(1)Deacon直接氧化法可按下列催化过程进行:
2CuCl2(s)=2CuCl(s)+Cl2(g) △H1=+166 kJ·mol-1
2CuCl(s)+O2(g)=2CuO(s)+Cl2(g) △H2=-40kJ·mol-1
CuO(s)+2HCl(g)=CuCl2(s)+H2O(g) △H3=-121 kJ·mol-1
则上述总反应的热化学方程式为
(2)在一刚性容器中,当进料浓度比c(HCl):c(O2)=5:1时,实验测得HCl平衡转化率随温度变化的a(HCl)—T曲线如图所示:
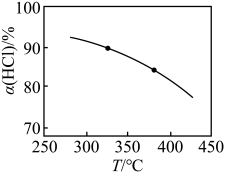
| 平衡时HCl转化率a | 93% | 90% | 87% | 85% | 82% |
| T ℃ | 250 | 300 | 350 | 400 | 450 |
A.HCl与O2的物质的量之比不改变
B.HCl与Cl2的物质的量之比等于2:1
C.容器内的压强不再改变
D.断裂n mol H-Cl键的同时形成 n mol H-O键
②250℃、400℃两点的平衡常数K250℃
③温度不变时,进一步提高HCl的转化率的方法是
(3)若在绝热的刚性容器中投入原料比为n(HCl):n(O2)=1:1的反应混合物,起始时总物质的量为8mol,测得反应过程中c(Cl2)的数据如下:
| t/min | 0 | 2.0 | 4.0 | 6.0 | 8.0 | 10.0 |
| c(Cl2)/10-1mol·L-1 | 0 | 1.5 | 5.4 | 6.5 | 7.4 | 8.0 |
②实验发现产生Cl2的速率先增大后减小,分析其原因:
③若在恒温的刚性容器中完成这个反应,15min时候测得Cl2产率为50%,问反应前后的压强之比:
相似题推荐
①2CH4(g)+O2(g)
 2CO(g)+4H2(g) △H
2CO(g)+4H2(g) △H已知如下反应:
②CH4(g)+2O2(g)
 CO2(g)+2H2O(g) △H1=-810kJ/mol
CO2(g)+2H2O(g) △H1=-810kJ/mol③CH4(g)+H2O(g)
 CO(g)+3H2(g) △H2=+225kJ/mol
CO(g)+3H2(g) △H2=+225kJ/mol④CO(g)+H2O(g)
 CO2(g)+H2(g) △H3=-35kJ/mol
CO2(g)+H2(g) △H3=-35kJ/mol请回答下列问题:
(1)试计算反应①的△H=
(2)在某体积固定的绝热密闭容器中发生反应④,下列事实能证明反应已达平衡的是
| A.容器内温度不再变化 | B.混合气体的平均分子量不再变化 |
| C.各组分的物质的量相等 | D.1molH—O键断裂同时有1molH—H键断裂 |

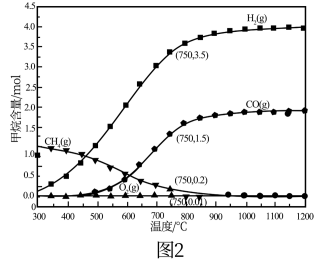
(4)现将2molCH4和1molO2加入某体积可变的密闭容器中,发生反应①。控制压强为1atm,测得不同温度下反应体系中各组分平衡含量如图2所示。实际生产中控制的温度为750℃,则此温度下CO的产率为
(1)已知:①
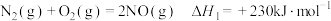
②
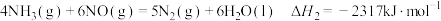
则表示氨气摩尔燃烧焓的热化学方程式为
(2)已知几种化学键的键能和热化学方程式如下:
| 化学键 |  |  |  |  |  |
键能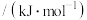 | 391 | 193 | 243 |  | 432 |
 ,则
,则
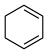
(3)4种不饱和烃分别与氢气发生加成反应生成
 环己烷(
环己烷(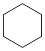 )的能量变化如图所示。根据图示计算反应
)的能量变化如图所示。根据图示计算反应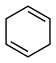 (1)=
(1)= (1)的
(1)的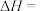
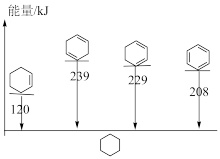
(4)用电解法处理有机废水是目前工业上一种常用手段,电解过程中阳极催化剂表面水被电解产生氧化性强的羟基自由基
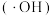 ,羟基自由基再进一步把有机物氧化为无毒物质。下图为电解含1,2-二氯乙烷的废水的装置图,写出电解池阴极的电极反应式:
,羟基自由基再进一步把有机物氧化为无毒物质。下图为电解含1,2-二氯乙烷的废水的装置图,写出电解池阴极的电极反应式: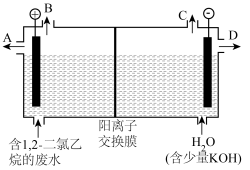
(1)工业上采用高温热分解H2S的方法制取H2,在膜反应器中分离H2,发生的反应为2H2S(g)
 2H2(g)+S2(g) △H。
2H2(g)+S2(g) △H。已知:①H2S(g)
 H2(g)+S(g) △H1;
H2(g)+S(g) △H1;②2S(g)
 S2(g) △H2。
S2(g) △H2。则△H=
(2)土壤中的微生物可将大气中的H2S经两步反应氧化成SO
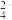 ,两步反应的能量变化示意图如图:
,两步反应的能量变化示意图如图: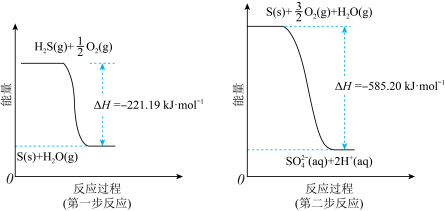
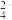 (aq)的热化学方程式为
(aq)的热化学方程式为(3)将H2S和空气的混合气体通入FeCl3、FeCl2、CuCl2的混合溶液中反应回收S,其物质转化如图所示。
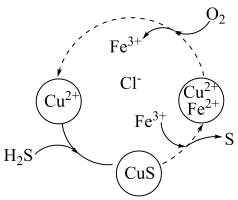
②在温度一定和不补加溶液的条件下,缓慢通入混合气体,并充分搅拌。欲使生成的硫单质中不含CuS,可采取的措施有
(4)二氧化硫—空气质子交换膜燃料电池可以利用大气中所含SO2快速启动,其装置示意图如图所示:
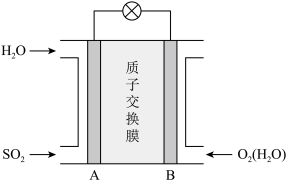
②负极的电极反应为
主反应I:

副反应II:

1.几种化学键的键能如表所示:
| 化学键 |  |  |  |  |
键能/( ) ) | 413 | 436 | 463 | a |

为了进一步研究上述两个反应,某小组在三个10L的刚性容器中,分别充入1mol
 和4mol
和4mol ,在三种不同实验条件(见下表)下进行两个反应,反应体系的总压强(p)随时间变化情况如图所示:
,在三种不同实验条件(见下表)下进行两个反应,反应体系的总压强(p)随时间变化情况如图所示:| 实验编号 | a | b | c |
| 温度/K | 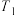 | 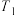 |  |
催化剂的比表面积/(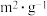 ) ) | 80 | 120 | 120 |
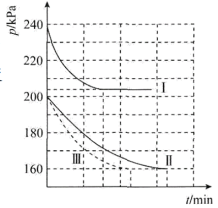
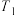 ___________
___________ 。
。| A.大于 | B.等于 | C.小于 | D.无法确定 |
| A.a | B.b | C.c | D.无法确定 |
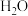 ,则10min内反应的平均速率
,则10min内反应的平均速率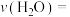
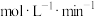 。
。5.该温度下反应II的平衡常数
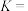 ___________。
___________。| A.1.44 | B.0.44 | C.2.55 | D.0.55 |
 或
或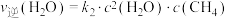 ,反应I的平衡常数
,反应I的平衡常数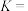
 、
、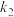 的代数式表示)。
的代数式表示)。
【推荐2】(1)3.2g硫完全燃烧生成稳定的氧化物放出297.2kJ,写出硫燃烧热的热化学方程式:
(2)氮的固定一直是科学家研究的重要课题,合成氨则是人工固氮比较成熟的技术,其原理为N2(g)+3H2(g) 2NH3(g)△H;已知每破坏1mol有关化学键需要的能量如下表:
2NH3(g)△H;已知每破坏1mol有关化学键需要的能量如下表:
H-H | N-H | N-N | N |
435.9kJ | 390.8kJ | 192.8kJ | 945.8kJ |
则△H=
(3)在某一容积为2L的恒容密闭容器中,A、B、C、D四种气体物质发生可逆反应,其物质的量n(mol)随时间t(min)的变化曲线如图所示。回答下列问题:
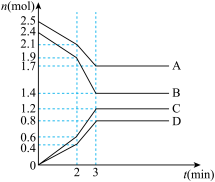
①该反应的化学方程式为
②在2min时,图像发生改变的原因可能是
A.增大B的物质的量B.降低温度
C.加入催化剂D.减小A的物质的量
③不同条件下的该反应的速率:①v(A)=1.0mol·L-1·s-1,②v(B)=1.0 mol·L-1·s-1,③v(C)=1.2 mol·L-1·s-1,④v(D)=1.2mol·L-1·s-1。其中反应速率最快的是
Ⅰ.研究表明,在催化剂存在下,CO2和H2可发生两个平行反应,分别生成CH3OH和CO,反应的热化学方程式如下:反应①
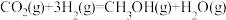
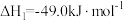
反应②

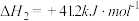
(1)关于上述反应①说法正确的是___________。
| A.△S>0 | B.△S<0 | C.低温自发 | D.高温自发 |
| A.混合气体的平均相对分子质量不变 | B.混合气体的密度不变 |
| C.v (H2)正=v (CO)正 | D.n(CO)/n(H2)不变 |
| A.降低反应温度 | B.增大压强(物质状态不变) |
| C.增大CO2的物质的量 | D.混合气体中掺入一定量惰性气体(不参与反应) |
Ⅱ.T1时,将1.00 mol CO2和3.00molH2充入体积为1.00L的恒容密闭容器中发生反应①,已知容器起始压强为p0,经过3h反应达到平衡,平衡后混合气体的物质的量为3.00mol。
(4)达到平衡时,CO2的转化率为
(5)该过程中H2的平均反应速率为
(6)写出反应①的平衡常数表达式K=
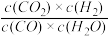 其平衡常数随温度变化如下表所示:请回答下列问题:
其平衡常数随温度变化如下表所示:请回答下列问题:| 温度/℃ | 400 | 500 | 850 |
| 平衡常数 | 9.94 | 9 | K1 |
(1)该反应的ΔH
(2)若在500 ℃时进行上述反应,某时刻测得四种物质的体积分数彼此相等,则此时反应
(3)850 ℃时在一个固定体(反应器中,投入2 molCO和3 molH2O(g),发生上述反应,CO和H2O(g)的浓度变化如图所示,则
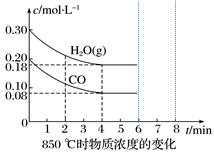
①4 min时H2的物质的量分数=
③若4分钟时测得反应的热效应数值为a kJ,则该反应的热化学方程式为:
④若第6分钟将容器压缩为5L,画出6~8分钟CO的浓度变化图象(注明起点坐标)
(4)t1℃(高于850℃)时,在相同容器中发生上述反应,容器内各物质的浓度随时间变化如下表。
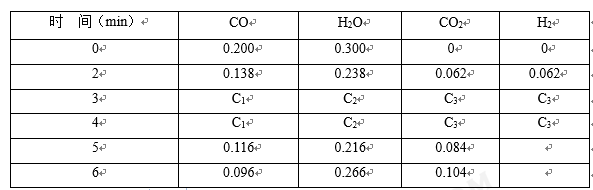
①3~4min时,v正
②反应在4 min~5 min,平衡向逆方向移动,可能的原因是
A.增加水蒸气 B.降低温度 C.使用催化剂 D.增加氢气浓度
 催化加氢合成甲醇。该技术是重要的碳捕获利用与封存技术,为我国碳达峰和碳中和目标提供技术支撑。回答下列问题:
催化加氢合成甲醇。该技术是重要的碳捕获利用与封存技术,为我国碳达峰和碳中和目标提供技术支撑。回答下列问题:(1)已知:反应I.

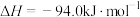
反应Ⅱ.

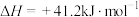
则反应Ⅲ.
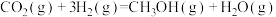 的
的 等于
等于(2)保持温度不变,向体积恒定为2L的密闭容器中,充入
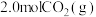 和
和 ,发生反应Ⅲ测得
,发生反应Ⅲ测得 的物质的量如下表所示:
的物质的量如下表所示:| 时间/min | 0 | 2 | 4 | 6 | 8 |
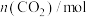 | 2.0 | 1.5 | 1.2 | 1.0 | 1.0 |
 表示的化学反应速率
表示的化学反应速率
②6min时,
 的消耗速率
的消耗速率 的生成速率(填“大于”“小于”或“等于”)。
的生成速率(填“大于”“小于”或“等于”)。③写出既能加快反应Ⅲ的反应速率,又能提高反应Ⅲ中
 转化率的措施:
转化率的措施:(3)在不同条件下,按起始量
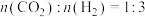 的配比发生反应Ⅲ。下图表示保持压强不变,不同温度时平衡体系中甲醇的物质的量分数
的配比发生反应Ⅲ。下图表示保持压强不变,不同温度时平衡体系中甲醇的物质的量分数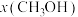 。若210℃到达平衡时容器的体积为1L,则该温度下反应Ⅲ到达平衡
。若210℃到达平衡时容器的体积为1L,则该温度下反应Ⅲ到达平衡 的转化率
的转化率
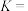
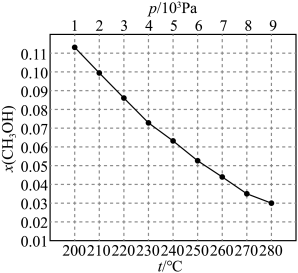
已知化学键键能数据如表:
| 化学键 | C﹣H | C=O | H﹣H | C≡O(CO) | ||
| 键能(kJ/mol) | 413 | 745 | 436 | 1075 |
(1)①CO2与CH4经催化重整,制得合成气:CH4(g)+CO2(g)
 2CO(g)+2H2(g)。则该反应的△H=
2CO(g)+2H2(g)。则该反应的△H=②在恒容绝热条件下,一定能说明该反应已达平衡状态的是
A.混合气体的密度不再变化B.混合气体的平均相对分子质量不再变化
C.容器内的温度不再变化D.v正(CH4)=2v逆(H2)
(2)利用CO2可制取甲醇,其反应为:CO2(g)+3H2(g)
 CH3OH(g)+H2O(g);△H=-49.0KJ/mol;为探究用CO2来生产燃料甲醇的反应原理,现进行如下实验:在恒温恒容密闭容器中,充入2molCO2和6molH2,进行上述反应。测得CO2和CH3OH(g)的浓度随时间变化如图1所示。从反应开始到平衡,v(H2)=
CH3OH(g)+H2O(g);△H=-49.0KJ/mol;为探究用CO2来生产燃料甲醇的反应原理,现进行如下实验:在恒温恒容密闭容器中,充入2molCO2和6molH2,进行上述反应。测得CO2和CH3OH(g)的浓度随时间变化如图1所示。从反应开始到平衡,v(H2)=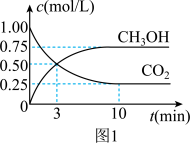
(3)下列措施能使
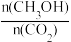 增大的是
增大的是A.降低温度
B.恒温恒容下,再充入2molCO2、6molH2
C.使用高效催化剂
D.恒温恒容充入He(g)使体系压强增大
(4)一种熔融碳酸盐燃料电池原理示意如图2。
①该电池的负极反应式为
②电池工作时,CO32﹣向电极
(1)在尿素合成塔中发生的反应分两步进行,其能量变化和反应历程如图所示:
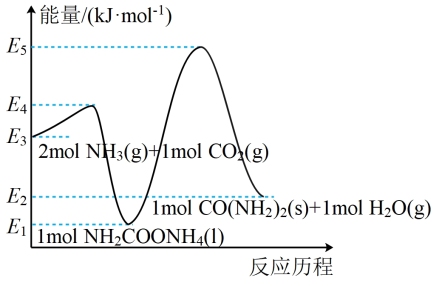
已知:E1=E2-15.5=E3-119.2=E4-129.3=E5-189.4
①图中所示两步反应中,第一步反应是
②有利于第一步反应正向进行的条件是
a.高温、高压 b.高温、低压 c.低温、高压 d.低温、低压
(2)T ℃时,向2 L的密闭容器中,通入2 mol NH3和1 mol CO2,保持体积不变,发生反应2NH3(g)+CO2(g)
 CO(NH2)2(s)+H2O(g),10 min时反应刚好达到平衡。测得起始压强为平衡时压强的1.5倍,则:
CO(NH2)2(s)+H2O(g),10 min时反应刚好达到平衡。测得起始压强为平衡时压强的1.5倍,则:①NH3的平衡转化率为
②能说明上述反应达到平衡状态的是
A.n(CO2)∶n(NH3)=1∶2
B.混合气体的密度不再发生变化
C.单位时间内消耗2 mol NH3,同时生成1 mol H2O
D.CO2的体积分数在混合气体中保持不变
③若10 min时保持温度和压强不变,再向容器中同时充入0.5 mol CO2和0.5 mol H2O(g),则此时平衡
(1)已知:甲烷和乙酸的燃烧热△H分别为-890.31kJ·mol-1、-876.72kJ·mol-1,试写出甲烷与CO2合成乙酸的热化学方程式:
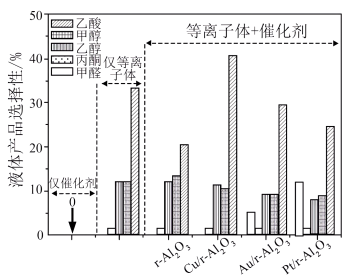
(2)甲烷和二氧化碳一步转化为液体产品的选择性如下图所示,其中选择性最高的产品是
(3)在某一钢性密闭容器中CH4、CO2的分压分别为25kPa、30kPa,加入Ni/α-Al2O3催化剂并加热至1123K使其发生反应:CH4(g)+CO2(g)
 2CO(g)+2H2(g)。
2CO(g)+2H2(g)。①研究表明CO的生成速率υco=1.28×10-2·p(CH4)·p(CO2) (mol·g-1·s-1),某时刻测得p(H2)=20kPa,则p(CH4)=
②达到平衡后测得体系总压是起始时的1.8倍,则该反应的平衡常数Kp=
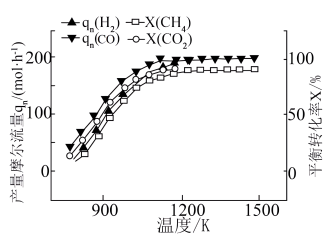
③温度对产物流量及平衡转化率的影响如图所示,可知反应△H
 、
、 等有毒气体,对汽车加装尾气净化装置,可使有毒气体相互反应转化为无毒气体。
等有毒气体,对汽车加装尾气净化装置,可使有毒气体相互反应转化为无毒气体。(1)已知
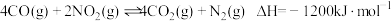
①该反应在
②某实验小组模拟上述净化过程,一定温度下,在容积为
 的恒容密闭容器中起始时按照甲、乙两种方式进行投料,经过一段时间后达到平衡状态,测得甲中
的恒容密闭容器中起始时按照甲、乙两种方式进行投料,经过一段时间后达到平衡状态,测得甲中 的转化率为50%,则该反应的平衡常数为
的转化率为50%,则该反应的平衡常数为 的体积分数:甲
的体积分数:甲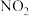 的浓度:甲
的浓度:甲| 甲 | 乙 |
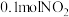 | 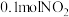 |
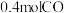 |  |
 和
和 可通过某含钴催化剂催化消除。不同温度下,将模拟尾气(成分如表所示)以相同的流速通过该催化剂测得所有产物(
可通过某含钴催化剂催化消除。不同温度下,将模拟尾气(成分如表所示)以相同的流速通过该催化剂测得所有产物( 、
、 、
、 )与
)与 的相关数据如图所示。
的相关数据如图所示。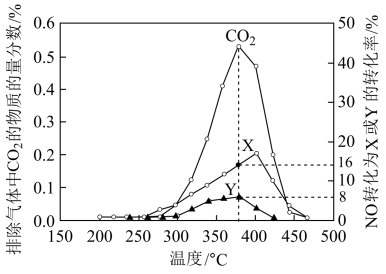
| 模拟尾气 | 气体 | 碳烟 | ||
 |  |  | ||
| 物质的量 | 0.025 | 0.5 | 9.475 | a |
 和
和 ,则Y的化学式为
,则Y的化学式为②实验过程中采用
 模拟
模拟 ,而不采用
,而不采用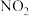 的原因是
的原因是
 N
N

