①

②
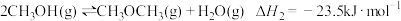
③

回答下列问题:
(1)反应3H2(g)+3CO(g)
 CH3OCH3 (g)+ CO2(g)的
CH3OCH3 (g)+ CO2(g)的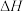 =
= 。
。(2)恒温恒容条件下反应②达到平衡后,下列措施能提高CH3OH的转化率的有
A.加入CH3OH(g) B.升高温度 C.增大压强 D.移出H2O(g) E.使用催化剂
(3)下列能说明反应3H2(g)+3CO(g)
 CH3OCH3 (g)+CO2(g)达到平衡状态的有
CH3OCH3 (g)+CO2(g)达到平衡状态的有A.H2和CO2的速率之比为3:1
B.单位时间内断裂3个H—H键的同时生成2个C=O键
C.恒温恒压条件下,混合气体的密度保持不变
D.恒温恒压条件下,混合气体的平均摩尔质量保持不变
E.绝热体系中,体系的温度保持不变
(4)一定量的CO2与足量碳在容积可变的恒压密闭容器中发生反应C(s)+CO2(g)
 2CO(g)。平衡时,体系中气体体积分数与温度的关系如图所示。已知:气体分压P分=气体总压P总
2CO(g)。平衡时,体系中气体体积分数与温度的关系如图所示。已知:气体分压P分=气体总压P总 体积分数。
体积分数。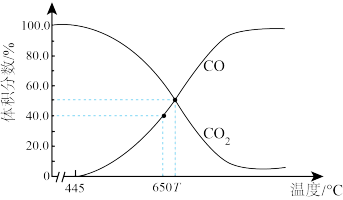
①反应的
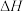
②650℃时,反应达到平衡后CO2的转化率为
③T℃时,用平衡分压代替平衡浓度表示的化学平衡常数Kp=
相似题推荐
(1)已知:Fe2O3(s)+3C(s)=2Fe(s)+3CO(g)ΔH1=a kJ·mol-1
CO(g)+
 O2(g)=CO2(g) ΔH2=b kJ·mol-1
O2(g)=CO2(g) ΔH2=b kJ·mol-14Fe(s)+3O2(g)=2Fe2O3(s) ΔH3=c kJ·mol-1
则C的燃烧热ΔH=
(2)①依据原电池的构成原理,下列化学反应在理论上可以设计成原电池的是
A.C(s)+CO2(g)=2CO(g)
B.NaOH(aq)+HCl(aq)=NaCl(aq)+H2O(l)
C.2H2O(l)=2H2(g)+O2(g)
D.2CO(g)+O2(g)=2CO2(g)
②若以熔融的K2CO3与CO2为反应的环境,依据所选反应设计成一个原电池,请写出该原电池的负极反应:
(3)某实验小组模拟工业合成氨反应N2(g)+3H2(g)
 2NH3(g) ΔH=-92.4 kJ·mol-1,开始他们将N2和H2混合气体20 mol(体积比1∶1)充入5 L合成塔中,反应前压强为P0,反应过程中压强用P表示,反应过程中
2NH3(g) ΔH=-92.4 kJ·mol-1,开始他们将N2和H2混合气体20 mol(体积比1∶1)充入5 L合成塔中,反应前压强为P0,反应过程中压强用P表示,反应过程中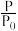 与时间t的关系如图所示。
与时间t的关系如图所示。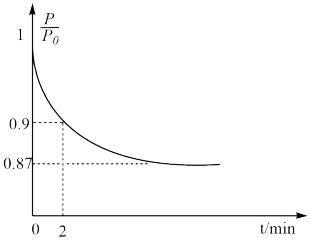
请回答下列问题:
①反应达平衡的标志是
A.压强保持不变
B.气体密度保持不变
C.NH3的生成速率是N2的生成速率的2倍
②0~2 min内,以c(N2)变化表示的平均反应速率为
③欲提高N2的转化率,可采取的措施有
A.向体系中按体积比1∶1再充入N2和H2
B.分离出NH3
C.升高温度
D.充入氦气使压强增大
E.加入一定量的N2
(4)25℃时,BaCO3和BaSO4的溶度积常数分别是8×10-9和1×10-10,某含有BaCO3沉淀的悬浊液中,c(
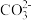 )=0.2 mol·L-1,如果加入等体积的Na2SO4溶液,若要产生BaSO4沉淀,加入Na2SO4溶液的物质的量浓度最小是
)=0.2 mol·L-1,如果加入等体积的Na2SO4溶液,若要产生BaSO4沉淀,加入Na2SO4溶液的物质的量浓度最小是(1)用CO2和H2可以合成二甲醚(CH3OCH3)。
已知:CO(g)+2H2(g)=CH3OH(g) ΔH1=-90.1 kJ·mol-1
CO(g)+H2O(g)=CO2(g)+H2(g) ΔH2=-41.1 kJ·mol-1
2CH3OH(g)=CH3OCH3(g)+H2O(g) ΔH3=-24.5 kJ·mol-1
则反应2CO2(g)+6H2(g)=CH3OCH3(g)+3H2O(g)ΔH=
(2)二甲醚(CH3OCH3)燃料电池可以提升能量利用率。利用二甲醚酸性介质燃料电池电解200 mL饱和食盐水(惰性电极),一段时间后,阴极收集到标况下的氢气2.24 L(设电解后溶液体积不变)。
①二甲醚燃料电池的正极反应式为:
②请写出电解食盐水的化学方程式:
③电解后溶液的c(OH-)=
(1)新型Zn-
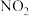 电池能将捕获的
电池能将捕获的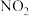 转化为
转化为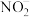 ,再将
,再将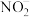 电解制氨,过程如下图所示。电极c上的电极反应式是
电解制氨,过程如下图所示。电极c上的电极反应式是
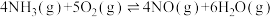
 ;
;
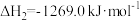 若
若 的正反应活化能为
的正反应活化能为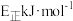 ,则其逆反应活化能为
,则其逆反应活化能为 (用含
(用含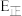 的代数式表示)。
的代数式表示)。(3)在合成氨工业中,原料气在进入合成塔前需用铜氨液除去其中的CO,其反应为:

 ,铜氨液吸收CO的适宜条件是
,铜氨液吸收CO的适宜条件是 中,配位原子是
中,配位原子是(4)在不同压强下,氮气、氢气以1∶3的体积比通入,反应达平衡时氨的体积分数与温度的计算结果如图所示,当
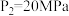 、
、 时,氮气的转化率
时,氮气的转化率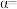
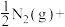
 的平衡常数
的平衡常数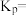
 (气体A的分压=气体总压强×气体A的体积分数,结果化为最简式)。
(气体A的分压=气体总压强×气体A的体积分数,结果化为最简式)。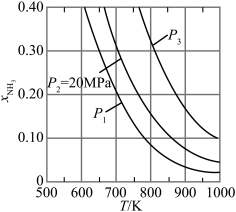

 .氨氮比、催化剂种类都会影响
.氨氮比、催化剂种类都会影响 的产率,保持其它初始条件不变重复实验,在相同时间内测得
的产率,保持其它初始条件不变重复实验,在相同时间内测得 产率与氨氮比、催化剂种类与温度的关系如下图。
产率与氨氮比、催化剂种类与温度的关系如下图。
A.温度越高,反应速率一定越快
B.氨化脱硝反应中氨氮物质的量之比越大,脱销效率越高
C.使用催化剂,可降低反应的活化能,加快反应速率,提高平衡产率
D.M点对应的速率
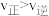
②分析420℃的脱硝效率低于390℃的脱硝效率可能的原因
已知(i)用
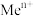 表示金属阳离子、HR表示有机萃取剂,“萃取”原理可以表示为:
表示金属阳离子、HR表示有机萃取剂,“萃取”原理可以表示为: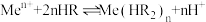
(ii)上述溶剂萃取法(利用萃取剂HR对杂质离子的特殊选择性)与化学净化除杂法(采用分步沉淀法逐一分离杂质离子)
除杂效果对比如下表
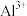 | 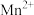 | 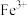 | 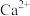 |  | 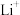 | |
| 化学净化法浸出率/% | 99.7 | 99.74 | 97.85 | 97.97 | 96.87 | 6.1 |
| 溶剂萃取法浸出率/% | 99.54 | 99.39 | 99.21 | 97.89 | 97.07 | 3.3 |
A.紫红色 B.紫色 C.黄色
(2)为提高“焙烧”效率,常采取的措施是
(3)操作1用到的主要玻璃仪器有烧杯、
(4)萃取剂的浓度对萃取率的影响如下图所示,则选择萃取剂浓度为
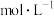 为宜;“反萃取”步骤用到的试剂A是
为宜;“反萃取”步骤用到的试剂A是 外,还有
外,还有
(5)“碳化反应”的化学反应方程式
(6)由除杂效果分析,溶剂萃取法相对于化学净化除杂法的优势为
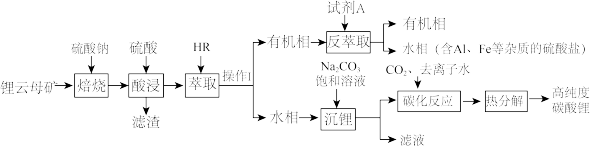
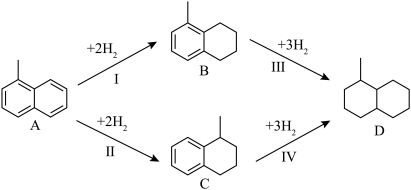
【推荐2】氮化镓(GaN)是一种重要的半导体材料。某工厂利用铝土矿(成分为Al2O3、Ga2O3、Fe2O3等)为原料制备GaN的流程如下图所示:
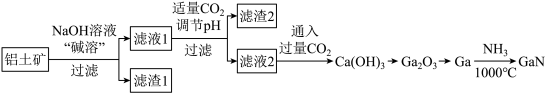
已知:镓与铝同主族,镓的熔点是29.8°C,沸点是2403°C;其氧化物和氢氧化物均为两性化合物;氢氧化物的电离常数如下:
两性氢氧化物 | Al( OH)3 | Ga( OH)3 |
酸式电离常数Ka | 2×10-11 | 1×10-7 |
碱式电离常数Kb | 1.3×10-33 | 1.4×10-34 |
回答下列问题:
(1)为了提高 “碱溶”效率应采用的措施是
(2)滤渣1的成分是
(3)滤液2中通入过量CO2的理由是
(4)流程中利用镓与NH3在1000℃反应生成固体半导体材料 GaN,每生成l molGaN时放出15. 45 kJ热量,写出该反应的热化学方程式
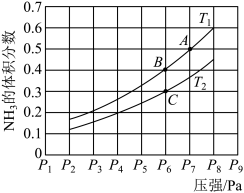
(5)在密闭容器中,充入一定量的Ga与NH3发生反应 ,测得反应平衡体系中NH3的体积分数与压强 P和温度T 的关系曲线如下图所示。图中A 点和C点化学平衡常数的大小关系: KA
(6)已知铝土矿中Ga2O3的质量分数为 a%,某工厂使用 w 吨铝土矿经过一系列反应, 反应过程中镓元素的总利用率为 b%能制得
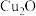 是重要的无机化工原料。某化学兴趣小组同学用下图所示装置(部分加热及夹持装置已省略)制取
是重要的无机化工原料。某化学兴趣小组同学用下图所示装置(部分加热及夹持装置已省略)制取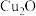 并测定其纯度。
并测定其纯度。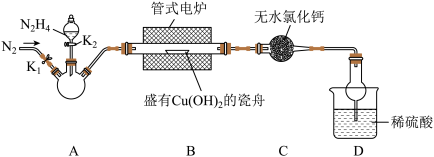
已知:①
 为二元弱碱,常温下为无色油状液体,沸点为113.5℃,有较强的还原性。
为二元弱碱,常温下为无色油状液体,沸点为113.5℃,有较强的还原性。②
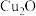 在潮湿的空气中易被氧化,在酸性溶液中易发生歧化反应。
在潮湿的空气中易被氧化,在酸性溶液中易发生歧化反应。③
 为深蓝色,
为深蓝色, 为无色。
为无色。④
 。
。回答下列问题:
(1)通入
 的作用是
的作用是 气流,装置A应选用的加热方式为
气流,装置A应选用的加热方式为(2)充分反应后,取B中固体
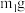 于试管中,加入足量稀硫酸,经过滤、洗涤、干燥后,所得固体质量为
于试管中,加入足量稀硫酸,经过滤、洗涤、干燥后,所得固体质量为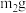 。B中产物溶于稀硫酸的离子方程式为
。B中产物溶于稀硫酸的离子方程式为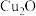 的百分含量为
的百分含量为(3)取B中少量固体产物于试管中,加入足量稀硫酸;继续滴加过量浓氨水,溶液变为深蓝色;一段时间后,溶液颜色逐渐变浅;再加入过量铜粉,久置溶液变为无色。
①取两份等体积的深蓝色溶液于试管中,分别加入等量的水和稀硫酸,观察到现象
 。
。②小组同学猜测最后无色溶液成份为
 ,请设计实验验证
,请设计实验验证(1)请结合下列数据分析,工业上选用氮气与氢气反应固氮,而没有选用氮气和氧气反应固氮的原因是
| 序号 | 化学反应 | K(298K)的数值 |
| ① | 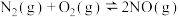 |  |
| ② |  | 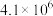 |
(2)合成氨的反应原理为
 ;在一容积为2L的密闭容器中加入一定量的
;在一容积为2L的密闭容器中加入一定量的 和0.5mol
和0.5mol ,在一定条件下发生合成氨反应,反应中
,在一定条件下发生合成氨反应,反应中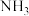 的物质的量浓度的变化情况如下图
的物质的量浓度的变化情况如下图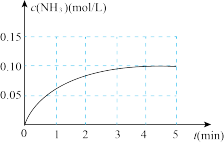
①根据上图,计算从反应开始到平衡时,平均反应速率
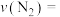
②平衡时测得
 的物质的量为0.10mol,则的转化率
的物质的量为0.10mol,则的转化率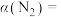
(3)对于反应
 ,在一定条件下氨的平衡含量如下表。
,在一定条件下氨的平衡含量如下表。| 温度/℃ | 压强/MPa | 氨的平衡含量 |
| 200 | 10 | 81.5% |
| 550 | 10 | 8.25% |
②其他条件不变时,温度升高,氨的平衡含量减小的原因是
③哈伯选用的条件是550℃、10MPa,而非200℃、10MPa,可能的原因是
(1)以上四个反应的平衡常数随温度变化的关系如图所示:
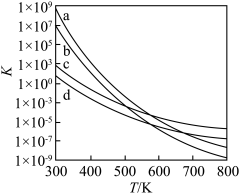
 、
、 、
、 的焓变分别为
的焓变分别为 、
、 、
、 ,则
,则
 的数值范围是
的数值范围是A.<-1 B.-1~0 C.0~1 D.>1
(2)650K,以
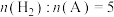 充料,反应达平衡时物质A、B、C、D的物质的量分数
充料,反应达平衡时物质A、B、C、D的物质的量分数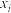 [例如:
[例如: ]随体系压强的变化关系如图所示:
]随体系压强的变化关系如图所示: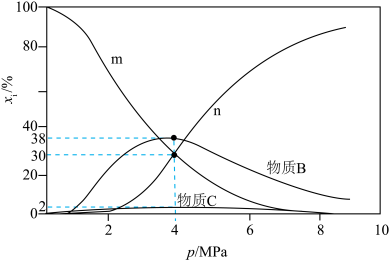
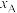 和
和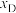 随压强p变化的曲线分别是
随压强p变化的曲线分别是②其他条件不变,若增大
 的比例,则
的比例,则 的值将
的值将③当压强
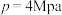 时,
时, 的平衡转化率
的平衡转化率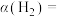
 压强平衡常数
压强平衡常数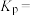
 (列出计算式即可)。
(列出计算式即可)。
(1)NO、NO2体积比为1:1可被NaOH溶液恰好吸收,该反应的化学方程式为
(2)CO在一定条件下可与H2发生如下反应:CO(g)+3H2(g)=CH4(g)+H2O(g)△H=-944 kJ/mol
| 化学键 | C≡O | C-H | O-H | H-H |
| 键能/kJ·mol-1 | x | 413 | 463 | 436 |
则x=
(3)某研究小组用NaOH溶液吸收尾气中的SO2,将得到的Na2SO3溶液进行电解,其中阴、阳膜组合电解装置如图一所示,电极材料为石墨。
①a表示
②阳极的电极反应式为


(4)SO2经过净化后与空气混合进行催化氧化可制取硫酸,其中SO2发生催化氧化的反应为2SO2(g)+O2(g)
 2SO3(g)。若在T1℃、0.1MPa条件下,往一密闭容器中通入SO2和O2[其中n(SO2):n(O2)=2:1],测得容器内总压强与反应时间的关系如图二所示。
2SO3(g)。若在T1℃、0.1MPa条件下,往一密闭容器中通入SO2和O2[其中n(SO2):n(O2)=2:1],测得容器内总压强与反应时间的关系如图二所示。①图中A点时,SO2的转化率为
②在其他条件不变的情况下,测得T2时压强的变化曲线如图二所示,则C点的正反应速率vC(正)与A点的逆反应速率vA(逆)的大小关系为vC(正)
③图中B点的压强平衡常数Kp=
(1)在一定温度下,氧化铁可以与一氧化碳发生反应:Fe2O3(s)+3CO(g)
 2Fe(s)+3CO2(g)。已知该反应在不同温度下的平衡常数如下表:
2Fe(s)+3CO2(g)。已知该反应在不同温度下的平衡常数如下表:| 温度/℃ | 1 000 | 1 150 | 1 300 |
| 平衡常数 | 64.0 | 50.7 | 42.9 |
①该反应的平衡常数表达式K=
②欲提高反应中CO的平衡转化率,可采取的措施是
A.减少Fe的量 B.加入合适的催化剂
C.增大容器的体积 D.降低反应的温度
③在一个容积为1 L的密闭容器中,1000 ℃时加入Fe2O3、CO各2 mol,反应经过10 min后达到平衡。求该时间范围内反应的平均反应速率v(CO)=
(2)饱和Mg(OH)2溶液中滴加FeCl3溶液,产生红褐色沉淀, 反应的离子方程式是
(3)已知:2Fe(s)+3/2O2(g)=Fe2O3(s) △H=−824 kJ·mol-1、2Al(s)+3/2O2(g)=Al2O3(s) △H=−1675.7 kJ·mol-1。则铝与Fe2O3发生反应生成Al2O3和Fe的热化学方程式为 :
请回答下列问题:
(1)煤的气化的主要化学反应方程式为:
(2)煤的气化过程中产生的有害气体H2S用Na2CO3溶液吸收,生成两种酸式盐,该反应的化学方程式为:
(3)利用水煤气合成二甲醚的三步反应如下:
① 2H2(g) + CO(g)
 CH3OH(g);ΔH = -90.8 kJ·mol-1
CH3OH(g);ΔH = -90.8 kJ·mol-1 ② 2CH3OH(g)
 CH3OCH3(g) + H2O(g);ΔH= -23.5 kJ·mol-1
CH3OCH3(g) + H2O(g);ΔH= -23.5 kJ·mol-1 ③ CO(g) + H2O(g)
 CO2(g) + H2(g);ΔH= -41.3 kJ·mol-1
CO2(g) + H2(g);ΔH= -41.3 kJ·mol-1 总反应:3H2(g) + 3CO(g)
 CH3OCH3(g) + CO2 (g)的ΔH=
CH3OCH3(g) + CO2 (g)的ΔH= 一定条件下的密闭容器中,该总反应达到平衡,要提高CO的转化率,可以采取的措施是
a.高温高压 b.加入催化剂 c.减少CO2的浓度
d.增加CO的浓度 e.分离出二甲醚
(4)已知反应②2CH3OH(g)
 CH3OCH3(g) + H2O(g)某温度下的平衡常数为400 。此温度下,在密闭容器中加入CH3OH ,反应到某时刻测得各组分的浓度如下:
CH3OCH3(g) + H2O(g)某温度下的平衡常数为400 。此温度下,在密闭容器中加入CH3OH ,反应到某时刻测得各组分的浓度如下:物质 | CH3OH | CH3OCH3 | H2O |
浓度/(mol·L-1) | 0.44 | 0.6 | 0.6 |
① 比较此时正、逆反应速率的大小:v正
② 若加入CH3OH后,经10 min反应达到平衡,此时c(CH3OH) =
(1)已知:
 变成
变成 吸收
吸收 的能量,在一定条件下,CO与水蒸气反应可制备氢能源,反应过程中的能量变化如图所示:
的能量,在一定条件下,CO与水蒸气反应可制备氢能源,反应过程中的能量变化如图所示:
反应
 为
为 的能量变化曲线,原因是
的能量变化曲线,原因是(2)某工业烟气中含
 ,需进行一系列处理后才能排入空气中,其中脱硝、脱硫就是重要一环,一种脱硝、脱硫的简易流程如图:
,需进行一系列处理后才能排入空气中,其中脱硝、脱硫就是重要一环,一种脱硝、脱硫的简易流程如图: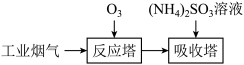
反应塔中涉及如下反应(忽略
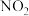 与
与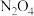 的相互转化):
的相互转化):Ⅰ.

Ⅱ.
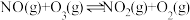
Ⅲ.

①恒温恒容条件下,反应塔中
 ,反应
,反应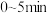 内测得
内测得 ,则
,则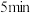 末时NO的转化率为
末时NO的转化率为②保持反应塔中起始浓度
 ,在相同时间内,测得吸收塔中脱硝率、脱硫率与
,在相同时间内,测得吸收塔中脱硝率、脱硫率与 的关系曲线如图所示:
的关系曲线如图所示: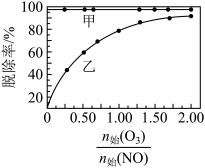
表示脱硫率的曲线是
③在某恒容密闭容器中充入一定量的
 ,发生上述反应Ⅰ、Ⅱ、Ⅲ,下列说法错误的是
,发生上述反应Ⅰ、Ⅱ、Ⅲ,下列说法错误的是a.升高温度,化学反应速率加快
b.容器中混合气体的密度不变说明反应达到平衡状态
c.容器中混合气体的压强不变说明反应达到平衡状态
d.容器中混合气体的颜色不变说明反应达到平衡状态
e.缩小容器的容积,压强增大,脱硝率和脱硫率均不变



