解题方法
1 . 微颗粒的氧化锌作为一种纳米材料开始在相关领域发挥作用,利用锌焙砂 主要成分为
主要成分为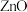 ,含
,含 及铅、铜、镉的氧化物
及铅、铜、镉的氧化物 生产高纯氧化锌的工业流程如图所示,回答下列问题:
生产高纯氧化锌的工业流程如图所示,回答下列问题:

已知: 微溶于水而生成亚砷酸
微溶于水而生成亚砷酸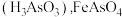 和
和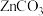 难溶于水。
难溶于水。
(1)基态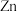 原子核外价层电子轨道表达式为
原子核外价层电子轨道表达式为______ ,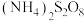 中
中 的化合价为
的化合价为______ 。
(2)“浸出”过程中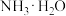 足量,则生成的主要配离子的化学式为
足量,则生成的主要配离子的化学式为______ ;增大浸出率可采取的措施是______  写出两条
写出两条 。
。
(3)“除砷”过程中,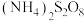 作氧化剂
作氧化剂 溶液呈酸性
溶液呈酸性 ,发生反应的离子方程式为
,发生反应的离子方程式为______ 。
(4)“除重金属”过程中加入 后
后 在溶液中的残留浓度为
在溶液中的残留浓度为 ,此时
,此时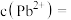
______ 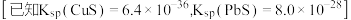 。
。
(5)“沉锌”前锌元素以 存在,写出“沉锌”的化学方程式
存在,写出“沉锌”的化学方程式______ 。
(6)流程中可循环利用的物质有 、
、______ 、______  填化学式
填化学式 。
。
 主要成分为
主要成分为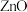 ,含
,含 及铅、铜、镉的氧化物
及铅、铜、镉的氧化物 生产高纯氧化锌的工业流程如图所示,回答下列问题:
生产高纯氧化锌的工业流程如图所示,回答下列问题:
已知:
 微溶于水而生成亚砷酸
微溶于水而生成亚砷酸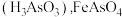 和
和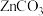 难溶于水。
难溶于水。(1)基态
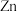 原子核外价层电子轨道表达式为
原子核外价层电子轨道表达式为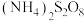 中
中 的化合价为
的化合价为(2)“浸出”过程中
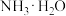 足量,则生成的主要配离子的化学式为
足量,则生成的主要配离子的化学式为 写出两条
写出两条 。
。(3)“除砷”过程中,
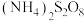 作氧化剂
作氧化剂 溶液呈酸性
溶液呈酸性 ,发生反应的离子方程式为
,发生反应的离子方程式为(4)“除重金属”过程中加入
 后
后 在溶液中的残留浓度为
在溶液中的残留浓度为 ,此时
,此时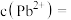
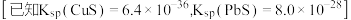 。
。(5)“沉锌”前锌元素以
 存在,写出“沉锌”的化学方程式
存在,写出“沉锌”的化学方程式(6)流程中可循环利用的物质有
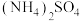 、
、 填化学式
填化学式 。
。
您最近一年使用:0次
2 .  、
、 、
、 、
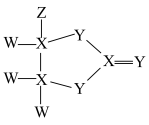
、 是原子序数依次增大且不相邻的短周期主族元素,由这四种元素组成的化合物的结构如图所示,下列有关说法正确的是
是原子序数依次增大且不相邻的短周期主族元素,由这四种元素组成的化合物的结构如图所示,下列有关说法正确的是
 、
、 、
、 、
、 是原子序数依次增大且不相邻的短周期主族元素,由这四种元素组成的化合物的结构如图所示,下列有关说法正确的是
是原子序数依次增大且不相邻的短周期主族元素,由这四种元素组成的化合物的结构如图所示,下列有关说法正确的是
A. 的原子结构示意图为 的原子结构示意图为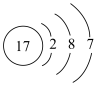 |
B. 、 、 、 、 的简单氢化物中沸点最高的是 的简单氢化物中沸点最高的是 |
C. 的基态原子有 的基态原子有 种空间运动状态的电子 种空间运动状态的电子 |
| D.该化合物在酸性条件下水解生成的气体为极性分子 |
您最近一年使用:0次
3 . 一种新型电池装置如下图所示。金刚石压砧把 压入储氢材料,在压力梯度的驱动下生成质子,电子先后通过下层
压入储氢材料,在压力梯度的驱动下生成质子,电子先后通过下层 电极和外接线路转移到
电极和外接线路转移到 电极参与反应,形成闭合回路。下列说法正确的是
电极参与反应,形成闭合回路。下列说法正确的是
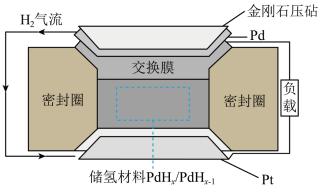
 压入储氢材料,在压力梯度的驱动下生成质子,电子先后通过下层
压入储氢材料,在压力梯度的驱动下生成质子,电子先后通过下层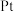 电极和外接线路转移到
电极和外接线路转移到 电极参与反应,形成闭合回路。下列说法正确的是
电极参与反应,形成闭合回路。下列说法正确的是
A. 电极发生的反应是氧化反应 电极发生的反应是氧化反应 |
B.电流方向是 负载 负载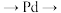 交换膜 交换膜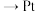 |
C.电池负极反应为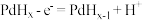 |
D.由于压力梯度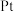 电极的电势会高于 电极的电势会高于 电极的电势 电极的电势 |
您最近一年使用:0次
4 . 三氯化铬是有机金属化学中的重要原料,为紫色晶体,熔点为83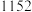 ℃,易潮解,高温下易被氧气氧化。实验室用
℃,易潮解,高温下易被氧气氧化。实验室用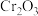 和
和 加热到
加热到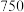 ℃时可制取
℃时可制取 ,同时还生成
,同时还生成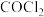 ,其装置图如下
,其装置图如下 加热及夹持装置略去
加热及夹持装置略去 ,请回答下列问题:
,请回答下列问题:
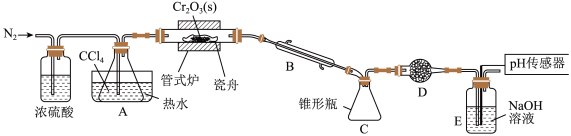
已知: 沸点
沸点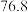 ℃;
℃;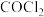 有毒,沸点
有毒,沸点 ℃,遇水剧烈水解生成刺激性气味的气体。
℃,遇水剧烈水解生成刺激性气味的气体。
Ⅰ.制备 :
:
(1)仪器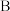 的名称是
的名称是______ 。
(2)装置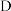 、
、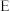 的作用分别是
的作用分别是______ 、______ 。
(3)实验前先通入一段时间 的目的是
的目的是______ 。
(4)制备 的化学方程式为
的化学方程式为______ 。
(5)判断反应结束的依据是______ 。
Ⅱ.测定 纯度:
纯度:
称取 产品,在强碱性条件下,加入过量
产品,在强碱性条件下,加入过量 溶液,并小火加热,使
溶液,并小火加热,使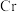 元素完全转化为
元素完全转化为 ;继续加热一段时间,冷却后配制成
;继续加热一段时间,冷却后配制成 溶液;取出
溶液;取出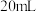 ,滴加适量硫酸酸化后,用新配制的
,滴加适量硫酸酸化后,用新配制的 标准液滴定至终点,消耗标准液
标准液滴定至终点,消耗标准液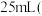 已知
已知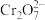 被
被 还原为
还原为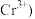 。
。
(6) 被
被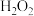 氧化成
氧化成 的离子方程式为
的离子方程式为______ 。
(7)产品中 的纯度为
的纯度为______  杂质不参与反应
杂质不参与反应 。
。
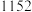 ℃,易潮解,高温下易被氧气氧化。实验室用
℃,易潮解,高温下易被氧气氧化。实验室用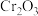 和
和 加热到
加热到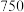 ℃时可制取
℃时可制取 ,同时还生成
,同时还生成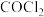 ,其装置图如下
,其装置图如下 加热及夹持装置略去
加热及夹持装置略去 ,请回答下列问题:
,请回答下列问题:
已知:
 沸点
沸点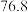 ℃;
℃;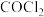 有毒,沸点
有毒,沸点 ℃,遇水剧烈水解生成刺激性气味的气体。
℃,遇水剧烈水解生成刺激性气味的气体。Ⅰ.制备
 :
:(1)仪器
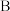 的名称是
的名称是(2)装置
 、
、 的作用分别是
的作用分别是(3)实验前先通入一段时间
 的目的是
的目的是(4)制备
 的化学方程式为
的化学方程式为(5)判断反应结束的依据是
Ⅱ.测定
 纯度:
纯度:称取
 产品,在强碱性条件下,加入过量
产品,在强碱性条件下,加入过量 溶液,并小火加热,使
溶液,并小火加热,使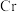 元素完全转化为
元素完全转化为 ;继续加热一段时间,冷却后配制成
;继续加热一段时间,冷却后配制成 溶液;取出
溶液;取出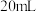 ,滴加适量硫酸酸化后,用新配制的
,滴加适量硫酸酸化后,用新配制的 标准液滴定至终点,消耗标准液
标准液滴定至终点,消耗标准液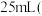 已知
已知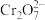 被
被 还原为
还原为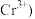 。
。(6)
 被
被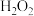 氧化成
氧化成 的离子方程式为
的离子方程式为(7)产品中
 的纯度为
的纯度为 杂质不参与反应
杂质不参与反应 。
。
您最近一年使用:0次
5 . 常温下,向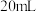 浓度为
浓度为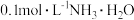 溶液中滴加等物质的量浓度的
溶液中滴加等物质的量浓度的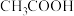 溶液,所得混合溶液的导电能力与加入
溶液,所得混合溶液的导电能力与加入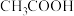 溶液体积
溶液体积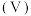 的关系如图所示,电离平衡常数
的关系如图所示,电离平衡常数 ,下列说法不正确的是
,下列说法不正确的是
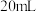 浓度为
浓度为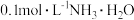 溶液中滴加等物质的量浓度的
溶液中滴加等物质的量浓度的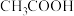 溶液,所得混合溶液的导电能力与加入
溶液,所得混合溶液的导电能力与加入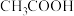 溶液体积
溶液体积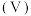 的关系如图所示,电离平衡常数
的关系如图所示,电离平衡常数 ,下列说法不正确的是
,下列说法不正确的是
A.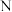 点溶液 点溶液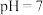 |
B.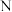 点溶液中: 点溶液中: |
C.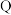 点溶液中: 点溶液中: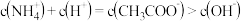 |
D.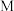 、 、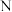 、 、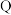 三点中, 三点中,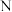 点溶液中水的电离程度最大 点溶液中水的电离程度最大 |
您最近一年使用:0次
解题方法
6 . 在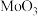 作用下,
作用下,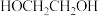 发生反应的历程如图所示。下列说法正确的是
发生反应的历程如图所示。下列说法正确的是
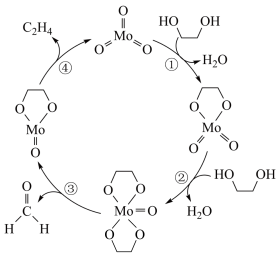
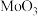 作用下,
作用下,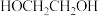 发生反应的历程如图所示。下列说法正确的是
发生反应的历程如图所示。下列说法正确的是
A.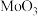 能提高 能提高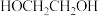 的平衡转化率 的平衡转化率 |
B.该反应历程中 、 、 键均发生断裂 键均发生断裂 |
C.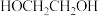 滴入过量酸性高锰酸钾溶液中生成 滴入过量酸性高锰酸钾溶液中生成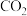 |
D.若将原料换为 ,则生成物为乙醛和丙烯 ,则生成物为乙醛和丙烯 |
您最近一年使用:0次
解题方法
7 . 氨硼烷 是一种新型储氢材料,分子中与
是一种新型储氢材料,分子中与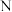 原子相连的
原子相连的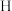 原子呈正电性,与
原子呈正电性,与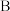 原子相连的
原子相连的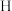 原子呈负催化剂电性。氨硼烷与水发生如下反应:
原子呈负催化剂电性。氨硼烷与水发生如下反应: 。下列说法正确的是
。下列说法正确的是
 是一种新型储氢材料,分子中与
是一种新型储氢材料,分子中与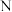 原子相连的
原子相连的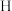 原子呈正电性,与
原子呈正电性,与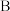 原子相连的
原子相连的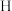 原子呈负催化剂电性。氨硼烷与水发生如下反应:
原子呈负催化剂电性。氨硼烷与水发生如下反应: 。下列说法正确的是
。下列说法正确的是A.中子数为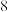 的 的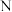 原子: 原子: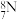 |
B.基态原子第一电离能由大到小: |
C.反应前后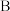 原子均采用 原子均采用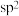 杂化 杂化 |
| D.氨硼烷中两种氢原子之间存在相互吸引作用 |
您最近一年使用:0次
解题方法
8 . 下列实验装置或操作能够达到实验目的的是
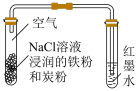 | 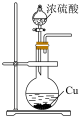 | 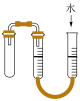 | 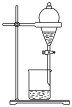 |
| A.验证铁粉发生析氢腐蚀 | B.验证浓硫酸的强氧化性 | C.检查装置气密性 | D.乙醇萃取碘水中的碘 |
A. | B. | C. | D.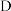 |
您最近一年使用:0次
9 . 下列叙述错误的是
A.工业上用 还原 还原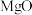 来冶炼金属 来冶炼金属 |
| B.石灰石是制造玻璃的主要原料之一 |
C.浓硫酸具有吸水性,可用于干燥 |
D.饱和 溶液可将水垢中的 溶液可将水垢中的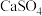 转化为 转化为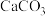 |
您最近一年使用:0次
解题方法
10 . 下列离子方程式书写正确的是
| A.AlCl3溶液显酸性的原因:Al3++3H2O=Al(OH)3+3H+ |
B.向 溶液中加入稀硫酸:S2O 溶液中加入稀硫酸:S2O +2H+=SO2↑+S↓+H2O +2H+=SO2↑+S↓+H2O |
C.向 溶液中通入少量CO2:CO2+OH—=HCO 溶液中通入少量CO2:CO2+OH—=HCO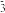 |
D.向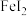 溶液中滴入少量 溶液中滴入少量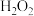 溶液:2Fe2++H2O2+2H+=2Fe3++2H2O 溶液:2Fe2++H2O2+2H+=2Fe3++2H2O |
您最近一年使用:0次



