(1)已知:
(a)CH4(g)+2O2(g)=CO2(g)+2H2O(l) ΔH1=-890.3 kJ·mol1
(b)2H2(g)+O2(g)=2H2O(l) ΔH2=-571.6 kJ·mol1
(c)C(s)+O2(g)=CO2(g) ΔH3=-393.5 kJ·mol1
①C(s)+2H2(g)=CH4(g) ΔH=
②CH4制备合成气的原理是CH4(g)+CO2(g)
 2CO(g)+2H2(g)。
2CO(g)+2H2(g)。A. 下列叙述能表示该反应在恒温恒容条件下达到平衡状态的是
a. 混合气体的压强不再发生变化
b. 混合气体的密度不再发生变化
c. 反应容器中CO2、CO的物质的量的比值不再发生变化
d. 混合气体的平均相对分子质量不再发生变化
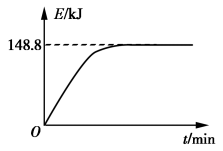
B. 若1 g CH4(g)完全反应吸收热量为15.5 kJ。在恒容密闭容器中充入1 mol CH4和1 mol CO2在一定条件下反应,体系吸收热量随着时间变化如图所示。在该条件下,甲烷的最大转化率(α)为
(2)在密闭容器中发生反应:2NO(g)+H2(g)= N2(g)+2H2O(g),其反应速率与浓度关系式为v=kcm(NO)·cn(H2)(k为常数,只与温度有关,m、n为反应级数,取正整数),在某温度下测得有关数据如表所示。
| 序数 | c(NO)/(mol·L1) | c(H2)/(mol·L1) | v/(mol·L1·min1) |
| Ⅰ | 0.10 | 0.10 | 0.414 |
| Ⅱ | 0.20 | 0.20 | 3.312 |
| Ⅲ | 0.10 | 0.20 | 0.828 |
总反应分两步进行:第i步,2NO+H2=N2+H2O2(很慢);第ii步,H2O2+H2=2H2O(很快)。在上述温度下,当c(NO)=c(H2)=0.50 mol·L1时v=
(3)在2 L恒容密闭容器中充入3 mol NO(g)和3 mol CO(g),发生反应:2NO(g)+2CO(g)
 N2(g)+2CO2(g),在一定温度下达到平衡,测得平衡体系中c(N2)=0.5 mol·L1。则该条件下平衡常数K值为
N2(g)+2CO2(g),在一定温度下达到平衡,测得平衡体系中c(N2)=0.5 mol·L1。则该条件下平衡常数K值为(4)汽车尾气中含有CO、NO2等有毒气体,对汽车加装尾气净化装置,可使有毒气体相互反应转化为无毒气体:4CO(g)+2NO2(g)
 4CO2(g)+N2(g) ΔH=-1 200 kJ·mol-1。对于该反应,温度不同(T2>T1),其他条件相同时,下列图象不正确的是
4CO2(g)+N2(g) ΔH=-1 200 kJ·mol-1。对于该反应,温度不同(T2>T1),其他条件相同时,下列图象不正确的是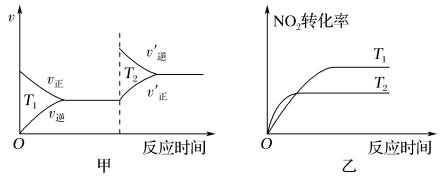
相似题推荐
反应Ⅰ:CH3OH(g)
 HCHO(g)+H2(g)ΔH>0
HCHO(g)+H2(g)ΔH>0反应Ⅱ:CH3OH(g)+
 O2(l)
O2(l) HCHO(g)+H2O(g)ΔH<0
HCHO(g)+H2O(g)ΔH<0反应Ⅲ:H2(g)+
 O2(g)
O2(g) H2O(g)ΔH<0
H2O(g)ΔH<0副反应:反应Ⅳ:CH3OH(g)+O2(g)
 CO(g)+2H2O(g)ΔH<0
CO(g)+2H2O(g)ΔH<0(1)在恒温恒压下的密闭容器中,充入1mol的甲醇,发生反应I,若起始压强为p0=aPa,达到平衡时甲醇的转化率为50%,计算反应平衡常数Kp=
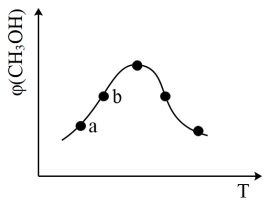
(2)Na2CO3是甲醇脱氢制甲醛的催化剂,有研究指出,催化反应的部分机理如下:
历程ⅰ:CH3OH→·H+·CH2OH
历程ⅱ:·CH2OH→·H+HCHO
历程ⅲ:·CH2OH→3·H+CO
历程ⅳ:·H+·H→H2
如图表示一定条件下温度对碳酸钠催化脱氢性能的影响,回答下列问题:
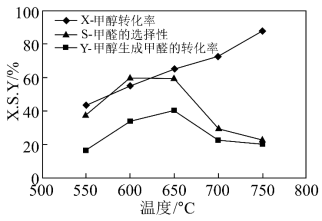
①从平衡角度解析550~650℃甲醇生成甲醛的转化率随温度升高的原因
②反应历程ⅰ的活化能
 HCHO(g)+H2(g)活化能。
HCHO(g)+H2(g)活化能。③650~750℃,反应历程ⅱ的速率反应
【推荐2】近年来,磁性材料的研究备受国际磁学界的关注。Fe3O4具有许多优异的性能,在磁性材料等领域应用广泛。回答下列问题:
(1)以太阳能为热源分解Fe3O4,经由热化学铁氧化合物循环分解水制H2的过程如图所示。
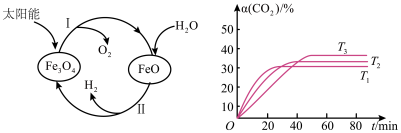
已知:H2O(l)= H2(g)+ O2(g)△H1+285.5kJ·mol-1
O2(g)△H1+285.5kJ·mol-1
过程I:2 Fe3O4(s)⇌6 FeO (s)+O2(g)△H2=+313.2kJ•mol-1
则过程II的热化学方程式为
(2)磁铁矿(Fe3O4)是工业上冶炼铁的原料之一,发生的主要反应为Fe3O4(s)+4CO(g)⇌3Fe(s)+4CO2(g)△H <0,T℃ 时,在 1L 恒容密闭容器中加入Fe3O4、CO各0.5mol,5min反应达到平衡时,固体质量减小了6.4g。
①从起始到反应达到平衡时v(CO2)=
②欲提高该反应中 CO的平衡转化率,可采取的措施是
③其他条件不变时,该反应在不同温度下,CO2的产率随时间的变化α(CO2)~t曲线如图所示,温度T1、T2、T3由大到小的关系是
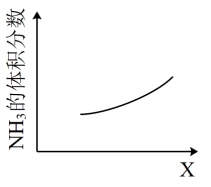
(1)脱硝。为消除NOx对环境的污染,利用NH3在一定条件下与NO反应生成无污染气体。
已知:4NH3(g)+5O2(g)
 4NO(g)+6H2O(g) △H=-905kJ/mol
4NO(g)+6H2O(g) △H=-905kJ/molN2(g)+O2(g)
 2NO(g) △H=+180 kJ/mol
2NO(g) △H=+180 kJ/mol①NH3(g)与NO(g)在一定条件下反应的热化学方程式为:
②下图是①中反应过程中NH3的体积分数随X变化的示意图,X代表的物理量可能是
(2)脱硫。下图电解装置可将雾霾中的SO2、NO转化为(NH4)2SO4。
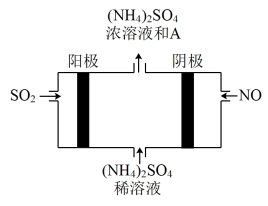
①装置中发生反应的化学方程式为
②阴极的电极反应式是
③某种脱硫工艺中将废气处理后,与一定量的氨气、空气反应,生成硫酸铵和硝酸铵的混合物,可作为化肥。硫酸铵和硝酸铵的水溶液pH<7,其原因用离子方程式表示为
(3)脱碳。将CO2转化为甲醇的热化学方程式为:CO2(g)+3H2(g)
 CH3OH(g)+H2O(g) △H 取五份等体积的CO2和H2的混合气体(物质的量之比均为1:3),分别加入温度不同、容积相同的恒容密闭容器中,发生上述反应,反应相同时间后,测得甲醇的体积分数
CH3OH(g)+H2O(g) △H 取五份等体积的CO2和H2的混合气体(物质的量之比均为1:3),分别加入温度不同、容积相同的恒容密闭容器中,发生上述反应,反应相同时间后,测得甲醇的体积分数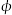 (CH3OH)与反应温度T的关系曲线如下图所示,则上述CO2转化为甲醇的反应的△H=
(CH3OH)与反应温度T的关系曲线如下图所示,则上述CO2转化为甲醇的反应的△H=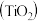 转化为
转化为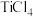 ,再进一步还原得到钛。回答下列问题:
,再进一步还原得到钛。回答下列问题:(1)
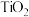 转化为
转化为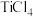 有直接氯化法和碳氯化法。在
有直接氯化法和碳氯化法。在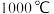 时反应的热化学方程式及其平衡常数如下:
时反应的热化学方程式及其平衡常数如下:(ⅰ)直接氯化:

(ⅱ)碳氯化:
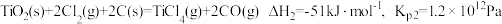
①反应
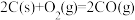 的
的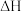 为
为 ,
,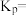
②碳氯化的反应趋势远大于直接氯化,其原因是
③对于碳氯化反应:增大压强,平衡
(2)在
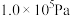 ,将
,将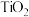 、C、
、C、 以物质的量比1∶2.2∶2进行反应。体系中气体平衡组成比例(物质的量分数)随温度变化的理论计算结果如图所示。
以物质的量比1∶2.2∶2进行反应。体系中气体平衡组成比例(物质的量分数)随温度变化的理论计算结果如图所示。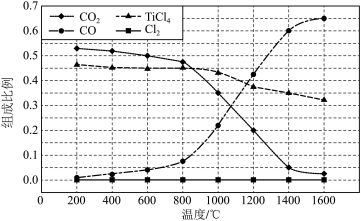
 的平衡常数
的平衡常数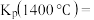
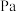 。
。②图中显示,在
 平衡时
平衡时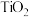 几乎完全转化为
几乎完全转化为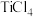 ,但实际生产中反应温度却远高于此温度,其原因是
,但实际生产中反应温度却远高于此温度,其原因是(3)
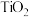 碳氯化是一个“气—固—固”反应,有利于
碳氯化是一个“气—固—固”反应,有利于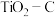 “固—固”接触的措施是
“固—固”接触的措施是 C2H4(g) △H。回答下列问题:
C2H4(g) △H。回答下列问题:(1)H2可通过反应CH4(g)+H2O(g)
 CO(g)+3H2(g)获取。T℃时,向2L恒容密闭容器中充入1.2mol水蒸气和1.2molCH4(g),经过2min反应达平衡,平衡后测得CO的浓度为0.3mol•L-1,则0~2min内H2的平均反应速率为
CO(g)+3H2(g)获取。T℃时,向2L恒容密闭容器中充入1.2mol水蒸气和1.2molCH4(g),经过2min反应达平衡,平衡后测得CO的浓度为0.3mol•L-1,则0~2min内H2的平均反应速率为(2)对于反应C2H2(g)+H2(g)
 C2H4(g),其他条件一定时,温度、压强对平衡时混合气体中C2H4体积分数的影响如图所示。
C2H4(g),其他条件一定时,温度、压强对平衡时混合气体中C2H4体积分数的影响如图所示。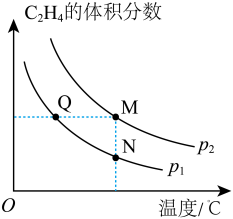
①该反应为
②Q、M、N三点的平衡常数KQ、KM、KN的大小关系为
(3)对于反应C2H2(g)+H2(g)
 C2H4(g),为了增大反应速率,且增大H2的平衡转化率,下列措施一定可行的是
C2H4(g),为了增大反应速率,且增大H2的平衡转化率,下列措施一定可行的是a.降低温度 b.使用合适的催化剂
c.缩小容器容积 d.减小原料气中
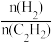 的比值
的比值
(1)氢气燃烧热值高。实验测得,在常温常压下1gH2完全燃烧生成液态水,放出142.9kJ热量。则表示H2燃烧热的热化学方程式为
 ,则氨气在空气中燃烧生成液态水和氮气时的热化学方程式为
,则氨气在空气中燃烧生成液态水和氮气时的热化学方程式为(2)氢气是合成氨的重要原料。
①当合成氨反应达到平衡后,改变某一外界条件(不改变
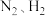 和
和 的量),反应速率与时间的关系如图所示。
的量),反应速率与时间的关系如图所示。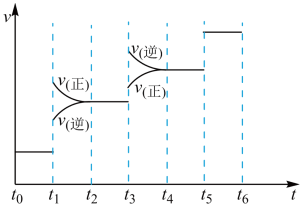
图中t1时引起平衡移动的条件可能是
②氨催化氧化可以制硝酸,此过程中涉及氮氧化物,如
 等。对于反应:
等。对于反应: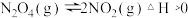 ,在温度为
,在温度为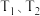 时,平衡体系中NO2的体积分数随压强变化曲线如图所示。
时,平衡体系中NO2的体积分数随压强变化曲线如图所示。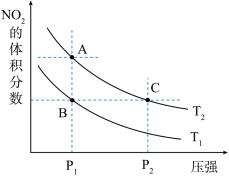
下列说法正确的是
a.
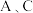 两点的化学平衡常数:
两点的化学平衡常数:
b.
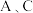 两点的气体颜色:
两点的气体颜色: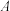 浅,
浅, 深
深 c.
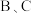 两点的气体平均相对分子质量:
两点的气体平均相对分子质量:
d.
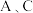 两点的反应速率:
两点的反应速率:
e.由状态B到状态A,可以用加热的方法
(1)汽车尾气净化的主要原理为:2NO(g)+2CO
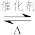 2CO2(g)+N2(g)△H<O
2CO2(g)+N2(g)△H<O①反应的速率-时间图象如图甲所示.若其他条件不变,仅在反应前加入合适的催化剂,其速率时间图象如图乙所示.以下说法正确的是
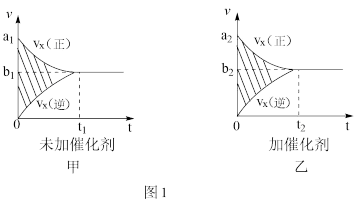
A.a1>a2B.b1<b2C.t1>t2
D.图乙中阴影部分面积更大 E.图甲中阴影部分面积更大
②若该反应在绝热、恒容的密闭体系中进行,下列示意图正确且能说明反应在进行到t1时刻达到平衡状态的是

(2)直接排放煤燃烧产生的烟气会引起严重的环境问题.煤燃烧产生的烟气含氮的氧化物,用CH4催化还原NOx可以消除氮氧化物的污染.
已知:CH4(g)+2NO2(g)⇌N2(g)+CO2(g)+2H2O(g)△H=﹣867kJ/mol
2NO2(g)⇌N2O4(g)△H=﹣56.9kJ/mol H2O(g)=H2O(l)△H=﹣44.0kJ/mol
写出CH4催化还原N2O4(g)生成N2和H2O(l)的热化学方程式:
(3)CH4和H2O(g) 在催化剂表面发生反应CH4+H2O⇌CO+3H2,该反应在不同温度下的化学平衡常数如表:
| 温度/℃ | 800 | 1000 | 1200 | 1400 |
| 平衡常数 | 0.45 | 1.92 | 276.5 | 1771.5 |
①该反应是
②T℃时,向1L密闭容器中投入l mol CH4和l mol H2O(g),平衡时c(CH4)=0.5mol/L,该温度下反应CH4+H2O⇌CO+3H2的平衡常数K=
(二)恒温时,将2molA和2molB气体投入固定容积为2L密闭容器中发生反应:2A(g)+B(g)⇌xC(g)+D(s),10s时,测得A的物质的量为1.7mol,C的反应速率为0.0225mol·L—1·s—1;40s时反应恰好处于平衡状态,此时B的转化率为20%。请填写下列空白:
(1)x=
(2)从反应开始到10s,B的平均反应速率为
(3)从反应开始到40s达平衡状态,A的平均反应速率为
(4)平衡时容器中B的体积分数为
(5)下列各项能表示该反应达到平衡状态是
A.消耗A的物质的量与生成D的物质的量之比为2∶1
B.容器中A、B的物质的量 n(A)∶n(B) =2∶1
C.气体的平均相对分子质量不再变化
D.压强不再变化
E.气体密度不再变化
(1)NO2与N2O4存在以下转化关系:2NO2(g)
 N2O4(g)。
N2O4(g)。①已知:标准状况(25℃、101kPa)下,由最稳定的单质生成1mol化合物的焓变(或反应热),称为该化合物的标准摩尔生成焓。几种氧化物的标准摩尔生成焓如表所示:
| 物质 | NO(g) | NO2(g) | N2O4(g) | CO(g) |
| 标准摩尔生成焓/(kJ·mol-1) | 90.25 | 33.18 | 9.16 | -110.53 |
则NO2转化成N2O4的热化学方程式为
②将一定量N2O4投入固定容积的恒温容器中,下述现象能说明反应达到平衡状态的是
A.v(N2O4)=2v(NO2) B.气体的密度不变
C.
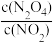 不变 D.气体的平均相对分子质量不变
不变 D.气体的平均相对分子质量不变③达到平衡后,保持温度不变,将气体体积压缩到原来的一半,再次达到平衡时,混合气体颜色
(2)在容积均为2L的甲、乙两个恒容密闭容器中,分别充入等量NO2,发生反应:2NO2(g)
 2NO(g)+O2(g)。保持温度分别为T1、T2,测得
2NO(g)+O2(g)。保持温度分别为T1、T2,测得 n(NO2)与n(O2)随时间的变化如图所示:
n(NO2)与n(O2)随时间的变化如图所示: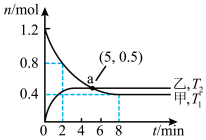
①T1时,反应从开始到第2min,平均速率v(O2)=
②实验测得:v正=k正c2(NO2),v逆=k逆c2(NO)·c(O2),k正、k逆为速率常数,受温度影响,下列有关说法正确的是
A.反应正向放热
B.a点处,v逆(甲)<v逆(乙)
C.k正(T2)>k正(T1)
D.温度改变,k正与k逆的比值不变
(3)硫代硫酸钠(Na2S2O3)在碱性溶液中是较强的还原剂,可用于净化氧化度较高的NOx废气,使之转化为无毒、无污染的N2。请写出NO2与硫代硫酸钠碱性溶液反应的离子方程式:
(1)甲烷、硫化氢重整反应制备氢气的原理为
 ,该反应体系的能量随反应过程的变化关系如图所示:
,该反应体系的能量随反应过程的变化关系如图所示: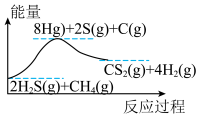
气态原子形成1 mol
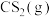 和4 mol
和4 mol 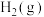 会释放热量2898 kJ,则1 mol
会释放热量2898 kJ,则1 mol 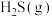 和0.5 mol
和0.5 mol 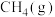 断键形成气态原子需要吸收的能量可能是___________(填字母)。
断键形成气态原子需要吸收的能量可能是___________(填字母)。| A.4056 J | B.1396 kJ | C.1506 kJ | D.20356 J |
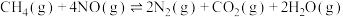 ,下列能说明该反应达到平衡状态的是___________(填字母)。
,下列能说明该反应达到平衡状态的是___________(填字母)。| A.体系总压强保持不变 |
| B.形成4mol C-H键的同时有4mol H-O键形成 |
C.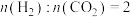 |
| D.NO的物质的量分数保持不变 |
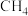 与
与 重整体系中通入适量
重整体系中通入适量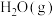 发生反应:
发生反应: 。在一定温度下,向恒容密闭容器中通入3 mol
。在一定温度下,向恒容密闭容器中通入3 mol 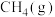 、1mol
、1mol 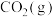 和4 mol
和4 mol 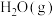 ,起始总压为12 MPa,体系中部分气体的含量与时间变化的关系如图所示:
,起始总压为12 MPa,体系中部分气体的含量与时间变化的关系如图所示: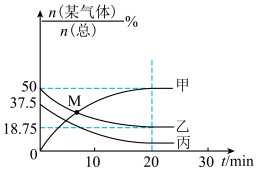
①曲线甲、丙表示的物质依次是
②M点时,
 的转化率为
的转化率为③20 min时达到平衡,0 ~20 min内用
 表示的平均反应速率为
表示的平均反应速率为 ,CO的分压p(CO)=
,CO的分压p(CO)= CH3OH(g) △H1=-116kJ/mol
CH3OH(g) △H1=-116kJ/mol(1)已知:CO(g)+
 O2(g)=CO2 (g) △H2=-283kJ/mol
O2(g)=CO2 (g) △H2=-283kJ/mol H2(g) +
 O2(g)=H2O (g) △H1=-242kJ/mol
O2(g)=H2O (g) △H1=-242kJ/mol则表示1mol气态甲醇完全燃烧生成CO2和水蒸气时的热化学方程式:
(2)在容积为1L的恒容容器中,分别研究在230℃、250℃、270℃三种温度下合成甲醇的规律。如图是上述三种温度下不同的H2和CO的起始组成比(起始时CO的物质的量均为1mol)与CO平衡转化率的关系。
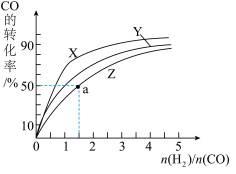
请回答:
①在上述三种温度中,曲线Z对应的温度是
②利用图中a点对应的数据,计算出曲线Z在对应温度下CO(g)+2H2(g)=CH3OH(g)的平衡常数 K=
③下列既能提高上述反应中 CO 的平衡转化率,又能增大反应速率的是
a.增大压强 b.降低温度 c.增大H2浓度 d.加高效催化剂
④若上述反应在某恒温恒容容器中发生,能说明该反应达到平衡的是
a.气体平均相对分子质量保持不变 b.△H1保持不变
c.
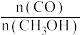 保持不变 d.气体密度保持不变
保持不变 d.气体密度保持不变(3)在某温度下,将一定量的CO和H2投入10L的密闭容器中,5min时达到平衡,各物质的物质的浓度(mol/L)变化如下表所示:
| 0min | 5min | 10min | |
| CO | 0.1 | 0.05 | |
| H2 | 0.2 | 0.2 | |
| CH3OH | 0 | 0.04 | 0.05 |
 合成气→高附加值产品”的工艺路线,可有效实现
合成气→高附加值产品”的工艺路线,可有效实现 的资源化利用。回答下列问题:
的资源化利用。回答下列问题:(1)
 加氢制合成气(CO、
加氢制合成气(CO、 )时发生下列反应:
)时发生下列反应:①
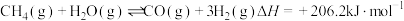
②
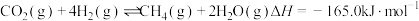
则

 。
。(2)
 经催化加氢可合成烯烃:
经催化加氢可合成烯烃: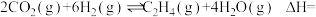 ?在0.1MPa时,按
?在0.1MPa时,按 投料,如图为不同温度(T)下,平衡时四种气态物质的物质的量(n)关系。
投料,如图为不同温度(T)下,平衡时四种气态物质的物质的量(n)关系。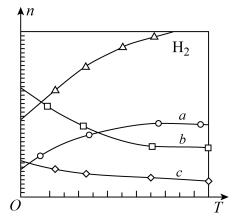
①在一个恒温恒容的密闭容器中,该可逆反应达到平衡的标志是
A.容器内各物质的浓度不随时间变化
B.
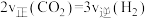
C.容器内压强不随时间变化
D.混合气体的密度不再改变
②该反应的

③曲线c表示的物质为
④为提高
 的转化率,可以采取什么措施
的转化率,可以采取什么措施(3)由
 与
与 反应合成甲醇:
反应合成甲醇: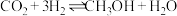
 。某温度下,将
。某温度下,将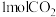 和
和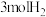 充入体积不变的2L密闭容器中,初始总压为8MPa,发生上述反应,测得不同时刻反应后与反应前的压强关系如下表:
充入体积不变的2L密闭容器中,初始总压为8MPa,发生上述反应,测得不同时刻反应后与反应前的压强关系如下表:| 时间/h | 1 | 2 | 3 | 4 | 5 |
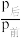 | 0.92 | 0.85 | 0.79 | 0.75 | 0.75 |
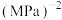 (用平衡分压代替平衡浓度计算,分压=总压×物质的量分数)。
(用平衡分压代替平衡浓度计算,分压=总压×物质的量分数)。(4)电催化
 制备有机物燃料可实现资源综合利用。如图装置工作时,阳极电极反应式为
制备有机物燃料可实现资源综合利用。如图装置工作时,阳极电极反应式为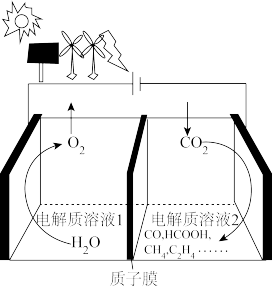
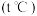 下,向
下,向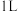 密闭容器中通入
密闭容器中通入 和
和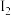 各
各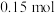 ,发生如下反应:
,发生如下反应: 一段时间后达到平衡,测得数据如下:
一段时间后达到平衡,测得数据如下: | 2 | 4 | 7 | 9 |
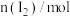 | 0.12 | 0.11 | 0.10 | 0.10 |
请回答:
(1)
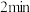 内,
内,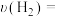
 。
。(2)下列能证明该反应已经达到平衡的是
a.
 b.混合气体的颜色不再发生变化
b.混合气体的颜色不再发生变化c.混合气体中各组分的物质的量不再发生变化 d.混合气体的密度不再变化。
(3)①该反应的化学平衡常数表达式为
 的平衡转化率是
的平衡转化率是(4)升高温度,化学平衡常数K要
(5)该温度
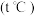 下,若开始通入一定量的
下,若开始通入一定量的 和
和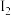 ,反应达到平衡时,测得
,反应达到平衡时,测得 ,
,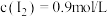 ,则平衡时
,则平衡时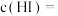
 。
。



