1 . (1)实验室有一瓶久置的亚硫酸钠溶液,同学分成甲、乙两小组对该瓶亚硫酸钠溶液成分进行实验探究。
【提出问题】该溶液中溶质成分是什么?该溶液中亚硫酸钠的物质的量浓度是多少?
【查阅资料】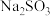 有较强还原性,在空气中易被氧气氧化
有较强还原性,在空气中易被氧气氧化
【作出猜想】猜想 :没有变质,成分是
:没有变质,成分是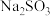
猜想 :完全变质,成分是
:完全变质,成分是
猜想 :部分变质,成分是
:部分变质,成分是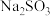 和
和
【实验探究Ⅰ】
(1)甲组设计实验探究溶液是否变质
甲组 | 实验操作 | 现象 | 结论 |
步骤 | 取少量样品于试管中加入过量稀盐酸 | 产生气泡 | 溶液中含有 |
步骤 | 另取样品于试管中,滴加足量氯化钡溶液,再滴加足量稀盐酸 | 溶液中含有 |
实验结论:样品部分变质。样品在空气中变质的化学方程式:
【实验探究Ⅱ】
(2)乙组设计如下实验测定
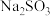 溶液物质的量浓度。
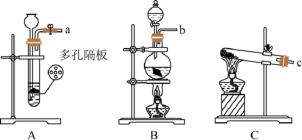
溶液物质的量浓度。①连好装置并检查装置气密性,在锥形瓶中放入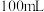 该样品,通入
该样品,通入 。
。
②称量C装置的质量为 。
。
③关闭活塞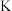 ,用注射器推入浓硫酸至不再产生气泡,注射器可以换为
,用注射器推入浓硫酸至不再产生气泡,注射器可以换为
④打开活塞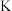 ,再缓缓鼓入一定量的
,再缓缓鼓入一定量的 ,继续通入
,继续通入 的作用是
的作用是
⑤再次称量C装置的质量为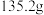 。
。

⑥计算出原溶液中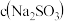 =
= 装置,实验结果将
装置,实验结果将 填“偏大”“不变”或“偏小”
填“偏大”“不变”或“偏小” 。
。
(1)实验室利用铜与浓硫酸制取SO2的化学方程式为
(2)焦亚硫酸钠易被氧化而变质,选用下列试剂设计实验方案,检验焦亚硫酸钠样品氧化变质的程度。
已知:2NaHSO3
 Na2S2O5+H2O。
Na2S2O5+H2O。试剂:稀盐酸、稀H2SO4、稀HNO3、BaCl2溶液、酸性KMnO4溶液、H2O2溶液
| 实验编号 | 实验步骤 | 现象 | 结论 |
| Ⅰ | 取少量样品,加入除氧蒸馏水 | 固体完全溶解得到无色溶液 | / |
| Ⅱ | 取实验I的溶液, | 样品已氧化变质 | |
| Ⅲ | 另取实验I的溶液, | 样品未完全氧化变质 |
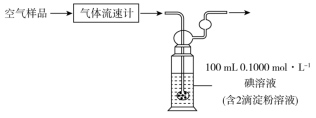
已知该反应的化学方程式为:SO2+I2+2H2O=H2SO4+2HI,若空气流速为a m3·min-1,当观察到
| 选项 | 目的 | 方案设计 | 现象和结论 |
| A | 探究溴乙烷消去反应的产物是乙烯 | 取溴乙烷,加入乙醇、氢氧化钠固体,加热,将产生的气体通入 溶液中 溶液中 | 若 溶液褪色,则溴乙烷消去反应的产物为乙烯 溶液褪色,则溴乙烷消去反应的产物为乙烯 |
| B | 证明酸性:碳酸>苯酚 | 将盐酸与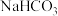 混合产生的气体直接通入苯酚钠溶液 混合产生的气体直接通入苯酚钠溶液 | 若溶液变浑浊,这证明碳酸酸性大于苯酚 |
| C | 探究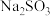 固体样品是否变质 固体样品是否变质 | 取少量待测样品溶于蒸馏水,加入足量稀盐酸,再加入足量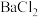 溶液 溶液 | 若有白色沉淀产生,则样品已经变质 |
| D | 验证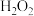 有还原性 有还原性 | 向HCl酸化的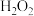 溶液中滴加 溶液中滴加 溶液,溶液褪色 溶液,溶液褪色 | 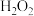 具有还原性 具有还原性 |
| A.A | B.B | C.C | D.D |
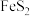 )为原料生产硫酸,并将产出的炉渣和尾气进行资源综合利用,减轻对环境的污染,其中一种流程如图所示:
)为原料生产硫酸,并将产出的炉渣和尾气进行资源综合利用,减轻对环境的污染,其中一种流程如图所示: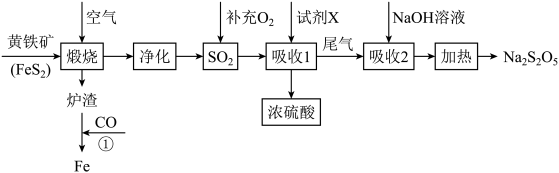
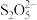 与
与 反应生成沉淀,与盐酸反应生成
反应生成沉淀,与盐酸反应生成 。
。回答下列问题:
(1)煅烧黄铁矿进行粉碎处理的目的是
(2)煅烧过程中主要发生的反应为
 通入酸性高锰酸钾溶液中,可观察到酸性高锰酸钾溶液褪色,,发生该反应的离子方程式为
通入酸性高锰酸钾溶液中,可观察到酸性高锰酸钾溶液褪色,,发生该反应的离子方程式为(3)“吸收1”中试剂
 是
是(4)“吸收2”后得到
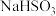 溶液,加热后得
溶液,加热后得 和水,写出加热反应的化学方程式
和水,写出加热反应的化学方程式(5)
 在保存过程中易变质生成
在保存过程中易变质生成 。欲检验
。欲检验 是否变质的具体实验操作方法为:
是否变质的具体实验操作方法为:(6)
 纯度测定:取
纯度测定:取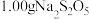 样品溶于水配成
样品溶于水配成 溶液,取
溶液,取 该溶液,向其滴加0.05mol/L碘水10mL,恰好反应完,反应的离子方程式为
该溶液,向其滴加0.05mol/L碘水10mL,恰好反应完,反应的离子方程式为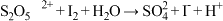 (未配平):样品中
(未配平):样品中 的质量分数为
的质量分数为目的 | 方案设计 | 现象和结论 | |
A | 探究食品脱氧剂中还原铁粉是否变质 | 取少量样品溶于盐酸,滴加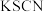 溶液 溶液 | 溶液未变红,说明铁粉未变质 |
B | 证明乙烯的氧化反应 | 将乙烯通入盛有溴水的试管中 | 溴水褪色,说明乙烯被溴水氧化 |
C | 检验某气体是否为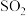 | 将该气体通入到品红溶液中 | 品红溶液褪色,说明该气体是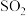 |
D | 比较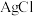 、 、 的 的 大小 大小 | 向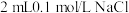 溶液中滴加2滴同浓度 溶液中滴加2滴同浓度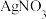 溶液,振荡后再加入4滴同浓度 溶液,振荡后再加入4滴同浓度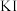 溶液 溶液 | 先产生白色沉淀,后产生黄色沉淀,说明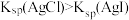 |
| A.A | B.B | C.C | D.D |
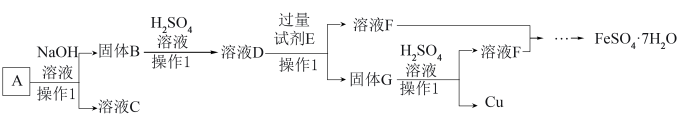
(1)操作1的名称是
(2)溶液C的溶质主要是
(3)检验溶液D中含Fe3+的方法是
(4)FeSO4·7H2O可处理工业废水中有毒的
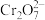 ,反应的离子方程式为Fe2++
,反应的离子方程式为Fe2++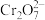 +H+→Fe3++Cr3++H2O(未配平),该反应中,氧化剂与还原剂的物质的量之比为
+H+→Fe3++Cr3++H2O(未配平),该反应中,氧化剂与还原剂的物质的量之比为(5)FeSO4·7H2O由于保存不当容易被空气中氧气氧化而变质,可用酸性KMnO4溶液测定其纯度,测定原理为5Fe2++
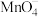 +8H+=5Fe3++Mn2++4H2O。现有一瓶绿矾样品,取出ag于试管中,往其中加入Vmlcmol/L酸性KMnO4溶液恰好完全反应,则绿矾样品的纯度为
+8H+=5Fe3++Mn2++4H2O。现有一瓶绿矾样品,取出ag于试管中,往其中加入Vmlcmol/L酸性KMnO4溶液恰好完全反应,则绿矾样品的纯度为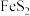 )为原料生产
)为原料生产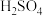 和
和 .
.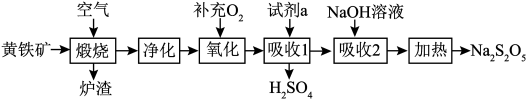
(1)
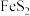 中铁元素的化合价为
中铁元素的化合价为(2)
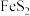 可和盐酸发生复分解反应生成一种不稳定的液态化合物,该化合物的电子式:
可和盐酸发生复分解反应生成一种不稳定的液态化合物,该化合物的电子式:(3)煅烧时主要反应的化学方程式为
(4)“氧化”时,反应的化学方程式为
(5)
 可用于葡萄酒的抗氧化剂,用碘液可以测定葡萄酒中
可用于葡萄酒的抗氧化剂,用碘液可以测定葡萄酒中 的含量,请写出其反应的离子方程式:
的含量,请写出其反应的离子方程式:若要检验
 是否变质生成了
是否变质生成了 ,所用的试剂是
,所用的试剂是(6)在测定某葡萄酒中
 的含量时,取
的含量时,取 葡萄酒样品,消耗
葡萄酒样品,消耗 碘液
碘液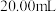 ,样品中
,样品中 的含量为
的含量为 。
。
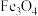 、
、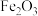 和FeO)制备绿矾(
和FeO)制备绿矾( ))的流程如图:
))的流程如图: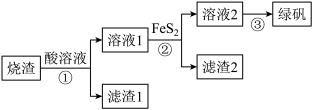
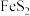 (S的化合价为-1)难溶于水。
(S的化合价为-1)难溶于水。(1)①中加入的酸为
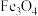 (烧渣的主要成分之一)与酸反应的离子方程式为
(烧渣的主要成分之一)与酸反应的离子方程式为(2)关于制备流程中含铁微粒的描述,错误的是___________。
A.步骤①,硫酸酸溶后溶液中主要有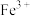 、 、 等正离子 等正离子 |
B.步骤①后可依次用KSCN溶液和酸性 溶液来检测溶液中铁元素的价态 溶液来检测溶液中铁元素的价态 |
| C.Fe元素位于周期表的第ⅥB族 |
D.步骤②中加入硫铁矿的目的是将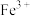 还原为 还原为 |
(3)反应②需在高温高压下进行,且随着反应温度的升高,硫铁矿烧渣酸浸液中
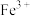 的还原率增加。已知滤渣2的成分中仅有
的还原率增加。已知滤渣2的成分中仅有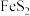 ,请你写出反应②的离子方程式
,请你写出反应②的离子方程式(4)通过③得到绿矾晶体的实验操作:加热浓缩、
(5)绿矾纯度测定:称量2.920g样品于锥形瓶中,溶解后加稀
 酸化,用
酸化,用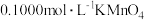 溶液滴定,消耗
溶液滴定,消耗 溶液20.00mL。该样品的纯度为
溶液20.00mL。该样品的纯度为(6)长期放置的
 溶液易被氧化而变质,实验室用绿矾
溶液易被氧化而变质,实验室用绿矾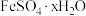 配制
配制 溶液时为了防止
溶液时为了防止 溶液变质,经常向其中加入
溶液变质,经常向其中加入(7)
 可转化为
可转化为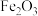 ,
,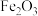 在空气中加热反应可制得铁系氧化物材料。已知
在空气中加热反应可制得铁系氧化物材料。已知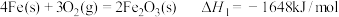
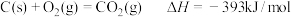

则:4FeCO3(s)+O2(g)
 2Fe2O3(s)+4CO2(g) ΔH=
2Fe2O3(s)+4CO2(g) ΔH=(8)用一氧化碳还原氮氧化物,可防止氮氧化物污染。已知:
①2C(s)+O2(g)=2CO(g) ΔH1=-221kJ•mol-1
②N2(g)+O2(g)=2NO(g) ΔH2=+181kJ•mol-1
③2CO(g)+2NO(g)=2CO2(g)+N2(g) ΔH3=-747kJ•mol-1
则C(s)的燃烧热为
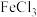 溶液与
溶液与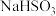 溶液的反应进行探究。
溶液的反应进行探究。(一)配制溶液
(1)配制
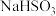 溶液前需检验固体是否变质,其操作方法为:取少量样品于试管中,加适量蒸馏水溶解,再加入
溶液前需检验固体是否变质,其操作方法为:取少量样品于试管中,加适量蒸馏水溶解,再加入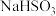 未变质。
未变质。(2)用离子方程式结合适当文字解释:配制氯化铁溶液时,先将氯化铁固体溶于稀盐酸,再稀释到所需浓度的原因
(二)预测实验
(3)该小组同学预测
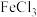 溶液与
溶液与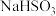 溶液混合时,溶液颜色由棕黄色变成浅绿色,他们预测的依据为(用离子方程式表示):
溶液混合时,溶液颜色由棕黄色变成浅绿色,他们预测的依据为(用离子方程式表示):(三)实施实验
向2mL
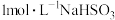 溶液中逐滴加入0.5mL
溶液中逐滴加入0.5mL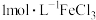 溶液
溶液| 装置 |  |
| 反应时间 | 实验现象 |
| 0~1min | 产生红褐色沉淀,有刺激性气味气体逸出 |
| 1~30min | 沉淀迅速溶解形成红色溶液,随后溶液逐渐变为橙色,之后几乎无色 |
| 30min后 | 与空气接触部分的上层溶液又变为浅红色,随后逐渐变为浅橙色 |
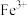 、
、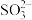 、
、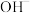 三种微粒会形成红色物质并存在如下转化:
三种微粒会形成红色物质并存在如下转化: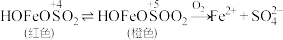
①用离子方程式解释0~1min的实验现象
②从反应速率和化学平衡两个角度解释1~30min的实验现象
(5)小组同学认为没有观察到预期的实验现象,是因为混合产生气体的反应干扰了氧化还原反应的发生,该同学通过如下实验证明了猜想的正确性。已知铁氰化钾溶液可与
 反应生成蓝色沉淀。
反应生成蓝色沉淀。| 装置 |  | |
| 实验步骤 | 操作 | 实验现象 |
| Ⅰ | 向烧杯中装好溶液、连接好装置、闭合开关 | 灵敏电流表指针未发生偏转 |
| Ⅱ | 向左侧烧杯中滴加少量饱和 溶液 溶液 | 灵敏电流表指针向左侧偏转 |
| Ⅲ | 向左侧烧杯中再滴加少量铁氰化钾溶液 | 左侧烧杯中产生蓝色沉淀,灵敏电流表指针向左侧偏转幅度更大 |
②实验Ⅱ中右侧烧杯发生的电极反应式为
| 选项 | 实验目的 | 实验方法或操作 |
| A | 检验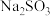 样品是否变质 样品是否变质 | 将样品溶解后加入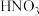 酸化的 酸化的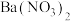 溶液,观察是否产生白色沉淀 溶液,观察是否产生白色沉淀 |
| B | 检验KI溶液是否变质 | 向样品中滴加少量稀硫酸,再滴入2~3滴淀粉溶液,观察溶液颜色变化 |
| C | 检验 样品是否变质 样品是否变质 | 将样品溶解后滴加几滴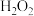 溶液,再滴加 溶液,再滴加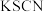 溶液,观察溶液颜色变化 溶液,观察溶液颜色变化 |
| D | 检验漂白粉是否变质 | 将漂白粉溶于水后,滴加浓盐酸,观察是否产生气泡 |
| A.A | B.B | C.C | D.D |



