(1)①已如:2SO2(g)+O2(g)⇌2SO3(g) △H=-196.6kJ•mol-1
2NO(g)+O2(g)⇌2NO2(g) △H=-113.0kJ•mol-1
请写出NO2与SO2反应生成SO3(g)和NO的热化学力程 式
②一定条件下,将NO2与SO2以体积比1:2置于2L密闭容器中发生上述反应,下列能说明反应达到平衡状态的是
a.体系压强保持不变 b.混合气体颜色保持不变
c.SO3和NO的体积比保持不变 d.每消耗1molSO2的同时生成1mlINO2
(2)①工 业上一般以CO和H2为原料合成甲醇。该反应的热化学方程式为:CO(g)+2H2(B)
 CH3OH(g) △H=-99kJ•mol-1。则反应物的总能量
CH3OH(g) △H=-99kJ•mol-1。则反应物的总能量②在恒温条件下体积为2L的密闭容器中,将1mol CO与3molH2的混合气体在催化剂作用下发生反应生成甲醇。2min时制得生成CH3OH0.2mol,则0~2min的平均反应速率v(H2)=
③恒温恒容时,改变下列条件能加快以上反应的速率的是
a.充入惰性气体 b.增加CO的量 C.升高温度 d.减小H2的量
相似题推荐
 捕获技术用于去除气流中的
捕获技术用于去除气流中的 或者分离出
或者分离出 作为气体产物,其中
作为气体产物,其中 催化合成甲醇是一种很有前景的方法。如图所示为该反应在无催化剂及有正催化剂时的能量变化(该反应为可逆反应)。
催化合成甲醇是一种很有前景的方法。如图所示为该反应在无催化剂及有正催化剂时的能量变化(该反应为可逆反应)。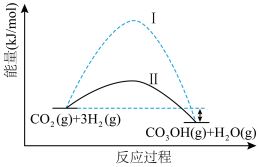
(1)温度为523 K时,测得上述反应中生成
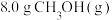 放出的热量为12.3 kJ。反应的热化学方程式为
放出的热量为12.3 kJ。反应的热化学方程式为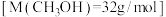
(2)从图可知,有催化剂存在的是过程
(3)一定温度下(各物质均为气态),向体积为2 L的恒容密闭容器中充入
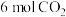 和
和 ,加入合适催化剂进行反应。某时刻测得
,加入合适催化剂进行反应。某时刻测得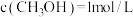 ,此时反应
,此时反应(4)关于
 催化合成甲醇的反应,下列说法中合理的是___________(填字母序号)。
催化合成甲醇的反应,下列说法中合理的是___________(填字母序号)。| A.该反应原子转化率为100% |
B.该反应可用于 的转化,有助于缓解温室效应 的转化,有助于缓解温室效应 |
C.使用催化剂可以降低该反应的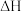 ,从而使反应放出更多热量 ,从而使反应放出更多热量 |
D.降温分离液态甲醇和水,将剩余气体重新通入反应器,可提高 与 与 的利用率 的利用率 |
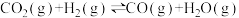
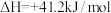 将混合气按进料比
将混合气按进料比 通入反应装置,选择合适催化剂发生反应。不同温度和压强下,
通入反应装置,选择合适催化剂发生反应。不同温度和压强下, 平衡产率和
平衡产率和 平衡转化率分别如图1、图2。
平衡转化率分别如图1、图2。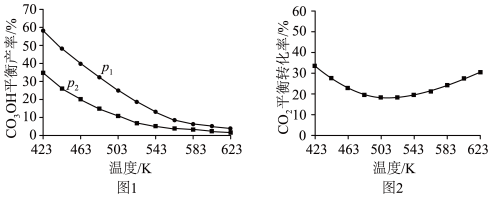
①图1中,压强
 (填“>”“=”或“<”),推断的依据是
(填“>”“=”或“<”),推断的依据是②图2中,压强为
 ,温度高于503 K后,
,温度高于503 K后, 平衡转化率随温度升高而增大的原因是
平衡转化率随温度升高而增大的原因是(1)CH4与CO2经催化反应可制得合成气:CH4(g)+CO2(g)=2CO(g)+2H2(g) ΔH
已知:反应1:CH4(g)=C(s)+2H2(g) ΔH1=+75kJ/mol
反应2: 2CO(g)=C(s)+CO2(g) ΔH2=-172kJ/mol 则该催化反应的ΔH=
(2)工业上可用CO和H2合成二甲醚,其反应为: 2CO(g)+4H2(g)=CH3OCH3(g)+H2O(g) ΔH=-204.7 kJ/mol 初始条件相同时,分别在A(恒温)、B(绝热)两个容器内反应。反应初始时两容器的反应速率A
(3)工业上可用CO和H2合成甲醇,其反应为CO(g)+2H2(g)=CH3OH(g) ΔH=-90.1kJ·md。温度T时,在容积为2L的某密闭容器中进行上述反应,反应过程中相关数据如图所示。
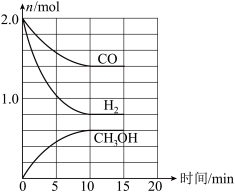
①下列能表明该反应已达到平衡状态的是
a v(H2)=2v(CH3OH) b c( CO)/c( CH3OH)=7: 3时
c c(H2)/c( CH3OH)=2: 1时 d 容器中气体的压强不再变化
②该化学反应10min内生成CH3OH的反应速率v(CH3OH)=
③该温度下,反应的平衡常数为
④若其他条件不变时,15min时再向容器中加入2 mol CO和 x mol H2,平衡时CO的转化率与原平衡相同,则x=
(1)已知:①2Fe(s)+O2(g)=2FeO(s)△H1=﹣544.0kJ•mol-1;
②4Al(s)+3O2(g)=2Al2O3(s)△H2=﹣3351.4kJ•mol-1。
Al和FeO发生铝热反应的热化学方程式是
(2)通常人们把拆开1mol某化学键吸收的能量看成该化学键的键能。下表是一些化学键的键能。
| 化学键 | C-H | C-F | H-F | F-F |
| 键能kJ/mol | 414 | 489 | 565 | 155 |
根据键能数据反应:CH4(g)+4F2(g)=CF4(g)+4HF(g)的反应热△H为
(3)在恒温(500K)、体积为1.0L的密闭容器中通入1.0molN2和1.0molH2发生合成氨反应N2+3H2⇌2NH3,20min后达到平衡,测得反应放出的热量为18.4kJ,混合气体的物质的量为1.6mol,该反应的热化学方程式为
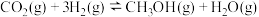 。一定温度下,相关物质的相对能量如下图:
。一定温度下,相关物质的相对能量如下图:
(1)反应
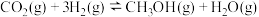 的
的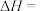
(2)若反应在恒温恒压的密闭容器中进行。
①能说明该反应已达到平衡状态的是
a.容器内混合气体的密度不再改变
b.容器内
 的体积分数保持不变
的体积分数保持不变c.断开
 键同时断开
键同时断开 键
键d.容器内
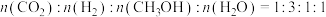
②反应中测得容器内CO2及H2O的物质的量随时间变化如图所示:

图中b点对应的速率关系是
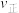
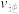 (填“>”“<”或“=”,下同),c点对应的正反应速率和d点对应的逆反应速率的关系是
(填“>”“<”或“=”,下同),c点对应的正反应速率和d点对应的逆反应速率的关系是
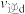 。
。(3)若反应在2L的恒温密闭容器中进行,起始时
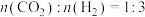 ,测得n(CO2)随时间t变化如表所示。
,测得n(CO2)随时间t变化如表所示。| t/min | 0 | 2 | 4 | 6 | 8 | 10 | 12 | 14 |
| n(CO2)/mol | 2.00 | 1.40 | 1.0 | 0.76 | 0.58 | 0.50 | 0.50 | 0.50 |
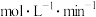 。
。②平衡时H2O(g)的体积分数为
 CH3OH+H2O(g)
CH3OH+H2O(g)(1)已知生成32 g CH3OH放出热量49 kJ,该反应的热化学方程式为
(2)在该反应中,表示该反应速率最快的是:_______
| A.v(CO2)=30 mol·L-1·min-1 | B.v(CH3OH)=0.3 mol·L-1·s-1 |
| C.v(H2)=0.8 mol·L-1·s-1 | D.v(H2O)=0.6 mol·L-1·s-1 |

(4)在一定温度下,将2 mol CO2与6 mol H2气体混合于2 L密闭容器中制甲醇,2 min末生成0.8 mol H2O(g),则:
①用单位时间内CO2浓度的减小来表示2 min内该反应的平均速率为
②2 min末容器中H2的物质的量浓度为
 2CO2+N2。为研究如何提高该转化过程反应速率,某课题组进行了以下实验探究。
2CO2+N2。为研究如何提高该转化过程反应速率,某课题组进行了以下实验探究。 【资料查阅】①不同的催化剂对同一反应的催化效率不同;
②使用相同的催化剂,当催化剂质量相等时,催化剂的比表面积对催化效率有影响。
【实验设计】课题组为探究某些外界条件对汽车尾气转化反应速率的影响规律,设计了以下对比实验。
(1)完成以下实验设计表(表中不要留空格)。
| 实验编号 | 实验目的 | T/℃ | NO初始浓度mol/L | CO初始浓度mol/L | 同种催化剂的比表面积m2/g |
| Ⅰ | 为以下实验作参照 | 280 | 6.00×10-3 | 4.00×10-3 | 80 |
| II | 120 | ||||
| Ⅲ | 探究温度对尾气转化速率的影响 | 360 | 6.00×10-3 | 4.00×10-3 | 80 |

(2)计算第Ⅰ组实验中,达平衡时NO的浓度为
(3)由曲线Ⅰ、Ⅱ可知,增大催化剂比表面积,汽车尾气转化速率_____________(填“增大”、“减小”、“无影响”)。
(II)随着世界粮食需求量的增长,农业对化学肥料的需求量越来越大,其中氮肥是需求量最大的一种化肥。而氨的合成为氮肥的生产工业奠定了基础,其原理为:N2+3H2
 2NH3
2NH3(4)在N2+3H2
 2NH3的反应中,一段时间后,NH3的浓度增加了0.9mol·L-1。用N2表示其反应速率为0.15 mol·L-1·s-1,则所经过的时间为_________;
2NH3的反应中,一段时间后,NH3的浓度增加了0.9mol·L-1。用N2表示其反应速率为0.15 mol·L-1·s-1,则所经过的时间为_________;| A.2 s | B.3 s | C.4 s | D.6 s |
| A.v(H2)=0.1 mol·L-1·min-1 | B.v(N2)=0.1 mol·L-1·min-1 |
| C.v(NH3)=0.15 mol·L-1·min-1 | D.v(N2)=0.002mol·L-1·s-1 |
 2NH3(g) △H<0。下列各项能说明该反应已经达到平衡状态的是 ___________。
2NH3(g) △H<0。下列各项能说明该反应已经达到平衡状态的是 ___________。| A.容器内气体密度保持不变 | B.容器内温度不再变化 | C.断裂1mol N≡N键的同时,生成6 mol N—H键 | D.2v正(H2)=3v逆(NH3) | E.反应混合气体的平均相对分子质量不变 | F.C(H2): C(N2): C(NH3)= 3:1:2 |
(1)已知高炉炼铁过程中会发生如下反应:
FeO(s)+CO(g)=Fe(s)+CO2(g) ΔH1
Fe2O3(s)+
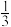 CO(g)=
CO(g)= Fe3O4(s)+
Fe3O4(s)+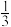 CO2(g) ΔH2
CO2(g) ΔH2Fe3O4(s)+CO(g)=3FeO(s)+CO2(g) ΔH3
Fe2O3(s)+3CO(g)=2Fe(s)+3CO2(g) ΔH4
上述总反应在高炉中大致分为三个阶段,各阶段主要成分与温度的关系如图:
| 温度 | 250℃ | 600℃ | 1000℃ | 2000℃ |
| 主要成分 | Fe2O3 | Fe3O4 | FeO | Fe |
1600℃时固体物质的主要成分为
(2)铁系金属常用作CO加氢反应的催化剂。已知某种催化剂可用来催化反应CO(g)+3H2(g)⇌CH4(g)+H2O(g) ΔH<0。在T℃,压强为p时将1molCO和3molH2加入容积为1L的密闭容器中。实验测得CO的体积分数χ(CO)如表:
| t/min | 0 | 10 | 20 | 30 | 35 |
| χ(CO) | 0.25 | 0.20 | 0.17 | 0.15 | 0.15 |
①前10min内,CO的平均反应速率υ(CO)=
②达到平衡时,体系的总压强p与初始压强之比p0,p:p0=
③如图是该反应CO的平衡转化率与温度、压强的关系。
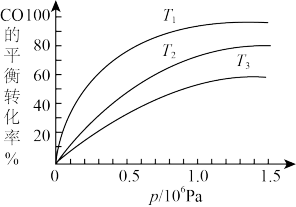
图中温度T1、T2、T3由高到低的顺序是
【推荐2】十八大以来,各地重视“蓝天保卫战”战略。作为煤炭使用大国,我国每年煤炭燃烧释放出的大量SO2严重破坏生态环境。现阶段主流煤炭脱硫技术通常采用石灰石-石膏法将硫元素以CaSO4的形式固定,从而降低SO2的排放。但是煤炭燃烧过程中产生的CO又会与CaSO4发生化学反应,降低脱硫效率。相关反应的热化学方程式如下:
反应Ⅰ:CaSO4(s)+CO(g)⇌CaO(s)+SO2(g)+CO2(g)活化能Ea1,ΔH1=+218.4kJ·mol-1
反应Ⅱ:CaSO4(s)+4CO(g)⇌CaS(s)+4CO2(g)活化能Ea2,ΔH2=-175.6kJ·mol-1
已知活化能Ea1< Ea2。请回答下列问题:
(1)反应CaO(s)+3CO(g)+SO2(g)⇌CaS(s)+3CO2(g);△H=
(2)恒温密闭容器中,加入足量CaSO4和一定物质的量的CO气体,此时压强为p0。t min中时反应达到平衡,此时CO和CO2体积分数相等,CO2是SO2体积分数的2倍,则反应I的平衡常数Kp=
(3)下图为1000 K时,在恒容密闭容器中同时发生反应I和II,c(SO2)随时间的变化图象。请分析下图曲线中c(SO2)在0~t2区间变化的原因

(4)下图为实验在恒容密闭容器中,测得不同温度下,反应体系中初始浓度比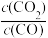 与SO2体积分数的关系曲线。下列有关叙述正确的是
与SO2体积分数的关系曲线。下列有关叙述正确的是

A.当体系中气体的密度不再变化时,反应I和反应Ⅱ都已达到平衡状态
B.提高CaSO4的用量,可使反应I正向进行,SO2体积分数增大
C.其他条件不变,升高温度,有利于反应I正向进行,SO2体积分数增大,不利于脱硫
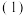 有科学家提出可利用FeO吸收和利用
有科学家提出可利用FeO吸收和利用 ,相关热化学方程式如下:
,相关热化学方程式如下: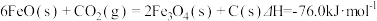
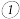 上述反应中每生成
上述反应中每生成 ,转移电子的物质的量为
,转移电子的物质的量为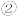 已知:
已知: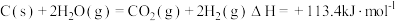 则反应:
则反应: 的
的
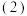 用氨水捕集烟气中的
用氨水捕集烟气中的 生成铵盐是减少
生成铵盐是减少 排放的可行措施之一。
排放的可行措施之一。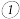 分别用不同pH的吸收剂吸收烟气中的
分别用不同pH的吸收剂吸收烟气中的 ,
, 脱除效率与吸收剂的pH关系如下图所示,若烟气中
脱除效率与吸收剂的pH关系如下图所示,若烟气中 的含量
的含量 体积分数)为
体积分数)为 ,烟气通入氨水的流量为
,烟气通入氨水的流量为 ,标准状况,用pH为
,标准状况,用pH为 的氨水吸收烟气30min,脱除的
的氨水吸收烟气30min,脱除的 的物质的量最多为
的物质的量最多为 精确到
精确到 。
。
 通常情况下温度升高,
通常情况下温度升高, 脱除效率提高,但高于
脱除效率提高,但高于 时,脱除
时,脱除 效率降低的主要原因是
效率降低的主要原因是 一定条件下,
一定条件下, 催化剂可使
催化剂可使 “甲烷化”从而变废为宝,其反应机理如上图所示
“甲烷化”从而变废为宝,其反应机理如上图所示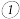 该反应的化学方程式为
该反应的化学方程式为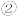 向一容积为2L恒容密闭容器中充入一定量的
向一容积为2L恒容密闭容器中充入一定量的 和
和 ,在
,在 时发生上述反应,达到平衡时各物质的浓度分别为
时发生上述反应,达到平衡时各物质的浓度分别为 :
: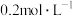 、
、 :
: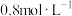 、
、 :
: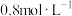 、
、 :
: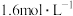 ,则
,则 的平衡转化率为
的平衡转化率为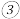 已知该反应正反应放热,现有两个相同的恒容绝热与外界无热量交换
已知该反应正反应放热,现有两个相同的恒容绝热与外界无热量交换 密闭容器Ⅰ、Ⅱ,在I中充入
密闭容器Ⅰ、Ⅱ,在I中充入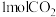 和
和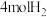 ,在Ⅱ中充入
,在Ⅱ中充入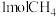 和
和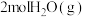 ,
, 下开始反应。达到平衡时,下列说法正确的是
下开始反应。达到平衡时,下列说法正确的是 填字母
填字母
A.容器Ⅰ、Ⅱ中正反应速率相同
B.容器Ⅰ、Ⅱ中
 的物质的量分数相同
的物质的量分数相同C.容器Ⅰ中
 的物质的量比容器Ⅱ中的多
的物质的量比容器Ⅱ中的多D.容器Ⅰ中
 的转化率与容器Ⅱ中
的转化率与容器Ⅱ中 的转化率之和小于1
的转化率之和小于1
(1)地下水中的氮污染主要是由
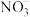 引起的,人们对
引起的,人们对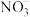 的转化进行了长时间的研究,目前主要有物理方法、化学方法和生物方法,其中化学方法主要包含活泼金属还原法和催化反硝化法。催化反硝化法是一种经济可行的脱氮方法,其原理是在Pd/Cu双金属催化剂作用下,
的转化进行了长时间的研究,目前主要有物理方法、化学方法和生物方法,其中化学方法主要包含活泼金属还原法和催化反硝化法。催化反硝化法是一种经济可行的脱氮方法,其原理是在Pd/Cu双金属催化剂作用下, 将硝酸盐还原成氮气:
将硝酸盐还原成氮气: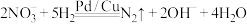 的
的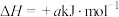 ,
,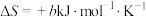 (a、b均为正值),该反应
(a、b均为正值),该反应(2)热解
 制
制 。根据文献,将
。根据文献,将 和
和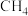 的混合气体导入石英管反应器热解(一边进料,另一边出料),发生如下反应:
的混合气体导入石英管反应器热解(一边进料,另一边出料),发生如下反应:Ⅰ.

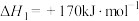
Ⅱ.
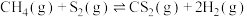
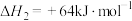
总反应:Ⅲ.
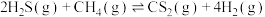
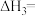
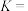
(3)将等物质的量的A、B混合于2L的密闭容器中,发生如下反应:
 ,经5min后,测得D的浓度为0.5mol/L,
,经5min后,测得D的浓度为0.5mol/L, ,C的平均反应速率为0.1mol/(L·min)。求:此时A的浓度
,C的平均反应速率为0.1mol/(L·min)。求:此时A的浓度
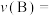
(4)已知可逆反应
 ,平衡常数为K。557℃时,若起始浓度:CO为
,平衡常数为K。557℃时,若起始浓度:CO为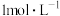 ,
, 为
为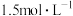 ,平衡时CO转化率为60%,则
,平衡时CO转化率为60%,则 的转化率为
的转化率为(1)已知甲醇是一种清洁燃料,制备甲醇是煤液化的重要方向。若已知H2(g)、CO(g)、CH3OH(l)的燃烧热分别为∆H=-285.8kJ/mol、△H=-283.0kJ/mol、△H=-726.5kJ/mol,CO(g)+2H2(g)
 CH3OH(l)△H=
CH3OH(l)△H=(2)一定温度下,一定可以提高甲醇合成速率的措施有
a.增大起始通入
 值
值b.恒温恒容,再通入氦气
c.使用新型的高效正催化剂
d.将产生的甲醇及时移走
e.压缩体积,增大压强
(3)在恒温恒容条件下,下列说法可以判定反应CO(g)+2H2(g)
 CH3OH(g)已经达到平衡状态的是
CH3OH(g)已经达到平衡状态的是a.体系中碳氢单键数目不再改变
b.体系中n(CO):n(H2)不再改变
c.体系中压强或者平均摩尔质量不再改变
d.单位时间内消耗氢气和CH3OH的物质的量相等
(4)在恒压的容器中,曲线X、Y、Z分别表示在T1°C、T2°C和T3°C三种温度下合成甲醇气体的过程。控制不同的原料投料比,CO的平衡转化率如图所示:
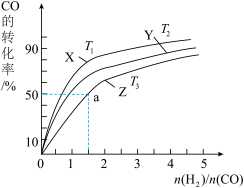
①温度T1°C、T2°C和T3°C由高到低的顺序为:
②若温度为T3°C时,体系压强保持50aMPa,起始反应物投料比n(H2)/n(CO)=1.5,则平衡时CO和CH3OH的分压之比为
 对资源综合利用有重要意义。工业上用
对资源综合利用有重要意义。工业上用 氧化
氧化 制
制 主要化学反应有:
主要化学反应有:a.


b.
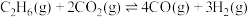
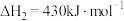
c.
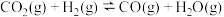
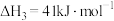
(1)上述反应体系在一定条件下建立平衡后,下列说法正确的有_______(填编号)。
A.增大的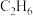 浓度,反应a、b的正反应速率都增加 浓度,反应a、b的正反应速率都增加 |
| B.反应b有助于乙烷脱氢,有利于乙烯生成 |
C.加入反应a的催化剂,可提高的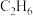 平衡转化率 平衡转化率 |
| D.降低反应温度,反应a~c的正、逆反应速率都减小 |
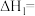
 。
。
(3)乙烷在一定条件可发生如下反应:
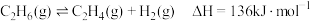
①提高该反应平衡转化率的方法有
②一定温度下,原料组成为
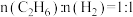 、初始总压为100kPa的恒容密闭容器中进行反应,体系达到平衡时
、初始总压为100kPa的恒容密闭容器中进行反应,体系达到平衡时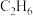 的平衡转化率为20%,平衡时体系的压强为
的平衡转化率为20%,平衡时体系的压强为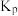 写出计算过程。(用平衡分压代替平衡浓度计算,分压=总压×物质的量分数)
写出计算过程。(用平衡分压代替平衡浓度计算,分压=总压×物质的量分数)



