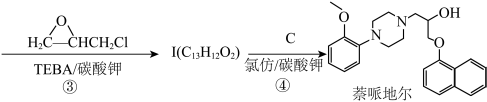
1 . 萘哌地尔是一种常用的肾上腺素受体阻滞药,可由以下工艺合成:
i.合成中间体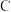 :
:
① ;
;
(1)②的反应类型为___________ ,D中含有的官能团名称为___________ 。
(2) 的化学方程式为
的化学方程式为___________ 。可用 溶液检验
溶液检验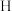 中是否混有
中是否混有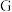 ,其现象为
,其现象为___________ 。
(3)D的同分异构体中,能使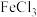 溶液变色的有
溶液变色的有___________ 种;I的结构简式为___________ 。
(4)根据上述信息,写出以1,1-二苯乙烷和苯胺为主要原料制备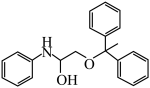 的路线
的路线___________ 。
i.合成中间体
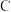 :
: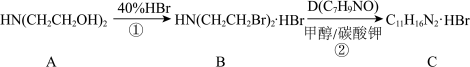
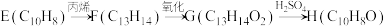
①
 ;
;②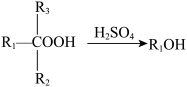 。
。
(1)②的反应类型为
(2)
 的化学方程式为
的化学方程式为 溶液检验
溶液检验 中是否混有
中是否混有 ,其现象为
,其现象为(3)D的同分异构体中,能使
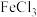 溶液变色的有
溶液变色的有(4)根据上述信息,写出以1,1-二苯乙烷和苯胺为主要原料制备
 的路线
的路线
您最近一年使用:0次
名校
解题方法
2 . 电化学中,常用电极电势表示一定条件下微粒得失电子能力,电极电势越高氧化性越强。标准状态下,氧化型电极反应 ,
,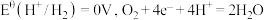 ,
,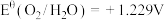 ,则
,则 的氧化性比
的氧化性比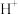 强。含钴微粒的电极电势随
强。含钴微粒的电极电势随 的变化如图所示,两条虚线分别表示“氧线”和“氢线”。下列说法错误的是
的变化如图所示,两条虚线分别表示“氧线”和“氢线”。下列说法错误的是
 ,
,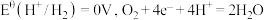 ,
,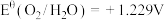 ,则
,则 的氧化性比
的氧化性比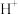 强。含钴微粒的电极电势随
强。含钴微粒的电极电势随 的变化如图所示,两条虚线分别表示“氧线”和“氢线”。下列说法错误的是
的变化如图所示,两条虚线分别表示“氧线”和“氢线”。下列说法错误的是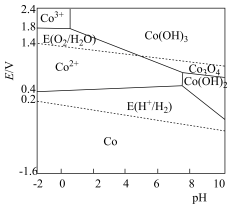
A.单质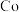 溶于稀硫酸能置换出 溶于稀硫酸能置换出 |
| B.氧气的氧化性与溶液的酸碱性有关 |
C.一定条件下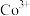 转化为 转化为 ,氧化能力减弱 ,氧化能力减弱 |
D.酸性条件下,反应 难以进行 难以进行 |
您最近一年使用:0次
7日内更新
|
222次组卷
|
5卷引用:2024届山东省潍坊市四县联考高三下学期三模化学试题
2024届山东省潍坊市四县联考高三下学期三模化学试题2024届山东省高三下学期联合模拟考试模拟预测化学试题山东省德州市第一中学2024届下学期三模化学试题湖南省雅礼中学2024届高三下学期热身训练(三模)化学试题(已下线)第04讲 氧化还原反应的基本概念和规律(讲义)-【上好课】2025年高考化学一轮复习讲练测(新教材新高考)
3 . 环戊酮是合成新型降压药物的中间体,实验室制备环戊酮(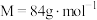 )的反应原理如下:
)的反应原理如下: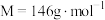 ,熔点为
,熔点为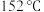 ,
, 左右升华;
左右升华;
b.环戊酮沸点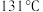 ,着火点
,着火点 。
。
步骤如下:
I.将 己二酸和适量
己二酸和适量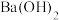 置于蒸馏烧瓶中,维持温度
置于蒸馏烧瓶中,维持温度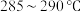 ,均匀加热20分钟;
,均匀加热20分钟;
Ⅱ.在馏出液中加入适量的 浓溶液,振荡锥形瓶、分液除去水层;向有机层中加入
浓溶液,振荡锥形瓶、分液除去水层;向有机层中加入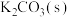 ,振荡分离出有机层;
,振荡分离出有机层;
Ⅲ.对Ⅱ所得有机层进行蒸馏,收集馏分,称量获得产品 ,计算产率。
,计算产率。
对于上述实验,下列说法正确的是
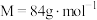 )的反应原理如下:
)的反应原理如下:
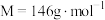 ,熔点为
,熔点为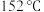 ,
, 左右升华;
左右升华;b.环戊酮沸点
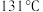 ,着火点
,着火点 。
。步骤如下:
I.将
 己二酸和适量
己二酸和适量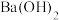 置于蒸馏烧瓶中,维持温度
置于蒸馏烧瓶中,维持温度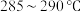 ,均匀加热20分钟;
,均匀加热20分钟;Ⅱ.在馏出液中加入适量的
 浓溶液,振荡锥形瓶、分液除去水层;向有机层中加入
浓溶液,振荡锥形瓶、分液除去水层;向有机层中加入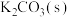 ,振荡分离出有机层;
,振荡分离出有机层;Ⅲ.对Ⅱ所得有机层进行蒸馏,收集馏分,称量获得产品
 ,计算产率。
,计算产率。对于上述实验,下列说法正确的是
| A.步骤I中,为增强冷凝效果应选用球形冷凝管 |
| B.步骤I中,为保证受热均匀应采取水浴加热的方式 |
| C.步骤Ⅱ中,分液操作需要的玻璃仪器有:分液漏斗、烧杯、玻璃棒 |
D.步骤Ⅲ中,应收集 范围内的馏分 范围内的馏分 |
您最近一年使用:0次
4 . 研究含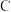 、
、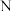 的污染排放物的性质,对建设美丽家乡,打造宜居环境具有重要意义。回答下列问题:
的污染排放物的性质,对建设美丽家乡,打造宜居环境具有重要意义。回答下列问题:
(1)城市中 和
和 污染主要来源于汽车尾气,
污染主要来源于汽车尾气, 在空气中存在如下反应:
在空气中存在如下反应: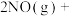
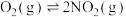 ,研究资料表明该反应的反应速率随温度的升高而减小,上述反应分两步完成,如图1所示。
,研究资料表明该反应的反应速率随温度的升高而减小,上述反应分两步完成,如图1所示。___________ 。
②由实验数据得到第二步反应的 关系如图2所示。当升高温度时,反应重新达到平衡,则
关系如图2所示。当升高温度时,反应重新达到平衡,则 变为相应的点为
变为相应的点为___________ (填字母)。
(2)将 转化为
转化为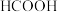 等物质,是实现“双碳”目标的途径之一。
等物质,是实现“双碳”目标的途径之一。
①在某催化剂作用下, 和
和 合成甲酸仅涉及以下反应:
合成甲酸仅涉及以下反应:
I.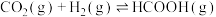

Ⅱ.

在 刚性容器中,加入
刚性容器中,加入 和
和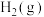 各
各 发生反应,平衡时
发生反应,平衡时 的转化率及
的转化率及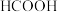 和
和 的选择性随温度变化如图所示。
的选择性随温度变化如图所示。 时,平衡后的压强与初始压强之比为
时,平衡后的压强与初始压强之比为___________ (用含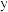 的代数式表示)。
的代数式表示)。 时,反应I的平衡常数
时,反应I的平衡常数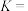
___________ (结果保留小数点后两位)。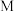 的DMSO(有机溶剂)溶液,反应过程中保持
的DMSO(有机溶剂)溶液,反应过程中保持 和
和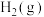 的压强不变,总反应
的压强不变,总反应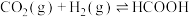
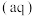 的反应速率为
的反应速率为 ,反应机理如下:
,反应机理如下:
Ⅲ.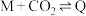
Ⅳ.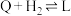
V.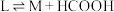
实验测得: ,
,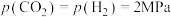 时,
时, 随催化剂
随催化剂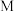 浓度
浓度 变化如图,
变化如图, 时,
时, 不再显著增大的原因是
不再显著增大的原因是___________ 。催化剂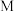 足量条件下,在溶液中加入三乙基胺
足量条件下,在溶液中加入三乙基胺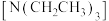 ,
, 的平衡转化率
的平衡转化率___________ (填“增大”“减小”或“不变”)。
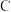 、
、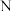 的污染排放物的性质,对建设美丽家乡,打造宜居环境具有重要意义。回答下列问题:
的污染排放物的性质,对建设美丽家乡,打造宜居环境具有重要意义。回答下列问题:(1)城市中
 和
和 污染主要来源于汽车尾气,
污染主要来源于汽车尾气, 在空气中存在如下反应:
在空气中存在如下反应: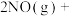
 ,研究资料表明该反应的反应速率随温度的升高而减小,上述反应分两步完成,如图1所示。
,研究资料表明该反应的反应速率随温度的升高而减小,上述反应分两步完成,如图1所示。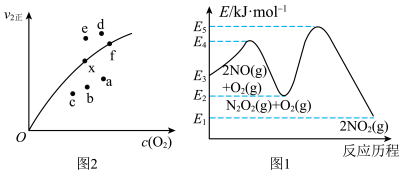
②由实验数据得到第二步反应的
 关系如图2所示。当升高温度时,反应重新达到平衡,则
关系如图2所示。当升高温度时,反应重新达到平衡,则 变为相应的点为
变为相应的点为(2)将
 转化为
转化为 等物质,是实现“双碳”目标的途径之一。
等物质,是实现“双碳”目标的途径之一。①在某催化剂作用下,
 和
和 合成甲酸仅涉及以下反应:
合成甲酸仅涉及以下反应:I.
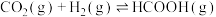

Ⅱ.


在
 刚性容器中,加入
刚性容器中,加入 和
和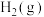 各
各 发生反应,平衡时
发生反应,平衡时 的转化率及
的转化率及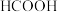 和
和 的选择性随温度变化如图所示。
的选择性随温度变化如图所示。 时,平衡后的压强与初始压强之比为
时,平衡后的压强与初始压强之比为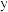 的代数式表示)。
的代数式表示)。 时,反应I的平衡常数
时,反应I的平衡常数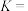

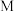 的DMSO(有机溶剂)溶液,反应过程中保持
的DMSO(有机溶剂)溶液,反应过程中保持 和
和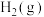 的压强不变,总反应
的压强不变,总反应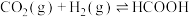
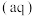 的反应速率为
的反应速率为 ,反应机理如下:
,反应机理如下:Ⅲ.
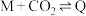
Ⅳ.
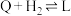
V.
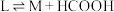
实验测得:
 ,
,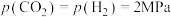 时,
时, 随催化剂
随催化剂 浓度
浓度 变化如图,
变化如图, 时,
时, 不再显著增大的原因是
不再显著增大的原因是 足量条件下,在溶液中加入三乙基胺
足量条件下,在溶液中加入三乙基胺 ,
, 的平衡转化率
的平衡转化率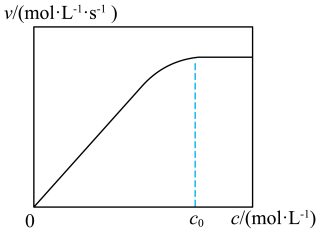
您最近一年使用:0次
解题方法
5 . 钠离子电池具有充电速度快和低温环境性能优越的特点,其电极材料的导电聚合物中掺杂磺酸基可增强其电化学活性,其工作原理如图所示。下列说法正确的是
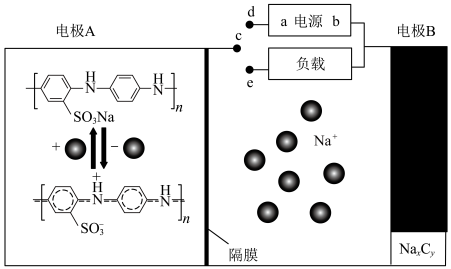
A. 连接 连接 时, 时, 透过隔膜进入电极 透过隔膜进入电极 室 室 |
B. 连接 连接 时,电源 时,电源 极的电势比 极的电势比 极低 极低 |
C. 连接 连接 时,每转移 时,每转移 ,两电极质量变化相差 ,两电极质量变化相差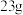 |
D. 连接 连接 时, 时, 的电极反应为: 的电极反应为: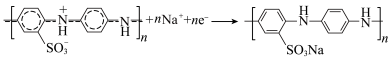 |
您最近一年使用:0次
6 . 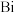 、
、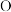 、
、 、
、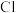 组成的化合物
组成的化合物 通过
通过 空位的掺杂可引入载流子诱发超导,
空位的掺杂可引入载流子诱发超导, 的晶体结构和氧原子沿
的晶体结构和氧原子沿 轴的投影如图所示。回答下列问题:
轴的投影如图所示。回答下列问题: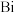 原子中
原子中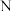 层上的电子空间运动状态有
层上的电子空间运动状态有___________ 种;O、S、Cl电负性由大到小的顺序是___________ 。
(2)氮族元素氢化物(MH3)NH3、PH3、AsH3、SbH3、BiH3键角H-M-H最大的是___________ (填化学式);键长 最长的是
最长的是___________ (填化学式)。已知:配离子的颜色与 电子跃迁的分裂能大小有关,1个电子从较低的
电子跃迁的分裂能大小有关,1个电子从较低的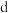 轨道跃迁到较高能量的
轨道跃迁到较高能量的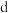 轨道所需的能量为
轨道所需的能量为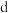 的分裂能,用符号
的分裂能,用符号 表示。分裂能
表示。分裂能
___________  (填“>”“<”或“=”),理由是
(填“>”“<”或“=”),理由是___________ 。
(3)A属四方晶系,晶胞参数如图所示,晶胞棱边夹角均为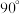 ,该晶胞中Bi原子的配位数为
,该晶胞中Bi原子的配位数为___________ 。若阿伏加德罗常数的值为 ,化合物
,化合物 的密度
的密度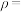
___________ 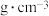 (用含
(用含 的代数式表示)。
的代数式表示)。
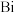 、
、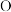 、
、 、
、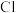 组成的化合物
组成的化合物 通过
通过 空位的掺杂可引入载流子诱发超导,
空位的掺杂可引入载流子诱发超导, 的晶体结构和氧原子沿
的晶体结构和氧原子沿 轴的投影如图所示。回答下列问题:
轴的投影如图所示。回答下列问题: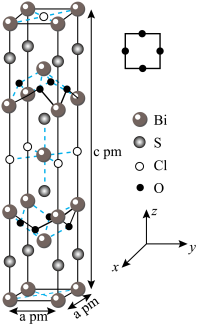
 原子中
原子中 层上的电子空间运动状态有
层上的电子空间运动状态有(2)氮族元素氢化物(MH3)NH3、PH3、AsH3、SbH3、BiH3键角H-M-H最大的是
 最长的是
最长的是 电子跃迁的分裂能大小有关,1个电子从较低的
电子跃迁的分裂能大小有关,1个电子从较低的 轨道跃迁到较高能量的
轨道跃迁到较高能量的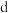 轨道所需的能量为
轨道所需的能量为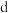 的分裂能,用符号
的分裂能,用符号 表示。分裂能
表示。分裂能
 (填“>”“<”或“=”),理由是
(填“>”“<”或“=”),理由是(3)A属四方晶系,晶胞参数如图所示,晶胞棱边夹角均为
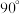 ,该晶胞中Bi原子的配位数为
,该晶胞中Bi原子的配位数为 ,化合物
,化合物 的密度
的密度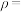
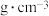 (用含
(用含 的代数式表示)。
的代数式表示)。
您最近一年使用:0次
解题方法
7 . 向 饱和溶液(有足量
饱和溶液(有足量 固体)中滴加
固体)中滴加 溶液,发生反应
溶液,发生反应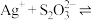
 和
和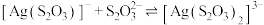 。lg
。lg 、
、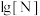 与
与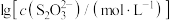 的关系如图所示。
的关系如图所示。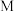 代表
代表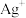 或
或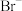 ;
;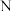 代表
代表 或
或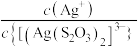 ;忽略
;忽略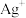 和
和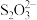 水解)
水解)
下列说法错误的是
 饱和溶液(有足量
饱和溶液(有足量 固体)中滴加
固体)中滴加 溶液,发生反应
溶液,发生反应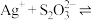
 和
和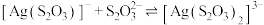 。lg
。lg 、
、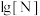 与
与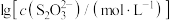 的关系如图所示。
的关系如图所示。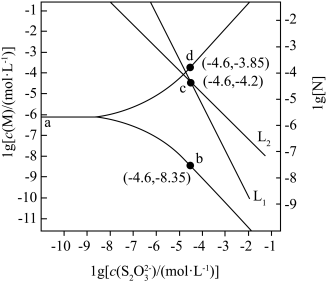
 代表
代表 或
或 ;
;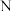 代表
代表 或
或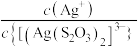 ;忽略
;忽略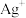 和
和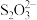 水解)
水解)下列说法错误的是
A. |
B.反应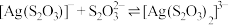 的平衡常数 的平衡常数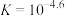 |
C. 点时, 点时,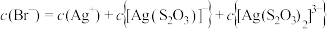 |
D.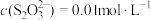 时,若溶液中 时,若溶液中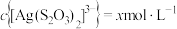 ,则 ,则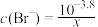 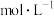 |
您最近一年使用:0次
解题方法
8 .  -月桂烯与
-月桂烯与 的加成反应的部分机理和反应势能变化如图所示。
的加成反应的部分机理和反应势能变化如图所示。
 -月桂烯与
-月桂烯与 的加成反应的部分机理和反应势能变化如图所示。
的加成反应的部分机理和反应势能变化如图所示。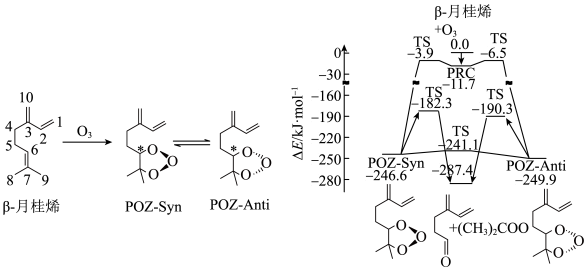
A. -月桂烯与丁基苯互为同分异构体 -月桂烯与丁基苯互为同分异构体 |
| B.POZ-Anti比POZ-Syn更稳定 |
C.升高温度,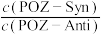 一定减小 一定减小 |
D. -月桂烯与 -月桂烯与 经历两种历程得到 经历两种历程得到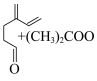 释放的能量相同 释放的能量相同 |
您最近一年使用:0次
9 . 一种工业上利用废旧三元锂离子电池正极材料(主要成分为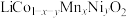 ,还含有铝箔、炭黑、有机粘合剂等)综合回收钴、锰、镍、锂的工艺流程如下图所示:
,还含有铝箔、炭黑、有机粘合剂等)综合回收钴、锰、镍、锂的工艺流程如下图所示: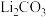 的溶解度随温度升高而减小;
的溶解度随温度升高而减小;
②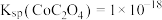 ,
,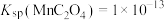 ,
,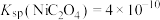 。
。
回答下列问题:
(1)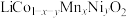 中
中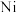 、
、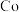 元素的化合价分别为+2价、+3价,若
元素的化合价分别为+2价、+3价,若 ,则
,则 元素的化合价为
元素的化合价为___________ 。锂离子电池中的电解质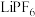 与水相遇会生成
与水相遇会生成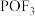 ,该反应的化学方程式为
,该反应的化学方程式为___________ 。
(2)“酸浸”时,下列试剂中最适宜替换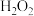 的是
的是___________ 。
a. 溶液 b.
溶液 b. 溶液 c.
溶液 c.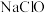 溶液 d.浓盐酸
溶液 d.浓盐酸
(3)若“酸浸”后溶液中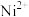 、
、 、
、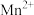 浓度均为
浓度均为 ,欲使
,欲使 完全沉淀(离子浓度小于
完全沉淀(离子浓度小于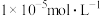 )而不沉淀
)而不沉淀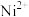 、
、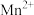 ,理论上需调节溶液
,理论上需调节溶液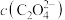 范围为
范围为___________ ;实际“沉钴”时,在加入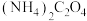 之后溶液中
之后溶液中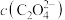 超出
超出 完全沉淀所需浓度,须先加入一定量氨水,所加氨水的作用为
完全沉淀所需浓度,须先加入一定量氨水,所加氨水的作用为___________ 。
(4)“沉锰”过程中加入 溶液后,溶液先变为紫红色,一段时间后紫红色又褪去,紫红色褪去的原因为
溶液后,溶液先变为紫红色,一段时间后紫红色又褪去,紫红色褪去的原因为___________ (用离子方程式表示)。
(5)“沉锂”操作中需将温度升高到 ,原因是:①加快反应速率;②
,原因是:①加快反应速率;②___________ 。
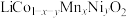 ,还含有铝箔、炭黑、有机粘合剂等)综合回收钴、锰、镍、锂的工艺流程如下图所示:
,还含有铝箔、炭黑、有机粘合剂等)综合回收钴、锰、镍、锂的工艺流程如下图所示: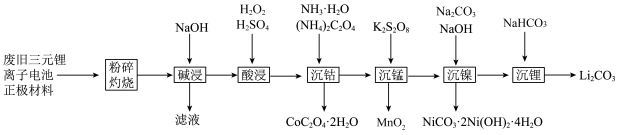
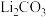 的溶解度随温度升高而减小;
的溶解度随温度升高而减小;②
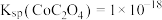 ,
,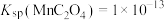 ,
,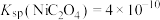 。
。回答下列问题:
(1)
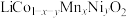 中
中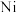 、
、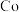 元素的化合价分别为+2价、+3价,若
元素的化合价分别为+2价、+3价,若 ,则
,则 元素的化合价为
元素的化合价为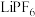 与水相遇会生成
与水相遇会生成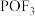 ,该反应的化学方程式为
,该反应的化学方程式为(2)“酸浸”时,下列试剂中最适宜替换
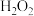 的是
的是a.
 溶液 b.
溶液 b. 溶液 c.
溶液 c.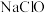 溶液 d.浓盐酸
溶液 d.浓盐酸(3)若“酸浸”后溶液中
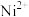 、
、 、
、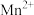 浓度均为
浓度均为 ,欲使
,欲使 完全沉淀(离子浓度小于
完全沉淀(离子浓度小于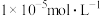 )而不沉淀
)而不沉淀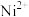 、
、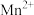 ,理论上需调节溶液
,理论上需调节溶液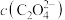 范围为
范围为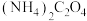 之后溶液中
之后溶液中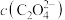 超出
超出 完全沉淀所需浓度,须先加入一定量氨水,所加氨水的作用为
完全沉淀所需浓度,须先加入一定量氨水,所加氨水的作用为(4)“沉锰”过程中加入
 溶液后,溶液先变为紫红色,一段时间后紫红色又褪去,紫红色褪去的原因为
溶液后,溶液先变为紫红色,一段时间后紫红色又褪去,紫红色褪去的原因为(5)“沉锂”操作中需将温度升高到
 ,原因是:①加快反应速率;②
,原因是:①加快反应速率;②
您最近一年使用:0次
10 . 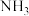 是生产化肥、含氮有机化学品、药物和聚合物的重要化合物,以乙醇为质子导体在电解池中利用连续
是生产化肥、含氮有机化学品、药物和聚合物的重要化合物,以乙醇为质子导体在电解池中利用连续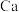 介导(
介导(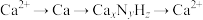 )合成
)合成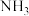 的装置如图所示。下列说法错误的是
的装置如图所示。下列说法错误的是
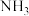 是生产化肥、含氮有机化学品、药物和聚合物的重要化合物,以乙醇为质子导体在电解池中利用连续
是生产化肥、含氮有机化学品、药物和聚合物的重要化合物,以乙醇为质子导体在电解池中利用连续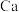 介导(
介导(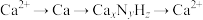 )合成
)合成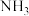 的装置如图所示。下列说法错误的是
的装置如图所示。下列说法错误的是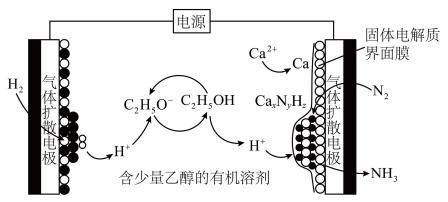
A.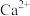 的作用是活化 的作用是活化 |
B.每产生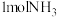 需补充 需补充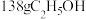 |
C.阴极反应包含如下过程: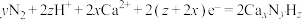 |
| D.电解质溶液可换为含有少量乙醇的水溶液 |
您最近一年使用:0次



