1 . 常温时,用石墨电极电解500mLNaCl和CuSO4混合溶液,当电路中通过2mol电子时,两极均产生标准状况下16.8L气体,其中阳极产生的O2在标准状况下是5.6L(假定产生的气体全部放出,电解前后溶液体积变化忽略不计),求:
(1)原溶液中c(NaCl)=___________ ,c(CuSO4)=___________ 。
(2)电解后溶液的c(OH-)=___________ 。
(1)原溶液中c(NaCl)=
(2)电解后溶液的c(OH-)=
您最近一年使用:0次
解题方法
2 . 用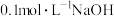 溶液滴定体积均为
溶液滴定体积均为 、浓度均为
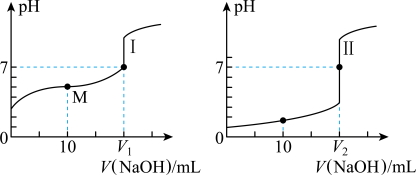
、浓度均为 盐酸和醋酸溶液得到如图所示的滴定曲线:
盐酸和醋酸溶液得到如图所示的滴定曲线:
(1)用 溶液滴定醋酸溶液通常所选择的指示剂为
溶液滴定醋酸溶液通常所选择的指示剂为______ (选填“酚酞”、“甲基橙”或“石蕊”),符合其滴定曲线的是______ (选填“Ⅰ”或“Ⅱ”)。
(2) 盐酸中,由水电离出的
盐酸中,由水电离出的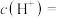
______ 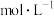 。
。
(3)图中 和
和 大小的比较:
大小的比较:
______ (选填“<”或“=”或“>”) 。
。
(4)在 时,有
时,有 为
为 的盐酸和
的盐酸和 为
为 的
的 溶液,
溶液, 该盐酸恰好中和
该盐酸恰好中和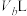 该
该 溶液。
溶液。
①若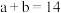 ,则
,则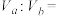
______ (填数字)。
②若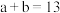 ,则
,则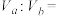
______ (填数字)。
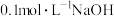 溶液滴定体积均为
溶液滴定体积均为 、浓度均为
、浓度均为 盐酸和醋酸溶液得到如图所示的滴定曲线:
盐酸和醋酸溶液得到如图所示的滴定曲线:
(1)用
 溶液滴定醋酸溶液通常所选择的指示剂为
溶液滴定醋酸溶液通常所选择的指示剂为(2)
 盐酸中,由水电离出的
盐酸中,由水电离出的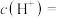
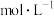 。
。(3)图中
 和
和 大小的比较:
大小的比较:
 。
。(4)在
 时,有
时,有 为
为 的盐酸和
的盐酸和 为
为 的
的 溶液,
溶液, 该盐酸恰好中和
该盐酸恰好中和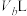 该
该 溶液。
溶液。①若
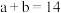 ,则
,则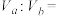
②若
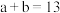 ,则
,则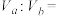
您最近一年使用:0次
23-24高二·全国·假期作业
3 . 已知由C、H、O元素组成的某化合物9.2 g完全燃烧后可以得到17.6 g CO2和10.8 g H2O,通过计算确定该化合物的实验式_______ 。(写出解题过程)
您最近一年使用:0次
4 . 已知: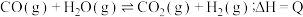 ,其平衡常数随温度变化如下表所示。
,其平衡常数随温度变化如下表所示。
(1)上述反应的化学平衡常数表达式为
___________ 。
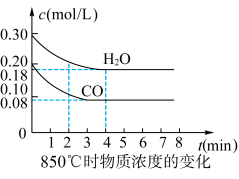
(2) 时,向体积为
时,向体积为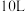 反应器中通入一定量的
反应器中通入一定量的 和
和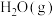 ,发生上述反应,
,发生上述反应, 和
和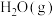 浓度变化如图所示,则
浓度变化如图所示,则 时,平均反应速率
时,平均反应速率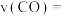
___________ 。
(3)若在 时进行,且
时进行,且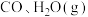 的起始浓度均为
的起始浓度均为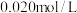 ,该条件下,
,该条件下, 的最大转化率为
的最大转化率为___________
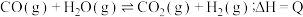 ,其平衡常数随温度变化如下表所示。
,其平衡常数随温度变化如下表所示。温度 | 400 | 500 | 850 |
| 平衡常数 | 9.94 | 9 | 1 |
(1)上述反应的化学平衡常数表达式为

(2)
 时,向体积为
时,向体积为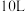 反应器中通入一定量的
反应器中通入一定量的 和
和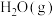 ,发生上述反应,
,发生上述反应, 和
和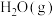 浓度变化如图所示,则
浓度变化如图所示,则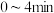 时,平均反应速率
时,平均反应速率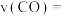

(3)若在
 时进行,且
时进行,且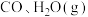 的起始浓度均为
的起始浓度均为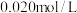 ,该条件下,
,该条件下, 的最大转化率为
的最大转化率为
您最近一年使用:0次
名校
解题方法
5 . 研究化学反应中的能量变化对生产、生活有重要的意义。
(1)甲烷是一种高效清洁的新能源, 完全燃烧生成液态水时放出的热量为
完全燃烧生成液态水时放出的热量为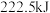 ,则表示
,则表示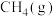 燃烧热的热化学方程式为
燃烧热的热化学方程式为___________ 。
(2)已知: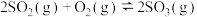

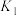
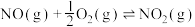

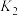
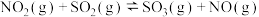

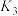
则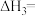
___________ (填含 、
、 的表达式),
的表达式),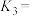
___________ (填含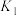 、
、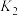 的表达式)。
的表达式)。
(3)已知: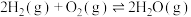
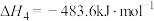 。查阅文献资料得部分化学键的键能如表所示:
。查阅文献资料得部分化学键的键能如表所示:
①合成氨反应 的活化能
的活化能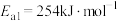 ,则氨分解反应
,则氨分解反应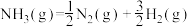 的活化能
的活化能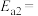
___________  。
。
②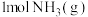 完全燃烧生成
完全燃烧生成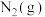 和水蒸气时放出
和水蒸气时放出___________ kJ的热量;又已知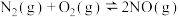
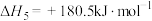 ,则氨催化氧化的热化学方程式为
,则氨催化氧化的热化学方程式为___________ 。
(4)研究氮氧化物与大气中悬浮的海盐粒子的相互作用时,涉及反应:

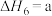
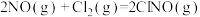
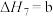
则反应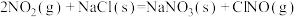 的
的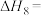
___________ (用含a、b的表达式表示)。
(1)甲烷是一种高效清洁的新能源,
 完全燃烧生成液态水时放出的热量为
完全燃烧生成液态水时放出的热量为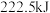 ,则表示
,则表示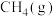 燃烧热的热化学方程式为
燃烧热的热化学方程式为(2)已知:
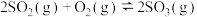

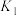
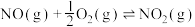

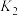
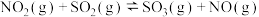

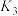
则
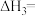
 、
、 的表达式),
的表达式),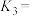
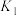 、
、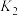 的表达式)。
的表达式)。(3)已知:
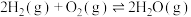
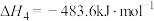 。查阅文献资料得部分化学键的键能如表所示:
。查阅文献资料得部分化学键的键能如表所示:| 化学键 | H—H |  | N—H |
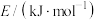 | 436 | 946 | 391 |
 的活化能
的活化能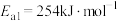 ,则氨分解反应
,则氨分解反应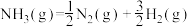 的活化能
的活化能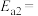
 。
。②
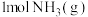 完全燃烧生成
完全燃烧生成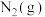 和水蒸气时放出
和水蒸气时放出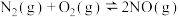
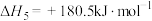 ,则氨催化氧化的热化学方程式为
,则氨催化氧化的热化学方程式为(4)研究氮氧化物与大气中悬浮的海盐粒子的相互作用时,涉及反应:

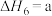
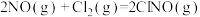
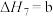
则反应
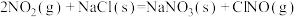 的
的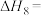
您最近一年使用:0次
2023-11-13更新
|
212次组卷
|
2卷引用:福建省泉州市2023-2024学年高二上学期期中考试化学试题
名校
解题方法
6 . 完成下列计算:
(1)已知工业上制取硫酸需要发生以下反应
Ⅰ. ;Ⅱ.
;Ⅱ.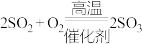 ;Ⅲ.
;Ⅲ.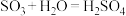
①假设在生产中所有反应完全进行,则每生产1.00吨98%的浓硫酸需要
______ 吨。(保留2位小数)
②实际生产中可能会有损耗(即存在反应物没有发生反应),假设反应Ⅰ中 损耗率为20%,反应Ⅱ中
损耗率为20%,反应Ⅱ中 损耗率为10%,则实际生产1吨98%浓硫酸需要
损耗率为10%,则实际生产1吨98%浓硫酸需要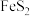
______ 吨。(保留2位小数)
(2)取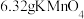 固体,加热一段时间后,剩余固体5.84g。该剩余固体与足量的浓盐酸在加热条件下充分反应生成氯气,产物中所有Mn元素均转化为
固体,加热一段时间后,剩余固体5.84g。该剩余固体与足量的浓盐酸在加热条件下充分反应生成氯气,产物中所有Mn元素均转化为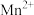 。
。
① 分解生成氧气的物质的量
分解生成氧气的物质的量
______ mol
② 分解的百分率为
分解的百分率为______ %;
③第二步反应中生成氯气的物质的量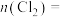
______ mol
(1)已知工业上制取硫酸需要发生以下反应
Ⅰ.
 ;Ⅱ.
;Ⅱ.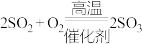 ;Ⅲ.
;Ⅲ.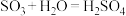
①假设在生产中所有反应完全进行,则每生产1.00吨98%的浓硫酸需要
②实际生产中可能会有损耗(即存在反应物没有发生反应),假设反应Ⅰ中
 损耗率为20%,反应Ⅱ中
损耗率为20%,反应Ⅱ中 损耗率为10%,则实际生产1吨98%浓硫酸需要
损耗率为10%,则实际生产1吨98%浓硫酸需要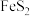
(2)取
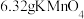 固体,加热一段时间后,剩余固体5.84g。该剩余固体与足量的浓盐酸在加热条件下充分反应生成氯气,产物中所有Mn元素均转化为
固体,加热一段时间后,剩余固体5.84g。该剩余固体与足量的浓盐酸在加热条件下充分反应生成氯气,产物中所有Mn元素均转化为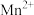 。
。①
 分解生成氧气的物质的量
分解生成氧气的物质的量
②
 分解的百分率为
分解的百分率为③第二步反应中生成氯气的物质的量
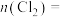
您最近一年使用:0次
2023-11-06更新
|
180次组卷
|
2卷引用:11-2023新东方高二上期中考化学11
名校
解题方法
7 . 将等物质的量的A、B混合于2升的密闭容器中,发生下列反应:3A(g)+B(g)=xC(g)+2D(g),经2min后测得D的浓度为0.5mol•L-1,c(A):c(B)=3:5,以C表示的平均反应速率v(C)=0.25mol•L-1•min-1。求(要求写出计算过程):
(1)v(B)=________ 。
(2)x=________ 。
(3)2min时A的物质的量浓度为_________ 。
(1)v(B)=
(2)x=
(3)2min时A的物质的量浓度为
您最近一年使用:0次
名校
8 . 回答下列问题:
金刚石和石墨均为碳的同素异形体,它们在氧气不足时燃烧生成一氧化碳,充分燃烧时生成二氧化碳,反应中放出的热量如图所示。
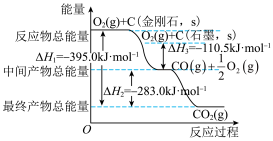
(1)在通常状况下,金刚石和石墨中___________ (填“金刚石”或“石墨”更稳定,石墨的标准燃烧热 为
为___________ 。
(2) 石墨在一定量空气中燃烧,生成气体
石墨在一定量空气中燃烧,生成气体 ,该过程放出的热量为
,该过程放出的热量为___________ 。
(3)已知: 、
、 分子中化学键的键能分别是
分子中化学键的键能分别是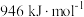 、
、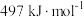 。
。
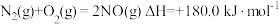
 分子中化学键的键能为
分子中化学键的键能为___________  。
。
(4)综合上述有关信息,请写出 与
与 反应的热化学方程式:
反应的热化学方程式:___________ 。
(5)根据盖斯定律计算
已知下列反应在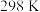 时的反应焓变:
时的反应焓变:
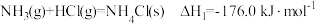
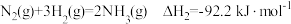
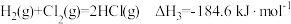
计算反应 在
在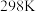 时的焓变
时的焓变___________ 。
金刚石和石墨均为碳的同素异形体,它们在氧气不足时燃烧生成一氧化碳,充分燃烧时生成二氧化碳,反应中放出的热量如图所示。

(1)在通常状况下,金刚石和石墨中
 为
为(2)
 石墨在一定量空气中燃烧,生成气体
石墨在一定量空气中燃烧,生成气体 ,该过程放出的热量为
,该过程放出的热量为(3)已知:
 、
、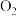 分子中化学键的键能分别是
分子中化学键的键能分别是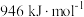 、
、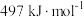 。
。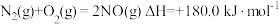
 分子中化学键的键能为
分子中化学键的键能为 。
。(4)综合上述有关信息,请写出
 与
与 反应的热化学方程式:
反应的热化学方程式:(5)根据盖斯定律计算
已知下列反应在
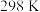 时的反应焓变:
时的反应焓变: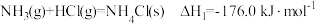
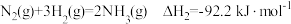
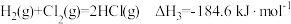
计算反应
 在
在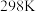 时的焓变
时的焓变
您最近一年使用:0次
9 . 现有含HCl和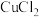 的混合溶液50g,向该溶液中逐滴加入溶质质量分数为10%的NaOH溶液,生成沉淀的质量与加入NaOH溶液的质量关系如图:
的混合溶液50g,向该溶液中逐滴加入溶质质量分数为10%的NaOH溶液,生成沉淀的质量与加入NaOH溶液的质量关系如图:
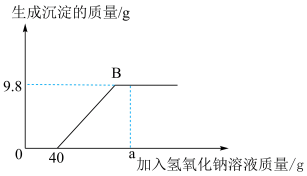
(1)当加入 溶液质量为ag时,溶液中的溶质有
溶液质量为ag时,溶液中的溶质有___________ (写出化学式);
(2)NaOH中Na元素的质量分数___________ (写出计算式);
(3)求混合溶液中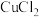 的质量分数
的质量分数___________ (写出计算过程);
(4)求恰好完全反应时,消耗NaOH溶液的总质量___________ (写出计算过程)。
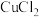 的混合溶液50g,向该溶液中逐滴加入溶质质量分数为10%的NaOH溶液,生成沉淀的质量与加入NaOH溶液的质量关系如图:
的混合溶液50g,向该溶液中逐滴加入溶质质量分数为10%的NaOH溶液,生成沉淀的质量与加入NaOH溶液的质量关系如图:
(1)当加入
 溶液质量为ag时,溶液中的溶质有
溶液质量为ag时,溶液中的溶质有(2)NaOH中Na元素的质量分数
(3)求混合溶液中
 的质量分数
的质量分数(4)求恰好完全反应时,消耗NaOH溶液的总质量
您最近一年使用:0次
2023-10-11更新
|
161次组卷
|
3卷引用:12-2023新东方高二上期中考化学12
12-2023新东方高二上期中考化学12北京市首都师范大学附属中学2023-2024学年高一上学期10月月考化学试题(已下线)【精品卷】3.2.2 物质的量在化学方程式计算中的应用课堂例题-人教版2023-2024学年必修第一册
名校
解题方法
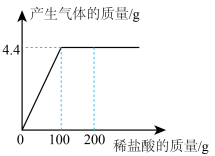
10 . 取CaCl2和CaCO3的混合物12.5g于烧杯中,向其中滴加一定溶质质量分数的稀盐酸,请加稀盐酸的质量与产生气体质量的关系如图所示。求:
(1)混合物CaCO3中的质量为___________ ;
(2)稀盐酸中的溶质质量分数为___________ ;
(3)恰好完全反应时,所得不饱和溶液中溶质的质量分数为___________ ?(写出计算过程,计算结果精确到0.1%)
(1)混合物CaCO3中的质量为
(2)稀盐酸中的溶质质量分数为
(3)恰好完全反应时,所得不饱和溶液中溶质的质量分数为

您最近一年使用:0次



