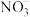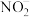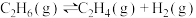
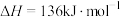 。
。(1)一定温度下,在乙烷热裂解制取乙烯时,向体系中充入一定量惰性气体,保持体系的总压强为100kPa,测得平衡时各组分的体积分数如下表所示:
| 物质 | 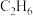 |  |  |
| 体积分数 | 5% | 20% | 20% |
(2)利用膜分离技术可以实现边反应边分离出生成的
 。在容积为1L的恒容密闭容器中充入1mol乙烷,测得不同氢气移出率α[
。在容积为1L的恒容密闭容器中充入1mol乙烷,测得不同氢气移出率α[ ]条件下,乙烷的平衡转化率与和温度的关系如图-1所示。
]条件下,乙烷的平衡转化率与和温度的关系如图-1所示。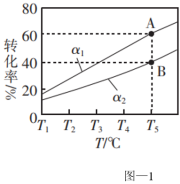
①α1
②若A点时平衡常数K=0.8,则α1=
(3)以
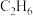 和
和 为原料可以协同制取
为原料可以协同制取 和CO,发生的反应如下:
和CO,发生的反应如下:反应1:
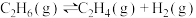
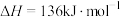
反应2:

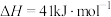
①已知
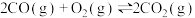
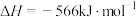 。
。则
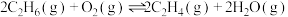 的
的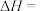
 。
。②0.1MPa时,按物质的量之比为1:1向密闭容器中充入
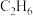 和
和 的混合气体,反应相同时间,测得
的混合气体,反应相同时间,测得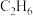 和
和 的转化率与温度的关系如图-2所示。X代表的物质是
的转化率与温度的关系如图-2所示。X代表的物质是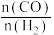 的值的变化情况是
的值的变化情况是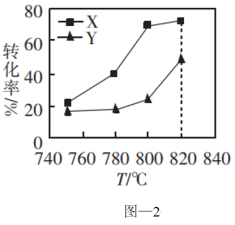
③乙烷热裂解时,会产生积碳,以
 和
和 为原料协同制取
为原料协同制取 和CO会减少积碳的产生,原因是
和CO会减少积碳的产生,原因是相似题推荐
(1)采用NaClO2溶液作为吸收剂可进行脱硝。反应一段时间后溶液中离子浓度的分析结果如下表:
离子 |
|
|
|
c/(mol·L-1) | 1.5×10-4 | 1.2×10-6 | 3.4×10-3 |
(2)用焦炭还原法将其转化为无污染的物质也可进行脱硝。已知:
2C(s)+O2(g)=2CO(g) △H1=-221.0kJ·mol-1;
N2(g)+O2(g)=2NO(g) △H2=+180.5kJ/mol
2NO(g)+2CO(g)=2CO2(g)+N2 (g) △H3=-746.0kJ /mol
回答下列问题
①用焦炭还原NO生成无污染气体的热化学方程式为
②在一定温度下,向甲、乙、丙三个恒容密闭容器中加入一定量的NO和足量的焦炭,反应过程中测得各容器中c(NO)(mo/L)随时间(s)的变化如下表:(已知:三个容器的反应温度分别为T甲=400℃、T乙=400℃、T丙=a℃)
时间 | 0s | 10s | 20s | 30s | 40s |
甲 | 2.00 | 1.50 | 1.10 | 0.80 | 0.80 |
乙 | 1.00 | 0.80 | 0.65 | 0.53 | 0.45 |
丙 | 2.00 | 1.45 | 1.00 | 1.00 | 1.00 |
(3)用NH3催化还原NOx,也可以消除氮氧化物的污染。已知:8NH3(g)+6NO2(g)
 7N2(g)+12H2O(l),ΔH<0。相同条件下,在2L密闭容器内,选用不同的催化剂进行反应,产生N2的量随时间的变化如图所示
7N2(g)+12H2O(l),ΔH<0。相同条件下,在2L密闭容器内,选用不同的催化剂进行反应,产生N2的量随时间的变化如图所示
①在催化剂A的作用下0-4min的v(NO2)=
②该反应活化能Ea(A)、Ea(B)、Ea(C)由小到大的顺序是
③下列说法不正确的是
a.使用催化剂A达平衡时,△H值更大
b升高温度可使容器内气体颜色加深
c.单位时间内形成N-H键与O-H键的数目相等时,说明反应已经达到平衡
d.若在恒容绝热的密闭容器中反应,当平衡常数不变时,说明反应已经达到平衡
(1)Deacon发明的直接氧化法为:4HCl(g)+O2(g)
 2Cl2(g)+2H2O(g)。下图为刚性容器中,进料浓度比c(HCl)∶c(O2)分别等于1∶1、4∶1、7∶1时HCl平衡转化率随温度变化的关系:
2Cl2(g)+2H2O(g)。下图为刚性容器中,进料浓度比c(HCl)∶c(O2)分别等于1∶1、4∶1、7∶1时HCl平衡转化率随温度变化的关系:
可知反应平衡常数K(300℃)
(2)Deacon直接氧化法可按下列催化过程进行:
CuCl2(s)=CuCl(s)+
 Cl2(g) ΔH1=+83 kJ·mol-1
Cl2(g) ΔH1=+83 kJ·mol-1CuCl(s)+
 O2(g)=CuO(s)+
O2(g)=CuO(s)+ Cl2(g) ΔH2=-20 kJ·mol-1
Cl2(g) ΔH2=-20 kJ·mol-1CuO(s)+2HCl(g)=CuCl2(s)+H2O(g) ΔH3=-121 kJ·mol-1
则4HCl(g)+O2(g)=2Cl2(g)+2H2O(g)的ΔH=
①


②


③

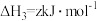
(1)(NH4)Fe3(SO4)2(OH)6净水时
(2)反应
 的△H=
的△H=(3)一定温度下,在恒容密闭容器中加入一定量的(NH4)Fe3(SO4)2(OH)6(s),只发生反应:
 ,下列情况表明该反应达到平衡状态的是___________(填标号)。
,下列情况表明该反应达到平衡状态的是___________(填标号)。| A.混合气体密度不随时间变化 | B.SO3体积分数不随时间变化 |
| C.气体平均摩尔质量不随时间变化 | D.Fe2O3质量不随时间变化 |
 ,达到平衡时测得
,达到平衡时测得 。保持温度不变,将容器体积变为原来的一半,达到新平衡时c(H2O)=
。保持温度不变,将容器体积变为原来的一半,达到新平衡时c(H2O)=(5)一定温度下,总压强恒定为8ρkPa时,向体积可变的密闭目容器中加入一定量的2Fe2O3·Fe2(SO4)3·(NH4)2SO4(s)和nmol氩气,仅发生反应②。测得平衡状态下n(NH3)与n(Ar)的关系如图所示。随着通入Ar的量增大,NH3的量增大的原因是

提示:用分压计算的平衡常数为KP,分压=总压X气体物质的量分数。
(6)(NH4)Fe3(SO4)2(OH)6失重时固体质量与温度的关系如图所示(变化过程中铁元素化合价不变)。a→b过程只失去一种气体,该气体是
(1)在中和反应反应热的测定实验中,为得到较准确的数值,适合用来做实验的一组试剂为
A.
 盐酸+适量
盐酸+适量 固体
固体B.
 盐酸
盐酸 溶液
溶液C.适量浓硫酸
 溶液
溶液(2)用下图甲、乙、丙三个容积相同的烧瓶收集等量的NO2气体,完成温度对化学平衡影响的实验,将操作步骤、预期现象和结论填入下表:(可以不填满)
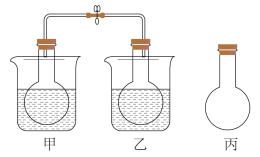
| 实验步骤与操作 | 预期现象 | 结论 |
| 当三只相同烧瓶中气体的颜色相同时,用弹簧夹夹紧甲、乙烧瓶间的橡皮管 | 不填 | 不填 |
| 同时将甲、乙两只烧瓶放入盛有冰水热水的两只烧杯中 | 放入热水中的烧瓶气体颜色比丙中的 | |
| 放入冰水中的烧瓶气体颜色比丙中的 |
(3)为比较Fe3+、Cu2+对H2O2分解的催化效果,某实验小组同学设计了如图所示的实验。
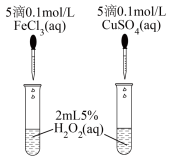
通过观察
(4)某实验小组欲探究某一外界条件对酸性KMnO4和H2C2O4(草酸)反应速率的影响,设计实验方案如下:
| 实验序号 | 草酸溶液 | 酸性高锰酸钾溶液 |
| ① |  、 、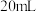 |  、 、 |
| ② | 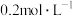 、 、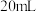 |  、 、 |
(1)已知反应
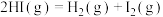
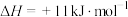 ,1mol
,1mol  (g)、1mol
(g)、1mol 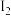 (g)分子中化学键断裂时分别需要吸收436 kJ、151 kJ的能量,则1mol HI(g)分子中化学键断裂时需吸收的能量为
(g)分子中化学键断裂时分别需要吸收436 kJ、151 kJ的能量,则1mol HI(g)分子中化学键断裂时需吸收的能量为(2)Bodensteins研究了下列反应:
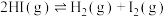 。在716K时,气体混合物中碘化氢的物质的量分数x(HI)与反应时间t的关系如表:
。在716K时,气体混合物中碘化氢的物质的量分数x(HI)与反应时间t的关系如表:| t/min | 0 | 20 | 40 | 60 | 80 | 120 | 160 |
| x(HI) | 1 | 0.91 | 0.85 | 0.815 | 0.795 | 0.784 | 0.784 |
②上述反应中,正反应速率为
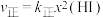 ,逆反应速率为
,逆反应速率为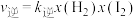 ,其中
,其中 、
、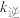 为速率常数,则
为速率常数,则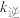 为
为 表示)。
表示)。③由上述实验数据计算得到
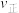 ~x(HI)和
~x(HI)和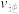 ~
~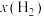 的关系可用下图表示。当升高到某一温度时,反应重新达到平衡,相应的点分别为
的关系可用下图表示。当升高到某一温度时,反应重新达到平衡,相应的点分别为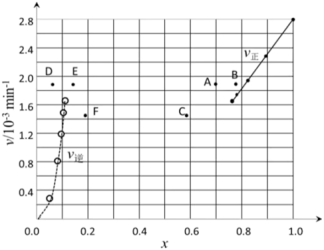
 催化加氢合成甲醇是重要的碳捕获利用与封
催化加氢合成甲醇是重要的碳捕获利用与封存技术,该过程主要发生下列反应:
反应①
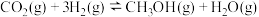

反应②
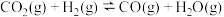
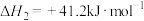
回答下列问题:
(1)CO继续加氢生成气态甲醇的热化学方程式为
(2)①现向2L恒容密闭容器中加入
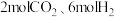 ,在恒温下发生上述反应。10s后反应达到平衡,此时容器内
,在恒温下发生上述反应。10s后反应达到平衡,此时容器内 的浓度为
的浓度为 ,CO为
,CO为 。则前10s内的平均反应速率
。则前10s内的平均反应速率
②若恒温恒容条件下只发生反应①,起始投料比
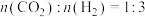 ,下列叙述能说明反应①达到平衡状态的是
,下列叙述能说明反应①达到平衡状态的是a.单位时间内消耗
 的同时生成
的同时生成
b.1mol
 生成的同时有3molH—H键断裂
生成的同时有3molH—H键断裂c.
 和
和 的浓度保持不变
的浓度保持不变d.容器内压强保持不变
e.
 和
和 的物质的量之比保持不变
的物质的量之比保持不变(3)
 催化加氢制甲醇反应历程中某一步基元反应的Arrhenius经验公式的实验数据如图1所示,已知Arrhenius经验公式为
催化加氢制甲醇反应历程中某一步基元反应的Arrhenius经验公式的实验数据如图1所示,已知Arrhenius经验公式为 (其中
(其中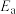 为活化能,
为活化能,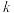 为速率常数,
为速率常数,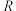 和
和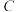 为常数)。该反应的活化能
为常数)。该反应的活化能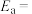
 。
。
(4)0.5MPa下,将
 的混合气体以一定流速通过装有催化剂的反应器,测得
的混合气体以一定流速通过装有催化剂的反应器,测得 的转化率、
的转化率、 或CO的选择性[
或CO的选择性[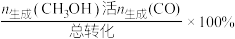 ]以及
]以及 的产率(
的产率( 的转化率×
的转化率× 的选择性)随温度的变化如图2所示。
的选择性)随温度的变化如图2所示。
曲线a表示
 ”或“CO”)的选择性随温度的变化,270℃时,对应
”或“CO”)的选择性随温度的变化,270℃时,对应 的转化率为
的转化率为 的产率增大的原因是
的产率增大的原因是(1)汽车气缸中发生复杂的化学反应,其中有如下三个重要反应:
反应1:N2(g)+O2(g)
 2NO(g) △H=+180.5kJ/mol
2NO(g) △H=+180.5kJ/mol反应2:2CO(g)
 2C(s)+O2(g) △H=+221 kJ/mol
2C(s)+O2(g) △H=+221 kJ/mol反应3:C(s)+O2(g)
 CO2(g) △H=-393.5 kJ/mol
CO2(g) △H=-393.5 kJ/mol①反应2NO(g)+2CO(g)
 N2(g)+2CO2(g)的△H=
N2(g)+2CO2(g)的△H=②下列有关反应1的说法中正确的是
A.反应物的总键能小于生成物的总键能
B.正反应的活化能大于逆反应的活化能
C.使用催化剂可使反应1的反应热减小
D.其它条件不变,加压,正逆反应速率不变,平衡不移动
③定温定容条件下,向容积为1L的密闭容器中充入一定量的NO和CO的混合气体(总物质的量保持4mol),平衡时NO、CO的转化率及N2的体积分数与
 的关系如图所示,CO转化率对应的曲线为
的关系如图所示,CO转化率对应的曲线为
(2)1073K时,H2与NO可发生反应2NO(g)+2H2(g)
 N2(g)+2H2O(g),该反应的正反应速率方程为:v=kca(H2)×cb(NO)(a、b分别为H2和NO的级数,k为速率常数)。
N2(g)+2H2O(g),该反应的正反应速率方程为:v=kca(H2)×cb(NO)(a、b分别为H2和NO的级数,k为速率常数)。在定容容器中,分别充入不同浓度的NO和H2,测得该反应的正反应速率如下表所示:
| 实验编号 | c(H2)/mol·L-1 | c(NO)/mol·L-1 | v/mol·L-1·s-1 |
| 1 | 0.0060 | 0.0010 | 7.9×10-7 |
| 2 | 0.0060 | 0.0020 | 3.2×10-6 |
| 3 | 0.0060 | 0.0040 | 1.3×10-5 |
| 4 | 0.0030 | 0.0040 | 6.4×10-6 |
| 5 | 0.0015 | 0.0040 | 3.2×10-6 |
【推荐2】甲醇是一种可再生能源,具有开发和应用的广阔前景,用Pt/Al2O3、Pd/C、Rh/SiO2作催化剂都可以采用如下反应来合成甲醇:2H2(g)+CO(g) CH3OH(g)
CH3OH(g)
(1)下表所列数据是各化学键的键能:
化学键 | H-H | C=O | C-H | C-O | O-H |
键能/(kJ●mol-1) | a | b | c | d | e |
则反应2H2(g)+CO(g) CH3OH(g)的△H=
CH3OH(g)的△H=
(2)三种不同催化剂作用时,CO的转化率如图1所示,则最适合作催化剂的是

(3)某化学研究性学习小组模拟工业合成甲醇的反应,在2 L的恒容密闭容器内充入2 mol H2和1molCO,加入合适催化剂后在某温度下开始反应,并用压力计监测容器内压强的变化如下:
反应时间/min | 0 | 5 | 10 | 15 | 20 | 25 |
压强/MPa | 12.6 | 10.8 | 9.5 | 8.7 | 8.4 | 8.4 |
则从反应开始到20 min时,CO的平均反应速率为
(4)近日,美国Neah和Novellusnn公司宣布,两家公司合作开发了多孔硅甲醇直接燃料电池,其工作原理如图所示。

①石墨2为
②石墨1极发生的电极反应式为
(1)如图是通过热化学循环在较低温度下由水或硫化氢分解制备氢气的反应系统原理。

通过计算,可知系统(Ⅰ)和系统(Ⅱ)制氢的热化学方程式分别为
(2)H2S与CO2在高温下发生反应:H2S(g)+CO2(g)
 COS(g)+H2O(g)。在610K时,将0.10molCO2与0.40molH2S充入2.5L的空钢瓶中,反应平衡后水的物质的量分数为0.02。
COS(g)+H2O(g)。在610K时,将0.10molCO2与0.40molH2S充入2.5L的空钢瓶中,反应平衡后水的物质的量分数为0.02。①H2S的平衡转化率α1=
②在620K重复试验,平衡后水的物质的量分数为0.03,H2S的转化率α2
③向反应器中再分别充入下列气体,能使H2S转化率增大的是
A.H2S B.CO2 C.COS D.N2
 N2(g)+2CO2(g)。
N2(g)+2CO2(g)。(1)上述反应的熵变ΔS
(2)假设在密闭容器中发生上述反应,达到平衡时下列措施能提高NO转化率的是
A.选用更有效的催化剂 B.升高反应体系的温度
C.降低反应体系的温度 D.缩小容器的体积
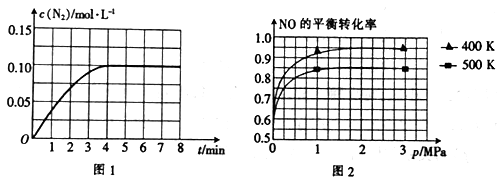
(3)若将1 molNO和2 mol CO通入2 L的恒容密闭容器中,在一定条件下发生上述反应,反应中生成的N2的物质的量浓度随时间的变化情况如图1所示。则NO从反应开始到平衡时的平均反应速率v(NO)=
(4)已知上述反应中NO的平衡转化率与压强、温度的关系如图2所示。工业上催化装置比较适合的温度和压强是
(5)研究表明:在使用等质量催化剂时,增大催化剂比表面积可提高化学反应速率。为了验证温度、催化剂比表面积对化学反应速率的影响规律,某同学设计了以下三组实验:
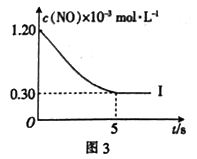
| 实验编号 | T(K) | NO的初始浓度 (mol·L-1) | CO的初始浓度 (mol·L-1) | 催化剂的比表面积 (m2·g-1) |
| I | 400 | 1.20×10-3 | 3.80×10-3 | 75 |
| Ⅱ | 400 | 1.20×10-3 | 3.80×10-3 | 100 |
| Ⅲ | 450 | 1.20×10-3 | 3.80×10-3 | 100 |
请在图3中分别画出上表中实验Ⅱ、Ⅲ条件下混合气体中NO的浓度随时间变化的曲线,并标明各条曲线的实验编号
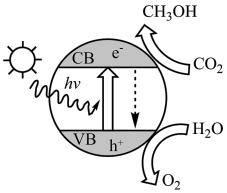
(1)如图为光催化转化二氧化碳的示意图,写出二氧化碳转化为甲醇的化学方程式
 :CO2(g)+3H2(g)
:CO2(g)+3H2(g) CH3OH(g)+H2O(g)
CH3OH(g)+H2O(g) 

反应
 :CO2(g)+H2(g)
:CO2(g)+H2(g) CO(g)+H2O(g)
CO(g)+H2O(g) 

试写出一氧化碳和氢气合成甲醇的热化学方程式
(3)
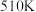 下,在压强为
下,在压强为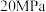 的恒压容器中通入
的恒压容器中通入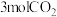 和
和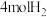 ,让其发生反应
,让其发生反应 后达到平衡,此时
后达到平衡,此时 的转化率为
的转化率为 ,则达到平衡时甲醇的物质的量分数为
,则达到平衡时甲醇的物质的量分数为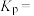
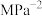 (保留
(保留 位有效数字
位有效数字 。
。(4)下图为在一定压强下二氧化碳与氢气合成甲醇时,二氧化碳的转化率和甲醇的选择性随温度的变化示意图:
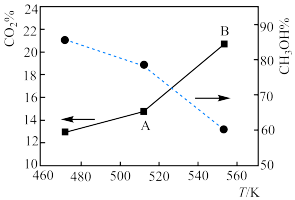
 随着温度的升高,二氧化碳的转化率升高而甲醇的选择性下降的原因是
随着温度的升高,二氧化碳的转化率升高而甲醇的选择性下降的原因是 对于反应
对于反应 来说,
来说, 点平衡常数
点平衡常数 填“
填“ ”“
”“ ”或者“
”或者“ ”
” 点平衡常数。
点平衡常数。(5)下图为在
 溶液中,通过电解的方法将二氧化碳转化为甲酸的机理图,试写出阴极的电极方程式
溶液中,通过电解的方法将二氧化碳转化为甲酸的机理图,试写出阴极的电极方程式
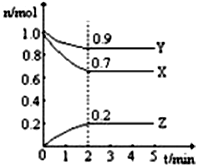
①该反应的化学方程式为
②从开始至2min,Z的平均反应速率为
③2min时X的转化率为
(2)在密闭容器中的一定量混合气体发生反应:xA(g)+yB(g)
 zC(g),平衡时测得A 的浓度为0.3 mol/L,保持温度不变,将容器的容积扩大到原来的两倍,再达到平衡时,测得A的浓度降低为0.18mol/L,则A 的转化率
zC(g),平衡时测得A 的浓度为0.3 mol/L,保持温度不变,将容器的容积扩大到原来的两倍,再达到平衡时,测得A的浓度降低为0.18mol/L,则A 的转化率(3)反应mA(g)+nB(g)
 pC(g)达到平衡后,当减压后混合体系中C 的百分含量减小。则加压后,C 的百分含量
pC(g)达到平衡后,当减压后混合体系中C 的百分含量减小。则加压后,C 的百分含量(4)将固体A放入密闭的真空容器中,反应2A(s)
 2B(g)+C(g)达到平衡。保持温度不变,增大容器容积,体系重新达到平衡,平衡
2B(g)+C(g)达到平衡。保持温度不变,增大容器容积,体系重新达到平衡,平衡(5)在一定温度下,有a、盐酸b、硫酸c、醋酸三种酸,若三者c(H+)相同时,物质的量浓度由大到小的顺序是