,将
,将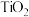 、C、
、C、 以物质的量比1:2.2:2进行碳氯化,平衡时体系中
以物质的量比1:2.2:2进行碳氯化,平衡时体系中 、CO、
、CO、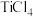 和C的组成比(物质的量分数)随温度变化如下图所示。下列说法不正确的是
和C的组成比(物质的量分数)随温度变化如下图所示。下列说法不正确的是
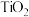 转化成
转化成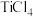 。在1000℃时发生如下:
。在1000℃时发生如下:①
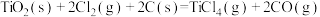
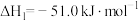
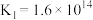
②
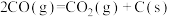
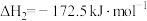
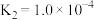
③
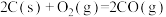
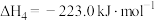
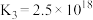 。
。在
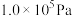 ,将
,将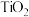 、C、
、C、 以物质的量比1:2.2:2进行碳氯化,平衡时体系中
以物质的量比1:2.2:2进行碳氯化,平衡时体系中 、CO、
、CO、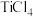 和C的组成比(物质的量分数)随温度变化如下图所示。下列说法不正确的是
和C的组成比(物质的量分数)随温度变化如下图所示。下列说法不正确的是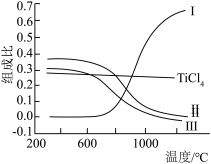
A.1000℃时,反应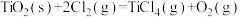 的平衡常数 的平衡常数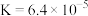 |
B.曲线III表示平衡时 的物质的量分数随温度的变化 的物质的量分数随温度的变化 |
| C.高于600℃,升高温度,主要对反应②的平衡产生影响 |
D.为保证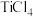 的平衡产率,选择反应温度应高于1000℃ 的平衡产率,选择反应温度应高于1000℃ |
您最近一年使用:0次
2023-03-23更新
|
1499次组卷
|
5卷引用:江苏省苏锡常镇四市2022-2023学年高三下学期教学情况调研(一)(3月)化学试题
江苏省苏锡常镇四市2022-2023学年高三下学期教学情况调研(一)(3月)化学试题(已下线)专题14 化学反应速率和化学平衡江苏省郑梁梅高级中学2022-2023学年高二下学期4月月考化学试题河北省衡水中学2023-2024学年高三上学期四调考试 化学试题(已下线)FHgkyldyjhx08
名校
解题方法
3 .  联合
联合 和
和 制取
制取 时,发生的主要反应如下:
时,发生的主要反应如下:
①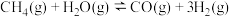

②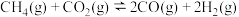
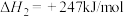
将 、
、 和
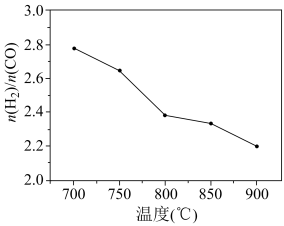
和 按一定比例通入填充有催化剂的恒容反应器,在不同温度下,反应相同时间内(反应均未达到化学平衡状态)测得
按一定比例通入填充有催化剂的恒容反应器,在不同温度下,反应相同时间内(反应均未达到化学平衡状态)测得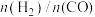 的值如下图所示。
的值如下图所示。
下列说法不正确 的是
 联合
联合 和
和 制取
制取 时,发生的主要反应如下:
时,发生的主要反应如下:①
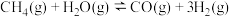

②
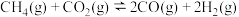
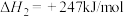
将
 、
、 和
和 按一定比例通入填充有催化剂的恒容反应器,在不同温度下,反应相同时间内(反应均未达到化学平衡状态)测得
按一定比例通入填充有催化剂的恒容反应器,在不同温度下,反应相同时间内(反应均未达到化学平衡状态)测得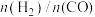 的值如下图所示。
的值如下图所示。
下列说法
| A.若反应足够长时间,①与②同时达到化学平衡状态 |
B.由①②可知,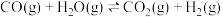 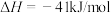 |
| C.其他条件不变时升高温度,反应①的正反应速率增大、逆反应速率减小 |
D.其他条件不变时,增大原料中 的浓度可以提高产物中 的浓度可以提高产物中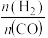 的值 的值 |
您最近一年使用:0次
名校
解题方法
4 . 乙烯是重要的工业原材料,中科院设计并完成了 催化氧化
催化氧化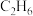 制取
制取 的新路径,
的新路径,
其主要反应为:反应Ⅰ: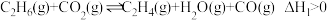
反应Ⅱ: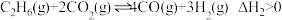
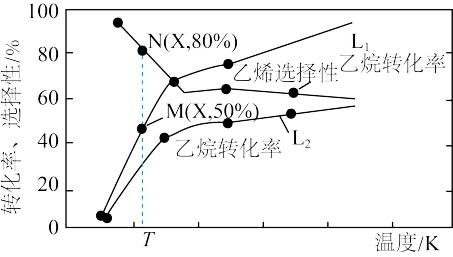
一定条件下,向容积为 的容器中通入
的容器中通入 和
和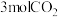 ,若仅考虑上述反应,
,若仅考虑上述反应,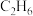 平衡转化率和平衡时
平衡转化率和平衡时 的选择性随温度、压强的关系如图所示,
的选择性随温度、压强的关系如图所示,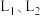 代表不同压强下乙烷的平衡转化率。
代表不同压强下乙烷的平衡转化率。
已知:
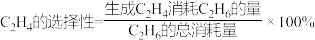
下列说法正确的是
 催化氧化
催化氧化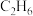 制取
制取 的新路径,
的新路径,其主要反应为:反应Ⅰ:
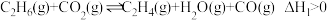
反应Ⅱ:
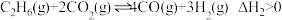
一定条件下,向容积为
 的容器中通入
的容器中通入 和
和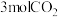 ,若仅考虑上述反应,
,若仅考虑上述反应,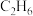 平衡转化率和平衡时
平衡转化率和平衡时 的选择性随温度、压强的关系如图所示,
的选择性随温度、压强的关系如图所示,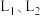 代表不同压强下乙烷的平衡转化率。
代表不同压强下乙烷的平衡转化率。
已知:
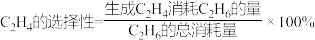
下列说法正确的是
A.压强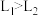 |
B.压强为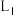 、温度为T时,反应达平衡时,混合气体中 、温度为T时,反应达平衡时,混合气体中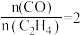 |
C.一定温度下,增大 可提高 可提高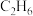 的平衡转化率 的平衡转化率 |
D.反应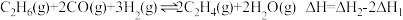 |
您最近一年使用:0次
2022-11-10更新
|
853次组卷
|
5卷引用:江苏省镇江市2022-2023学年高三上学期期中调研考试化学试题



