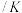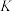工艺1:丙烷无氧脱氢工艺:
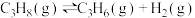
工艺2:丙烷氧化脱氢工艺:

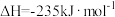
回答下列问题:
(1)根据下表数据,计算无氧脱氢工艺中的
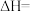
 ,与工艺1相比,工艺2的优点是
,与工艺1相比,工艺2的优点是| 物质 | C3H8(g) | C3H6(g) | H2(g) |
燃烧热( ) ) | 2217.8 | 2058.0 | 285.8 |
(2)若提高工艺1丙烷的平衡转化率,下列措施不可行的是___________(填字母)。
| A.适当的升高温度 |
| B.增大压强 |
| C.提高恒容反应容器中丙烷的初始浓度 |
| D.向恒压反应容器中的丙烷掺入水蒸气(水蒸气不参与反应) |
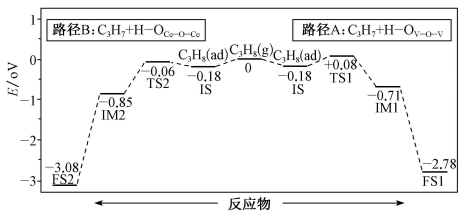
用图中数据可知,脱第1个氢原子时,最有可能按路径
(4)工艺1脱氢时,还会产生副产物C2H4,写出生成副产物的化学方程式:
 ,丙烷的总平衡转化率为40%,丙烯的选择性为50%,则工艺1的平衡常数K=
,丙烷的总平衡转化率为40%,丙烯的选择性为50%,则工艺1的平衡常数K=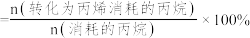 ]。
]。
相似题推荐
 已知某温度下某反应的化学平衡常数表达式为:
已知某温度下某反应的化学平衡常数表达式为: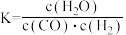 ,它所对应的化学反应为:
,它所对应的化学反应为: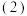 已知在一定温度下,
已知在一定温度下,①
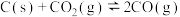
 平衡常数
平衡常数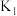
②
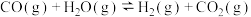
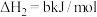 平衡常数
平衡常数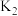
③
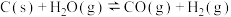
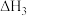 平衡常数
平衡常数
则
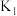 、
、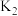 、
、 之间的关系是
之间的关系是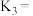
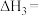
 用含a、b的代数式表示
用含a、b的代数式表示 。
。 煤化工通常通过研究不同温度下平衡常数以解决各种实际问题。已知等体积的一氧化碳和水蒸气进入反应器时,发生如下反应:
煤化工通常通过研究不同温度下平衡常数以解决各种实际问题。已知等体积的一氧化碳和水蒸气进入反应器时,发生如下反应: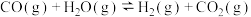 ;该反应平衡常数随温度的变化如表所示:
;该反应平衡常数随温度的变化如表所示:温度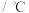 | 400 | 500 | 800 |
| 平衡常数K |  | 9 | 1 |
 填“吸热”或“放热”
填“吸热”或“放热” ,若在
,若在 时进行,设起始时CO和
时进行,设起始时CO和 的起始浓度均为
的起始浓度均为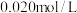 ,在该条件下,CO的平衡转化率为
,在该条件下,CO的平衡转化率为 高炉炼铁产生的废气中的CO可进行回收,使其在一定条件下和
高炉炼铁产生的废气中的CO可进行回收,使其在一定条件下和 反应制备甲醇:
反应制备甲醇: 。请回答下列问题:
。请回答下列问题:①若在相同温度和恒容且容积相同的三个密闭容器中,按不同方式投入反应物,测得反应达到平衡时的有关数据如下表:
| 容器 | 反应物投入的量 | 反应物的转化率 |  的浓度 的浓度 | 能量变化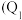 、 、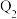 、 、 均大于 均大于 |
| 甲 | 1molCO和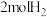 | 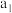 | 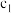 | 放出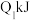 热量 热量 |
| 乙 |  | 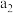 | 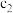 | 吸收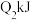 热量 热量 |
| 丙 | 2molCO和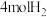 | 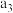 | 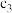 | 放出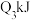 热量 热量 |

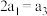
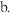


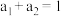
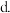
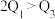
 该反应若生成
该反应若生成 ,则放出
,则放出 热量
热量②若在一体积可变的密闭容器中充入
 、
、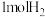 和
和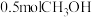 ,达到平衡时测得混合气体的密度是同温同压下起始的
,达到平衡时测得混合气体的密度是同温同压下起始的 倍,则该反应向
倍,则该反应向 填“正”或“逆”
填“正”或“逆” 反应方向移动。
反应方向移动。
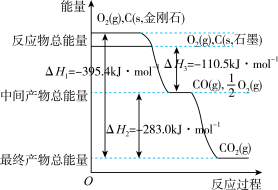
写出石墨和二氧化碳反应生成一氧化碳的热化学方程式
(2)核内中子数为N的R2+,质量数为A,则n g它的氧化物(RO)中所含质子的物质的量是
(3)已知:C(s)+O2(g)=CO2(g) △H1
CO2(g)+C(s)=2CO(g) △H2
2CO(g)+O2(g)=2CO2(g) △H3
4Fe(s)+3O3(g)=2Fe2O3(s) △H4
3 CO(g)+Fe2O3(s)=3CO2(g)+2Fe(s) △H5
△H3
(4)Cl2通入FeBr2溶液中,Cl2与FeBr2物质的量之比4︰5,请写出离子方程式
CuCl2(s)=CuCl(s)+
 Cl2(g) ΔH1=83 kJ·mol− 1
Cl2(g) ΔH1=83 kJ·mol− 1CuCl(s)+
 O2(g)=CuO(s)+
O2(g)=CuO(s)+ Cl2(g) ΔH2=− 20 kJ·mol− 1
Cl2(g) ΔH2=− 20 kJ·mol− 1CuO(s)+2HCl(g)=CuCl2(s)+H2O(g) ΔH3=− 121 kJ·mol− 1
则4HCl(g)+O2(g)=2Cl2(g)+2H2O(g)的ΔH=
(2)硅粉与HCl在300℃时反应生成1mol SiHCl3气体和H2,放出225KJ热量,该反应的热化学方程式为
(3)将SiCl4氢化为SiHCl3有三种方法,对应的反应依次为:
①SiCl4(g)+H2(g)
 SiHCl3(g)+HCl(g) ΔH1>0
SiHCl3(g)+HCl(g) ΔH1>0②3SiCl4(g)+2H2(g)+Si(s)
 4SiHCl3(g) ΔH2<0
4SiHCl3(g) ΔH2<0③2SiCl4(g)+H2(g)+Si(s)+HCl(g)
 3SiHCl3(g) ΔH3
3SiHCl3(g) ΔH3则反应③的ΔH3
(4)二氧化氯是目前国际上公认的第四代高效、无毒的广谱消毒剂,它可由KClO3在H2SO4存在下与Na2SO3反应制得.请写出反应的离子方程式
(5)氯化铵常用作焊接.如:在焊接铜器时用氯化铵除去铜器表面的氧化铜以便焊接,其反应为:_______CuO+______NH4Cl
 ______Cu+______CuCl2+______N2↑+______H2O
______Cu+______CuCl2+______N2↑+______H2O①配平此氧化还原反应方程式
②此反应中若产生0.2mol的气体,则有
| 编 号 | 往烧杯中加入的试剂及其用量(mL) | 催化剂 | 溶液开始变蓝时间(min) | ||||
| 0.1 mol/LKI溶液 | H2O | 0.01 mol/LX溶液 | 0.1 mol/L双氧水 | 0.1 mol/L稀盐酸 | |||
| 1 | 20.0 | 10.0 | 10.0 | 20.0 | 20.0 | 无 | 1.4 |
| 2 | 20.0 | m | 10.0 | 10.0 | n | 无 | 2.8 |
| 3 | 10.0 | 20.0 | 10.0 | 20.0 | 20.0 | 无 | 2.8 |
| 4 | 20.0 | 0 | 10.0 | 10.0 | 40.0 | 无 | t |
| 5 | 20.0 | 10.0 | 10.0 | 20.0 | 20.0 | 5滴Fe2(SO4)3 | 0.6 |
(1)已知:实验1、2的目的是探究H2O2浓度对H2O2+2H++2I-=2H2O+I2反应速率的影响。实验2中m=
(2)一定温度下,H2O2+2H++2I-=2H2O+I2,反应速率可以表示为:v=k·ca(H2O2)·cb(I-)·c(H+) (k为反应速率常数),则:
①实验4烧杯中溶液开始变蓝的时间t=
②根据上表数据可知,b =
(3)已知I2与X反应时,两者物质的量之比为1∶2。按上面表格中的X和KI的加入量,加入V(H2O2)>
(4)若要探究温度对H2O2+2H++2I-=2H2O+I2反应速率影响,在实验中温度不宜过高且采用水浴加热,其原因是
| 实验编号 | 反应物 | 催化剂 |
| ① | 10 mL 2% H2O2溶液 | 无 |
| ② | 10 mL 5% H2O2溶液 | 无 |
| ③ | 10 mL 5% H2O2溶液 | 1 mL 0.1 mol·L-1 FeCl3溶液 |
| ④ | 10 mL 5% H2O2溶液+少量HCl溶液 | 1 mL 0.1 mol·L-1 FeCl3溶液 |
| ⑤ | 10 mL 5% H2O2溶液+少量NaOH溶液 | 1 mL 0.1 mol·L-1 FeCl3溶液 |
(1)实验①和②的目的是
(2)实验③④⑤中,测得生成氧气的体积随时间变化的关系如图所示。
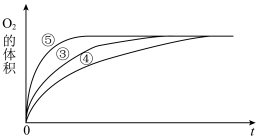
分析该图能够得出的实验结论是
【推荐3】氨是最重要的化学品之一,我国目前氨的生产能力位居世界首位。回答下列问题:
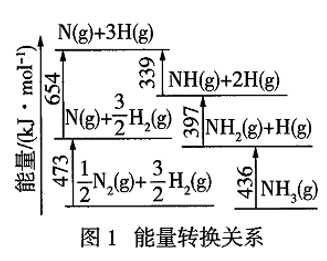
(1)Ⅰ.根据图1数据计算反应
 的
的
 。
。
Ⅱ.在一定温度、催化剂存在的条件下,密闭容器中CO与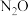 转化为
转化为 与
与 ,相关反应如下:
,相关反应如下:
①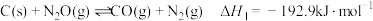
②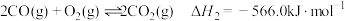
③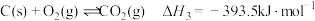
(2)则反应I:
 的
的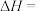
(3)合成氨工厂以“水煤气”和
 为原料,采用两段间接换热式绝热反应器,由进气口充入一定量含CO、
为原料,采用两段间接换热式绝热反应器,由进气口充入一定量含CO、 、
、 、
、 的混合气体,在反应器A进行合成氨,其催化剂Ⅲ铁触媒,在500℃活性最大,反应器B中主要发生的反应为
的混合气体,在反应器A进行合成氨,其催化剂Ⅲ铁触媒,在500℃活性最大,反应器B中主要发生的反应为
 ,装置如图2。
,装置如图2。
①温度比较:气流a
②气体流速一定,经由催化剂Ⅰ到催化剂Ⅱ,原料转化率有提升,其可能原因是:
③下列说法正确的是
A.合成氨是目前自然固氮最重要的途径
B.利用焦炭与水蒸气高温制备水煤气时,适当加快通入水蒸气流速,有利于水煤气生成
C.体系温度升高,可能导致催化剂失活,用热交换器将原料气可预热并使反应体系冷却
D.终端出口2得到的气体,通过液化可分离出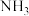
(4)LiH-3d过渡金属复合催化剂也可用于催化合成氨,已知
 先被吸附发生反应
先被吸附发生反应
 ,紧接着
,紧接着 被吸附发生的反应方程式
被吸附发生的反应方程式方法一:2FePO4(s)+ ___Li2CO3(s)+ ___C(s)⇌ ___LiFePO4(s)+ ___CO(g)
(1)配平该化学方程式
(2)该反应的平衡常数表达式为
(3)一定温度下,在2L密闭容器中发生上述反应。反应进行到20min时,容器内固体的质量减少了5.6g,则0∼20min内一氧化碳的平均反应速率是
方法二:LiFePO4可以通过(NH4)2Fe(SO4)2、H3PO4与LiOH溶液发生共沉淀反应,所得沉淀经80℃真空干燥、高温成型而制得。
(4)共沉淀反应投料时,不将(NH4)2Fe(SO4)2和LiOH溶液直接混合,其原因是
(5)磷酸亚铁锂电池总反应为:FePO4+Li→LiFePO4,放电时,负极为
 )材料作催化剂实现了乙醇水蒸汽重整制氢。其主要反应为:
)材料作催化剂实现了乙醇水蒸汽重整制氢。其主要反应为:反应Ⅰ:

 。
。反应Ⅱ:
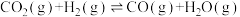
 。
。已知:在标准压力(
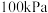 )和反应进行的温度时,由最稳定的单质合成
)和反应进行的温度时,由最稳定的单质合成 物质
物质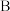 的反应焓变,叫做物质
的反应焓变,叫做物质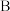 的标准摩尔生成焓。一些物质的标准摩尔生成焓如下表。
的标准摩尔生成焓。一些物质的标准摩尔生成焓如下表。| 物质 |  |  |  | 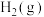 |
标准摩尔生成焓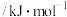 |  |  |  | 0 |
(1)
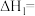
 。
。(2)一定温度下,向
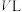 刚性密闭容器中通入
刚性密闭容器中通入 和
和 ,发生反应Ⅰ和反应Ⅱ,达到平衡后,
,发生反应Ⅰ和反应Ⅱ,达到平衡后, 的转化率为
的转化率为 ,
,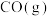 的物质的量为
的物质的量为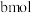 。
。①下列操作中,能提高
 平衡转化率的是
平衡转化率的是A.增加
 用量 B.将刚性容器体积扩大1倍 C.加入高效催化剂
用量 B.将刚性容器体积扩大1倍 C.加入高效催化剂②平衡时容器中

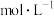 ,反应Ⅰ的平衡常数
,反应Ⅰ的平衡常数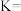
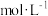 (用含字母的代数式表示)。
(用含字母的代数式表示)。(3)吸附在催化剂表面的
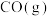 与
与 逐步反应机理及其相对能量如图。
逐步反应机理及其相对能量如图。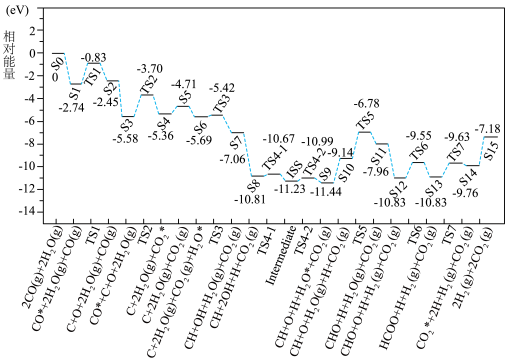

 ”、“
”、“ ”或“
”或“ ”)。反应Ⅱ历程中决速步骤是
”)。反应Ⅱ历程中决速步骤是 ”这样的格式,
”这样的格式, 和
和 代表0~15之间的正整数)。
代表0~15之间的正整数)。②已知反应Ⅱ的正反应速率
 ,逆反应速率
,逆反应速率 ,
,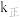 、
、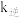 分别为正、逆反应的速率常数,
分别为正、逆反应的速率常数,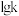 (
(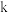 表示
表示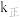 或
或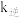 )与
)与 的关系如下图所示,则表示
的关系如下图所示,则表示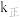 随温度变化的曲线是
随温度变化的曲线是 ”、“
”、“ ”、“
”、“ ”或“
”或“ ”)。
”)。
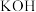 溶液,该电池的负极反应式为
溶液,该电池的负极反应式为
其中NO2二聚为N2O4的反应可以迅速达到平衡,体系的总压强p随时间t的变化如下表所示(t=∞时,N2O5(g)完全分解):
t/min | 0 | 40 | 80 | 160 | 260 | 1300 | 1700 | ∞ |
p/kPa | 35.8 | 40.3 | 42.5 | 45.9 | 49.2 | 61.2 | 62.3 | 63.1 |
N2O4(g)
 2NO2(g) △H2=+55.3 kJ· mol-1
2NO2(g) △H2=+55.3 kJ· mol-1则反应N2O5(g)=2NO2(g)+
 O2(g)的 △H=
O2(g)的 △H=(2)若提高反应温度至35℃,则N2O5(g)完全分解后体系压强介p∞(35℃)
(3)25℃时N2O4(g)
 2NO2(g)反应的平衡常数Kp=
2NO2(g)反应的平衡常数Kp=
(4)邻二甲苯经硝化得到两种一硝化产物:3-硝基邻二甲苯(3-NOX)和4-硝基邻二甲苯(4-NOX)。某研究小组研究了采用N2O5为硝化剂时催化剂用量对该反应的影响。将一定量的催化剂和邻二甲苯置于反应瓶中,控制温度反应一定时间。测得邻二甲苯的转化率和3-NOX的选择性如图所示:

①描述并解释随着催化剂用量的增加,邻二甲苯转化率的变化趋势:
②在图中画出4-NOX的选择性随催化剂用量变化的曲线
(1)CO还原NO的脱硝反应:2CO(g)+2NO(g)
 2CO2(g)+N2(g) △H
2CO2(g)+N2(g) △H已知:CO(g)+NO2(g)
 CO2(g)+NO(g) △H1=-226kJ·mol-1
CO2(g)+NO(g) △H1=-226kJ·mol-1N2(g)+2O2(g)
 2NO2(g) △H2=+68kJ·mol-1
2NO2(g) △H2=+68kJ·mol-1N2(g)+O2(g)
 2NO(g) △H3=+183kJ·mol-1
2NO(g) △H3=+183kJ·mol-1脱硝反应△H=
(2)某温度下,向体积为2L的恒容容器中通入2.00molNO2,发生反应:2NO2(g)
 N2O4(g)∆H=-57.0kJ·mol-1,已知:v正(NO2)=k1·c2(NO2),v逆(N2O4)=k2·c(N2O4),其中k1、k2为速率常数。测得NO2的体积分数[φ((NO2)]与反应时间(t)的关系如表:
N2O4(g)∆H=-57.0kJ·mol-1,已知:v正(NO2)=k1·c2(NO2),v逆(N2O4)=k2·c(N2O4),其中k1、k2为速率常数。测得NO2的体积分数[φ((NO2)]与反应时间(t)的关系如表:t/min | 0 | 20 | 40 | 60 | 80 |
φ((NO2) | 1.0 | 0.75 | 0.52 | 0.50 | 0.50 |
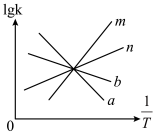
②
 的数值为
的数值为(3)用间接电化学法除去NO的过程,如图所示:
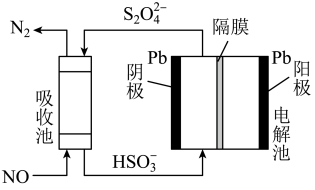
①已知电解池的阴极室中溶液的pH在4~7之间,写出阴极的电极反应式:
②用离子方程式表示吸收池中除去NO的原理:
【推荐2】 资源利用具有重要意义。
资源利用具有重要意义。
(1)工业上用二氧化碳催化加氢可合成乙醇,其反应原理为
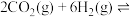
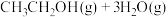
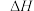 。
。温度 | 400 | 500 |
平衡常数 | 9 | 5.3 |
①通过表格中的数值可以推断:该反应在
② 的平衡转化率与氢碳比
的平衡转化率与氢碳比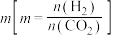 及压强随温度变化的关系分别如图
及压强随温度变化的关系分别如图 和图
和图 所示。
所示。
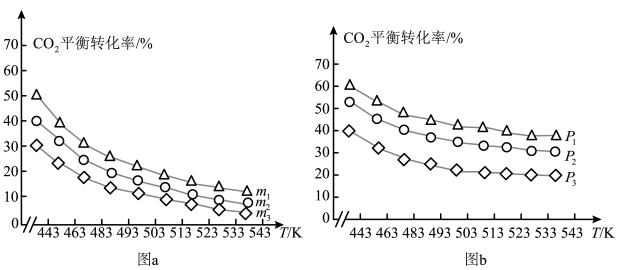
图 中氢碳比
中氢碳比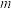 从大到小的顺序为
从大到小的顺序为
(2)
 催化加氢法可制备乙烯,反应原理为:
催化加氢法可制备乙烯,反应原理为: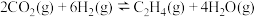
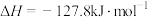 。向
。向 恒容密闭容器中加入
恒容密闭容器中加入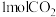 和
和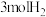 ,在催化剂作用下发生反应,测得平衡时体系中各气体的物质的量随温度的变化关系如图所示。
,在催化剂作用下发生反应,测得平衡时体系中各气体的物质的量随温度的变化关系如图所示。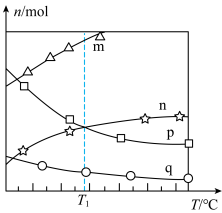
①图中曲线代表乙烯的物质的量随温度的变化关系的是
②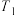 ℃时,化学平衡常数
℃时,化学平衡常数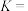
 的平衡转化率为
的平衡转化率为
(3)
 可用于制备苯乙烯。
可用于制备苯乙烯。在催化剂作用下, 参与反应的机理如图所示
参与反应的机理如图所示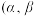 表示乙苯分子中
表示乙苯分子中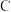 或
或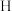 原子的位置; A、B为催化剂的活性位点,其中
原子的位置; A、B为催化剂的活性位点,其中 位点带部分正电荷,
位点带部分正电荷,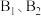 位点带部分负电荷)。根据元素电负性的变化规律,图中所示的反应机理中步骤I和步骤II可描述为
位点带部分负电荷)。根据元素电负性的变化规律,图中所示的反应机理中步骤I和步骤II可描述为

 转化合成气(
转化合成气( 、
、 )可有效实现碳资源的综合充分利用,有利于实现我国的“碳中和”。
)可有效实现碳资源的综合充分利用,有利于实现我国的“碳中和”。方法Ⅰ:
 热催化制备合成气:一定条件下用甲烷还原
热催化制备合成气:一定条件下用甲烷还原 制备合成气。
制备合成气。相关化学键的键能数据
| 化学键 |  |  |  |  |
键能 | 742 | 414 | 1046 | 436 |

 ,该反应为
,该反应为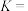
(2)已知某一温度时
 ,测得某时刻反应混合物中
,测得某时刻反应混合物中 、
、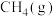 、
、 、
、 的浓度分别为
的浓度分别为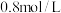 、
、 、
、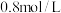 、
、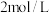 ,则此刻上述反应的平衡移动方向为
,则此刻上述反应的平衡移动方向为(3)热力学分析表明
 时易发生
时易发生 的歧化反应和
的歧化反应和 以上易发生
以上易发生 的裂解反应(方程式如下所示),一段时间后Ni基催化剂的催化效果会明显下降,原因是
的裂解反应(方程式如下所示),一段时间后Ni基催化剂的催化效果会明显下降,原因是

方法Ⅱ:
 加氢备合成气:将原料气
加氢备合成气:将原料气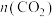 与
与 按一定比例在催化剂作用下发生反应a,同时伴随反应b发生,反应方程式如下所示。
按一定比例在催化剂作用下发生反应a,同时伴随反应b发生,反应方程式如下所示。反应a:

反应b:

(4)将原料气按
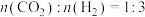 置于恒容密闭容器中发生反应,在相同时间内测得平衡时各组分摩尔分数与温度的变化曲线如图所示,则制合成气适宜的温度为
置于恒容密闭容器中发生反应,在相同时间内测得平衡时各组分摩尔分数与温度的变化曲线如图所示,则制合成气适宜的温度为
(5)理论上,适当增大氢气在混合物中的比例,
 的平衡转化率会
的平衡转化率会