名校
解题方法
1 . 为实现“碳中和”“碳达峰”,碳的循环利用是重要措施。利用氢气和CO反应生成甲烷,涉及的反应如下:
i.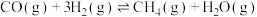
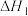
ii.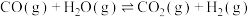

iii.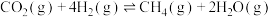

回答下列问题:
(1)在25℃和101kPa下,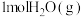 转变为
转变为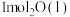 时放出44.0kJ热量,
时放出44.0kJ热量, 的燃烧热为
的燃烧热为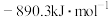 ,CO的燃烧热为
,CO的燃烧热为 ,
, 的燃烧热为
的燃烧热为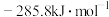 ,则
,则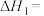
________  。
。
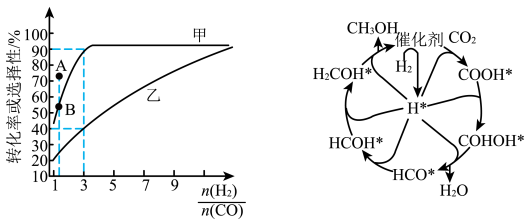
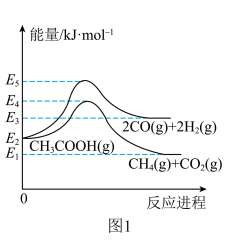
(2)一定温度下,在恒容的密闭容器中进行上述反应,平衡时CO的转化率及 的选择性随
的选择性随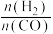 变化的情况如图所示[已知
变化的情况如图所示[已知 的选择性
的选择性 ]。
]。 选择性变化的曲线是
选择性变化的曲线是________ (填“甲”或“乙”),保持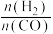 不变,曲线甲由B点达到A点需要的条件为
不变,曲线甲由B点达到A点需要的条件为________ 。
②相同温度下,向恒容容器内加入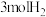 和1molCO,初始压强为10MPa,平衡时
和1molCO,初始压强为10MPa,平衡时 的物质的量为
的物质的量为________ mol,反应ii的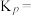
________ (保留小数点后一位)。
(3) 催化加氢制甲醇也是碳循环的重要途径。
催化加氢制甲醇也是碳循环的重要途径。 在某催化剂表面与氢气作用制备甲醇的反应机理如图所示。催化循环中产生的中间体微粒共
在某催化剂表面与氢气作用制备甲醇的反应机理如图所示。催化循环中产生的中间体微粒共________ 种, 催化加氢制甲醇总反应的化学方程式为
催化加氢制甲醇总反应的化学方程式为________ 。
i.
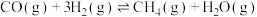
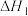
ii.
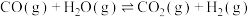

iii.
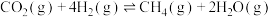

回答下列问题:
(1)在25℃和101kPa下,
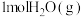 转变为
转变为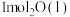 时放出44.0kJ热量,
时放出44.0kJ热量, 的燃烧热为
的燃烧热为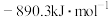 ,CO的燃烧热为
,CO的燃烧热为 ,
, 的燃烧热为
的燃烧热为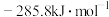 ,则
,则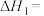
 。
。(2)一定温度下,在恒容的密闭容器中进行上述反应,平衡时CO的转化率及
 的选择性随
的选择性随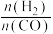 变化的情况如图所示[已知
变化的情况如图所示[已知 的选择性
的选择性 ]。
]。
 选择性变化的曲线是
选择性变化的曲线是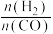 不变,曲线甲由B点达到A点需要的条件为
不变,曲线甲由B点达到A点需要的条件为②相同温度下,向恒容容器内加入
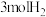 和1molCO,初始压强为10MPa,平衡时
和1molCO,初始压强为10MPa,平衡时 的物质的量为
的物质的量为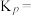
(3)
 催化加氢制甲醇也是碳循环的重要途径。
催化加氢制甲醇也是碳循环的重要途径。 在某催化剂表面与氢气作用制备甲醇的反应机理如图所示。催化循环中产生的中间体微粒共
在某催化剂表面与氢气作用制备甲醇的反应机理如图所示。催化循环中产生的中间体微粒共 催化加氢制甲醇总反应的化学方程式为
催化加氢制甲醇总反应的化学方程式为
您最近一年使用:0次
2024-06-08更新
|
225次组卷
|
7卷引用:2024届青海省西宁市大通县高三下学期三模理科综合试题-高中化学
2 . 火箭推进剂在航天和军事领域具有广泛的应用。
Ⅰ.肼(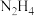 )常温下呈液态,具有弱碱性和强还原性,可与
)常温下呈液态,具有弱碱性和强还原性,可与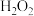 等氧化剂用作火箭燃料。
等氧化剂用作火箭燃料。
(1)肼溶于水形成二元弱碱,碱性比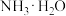 弱,在水中的电离与
弱,在水中的电离与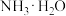 相似,写出肼在水中电离方程式
相似,写出肼在水中电离方程式___________ 。
(2)在标准压强和指定温度(298.15K)下,由元素最稳定的单质生成1mol化合物时的反应热称为该化合物的标准摩尔生成焓。已知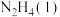 ,
,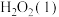 ,
, 的标准摩尔生成焓分别为50.63kJ/mol,-187.78kJ/mol,-241.82kJ/mol。,写出火箭燃料
的标准摩尔生成焓分别为50.63kJ/mol,-187.78kJ/mol,-241.82kJ/mol。,写出火箭燃料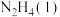 和氧化剂
和氧化剂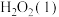 反应生成
反应生成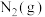 和
和 的热化学方程式:
的热化学方程式:____________ 。
(3)肼(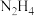 )-双氧水燃料电池由于绿色环保且具有较高的能量密度而被广泛使用,其工作原理如图所示。
)-双氧水燃料电池由于绿色环保且具有较高的能量密度而被广泛使用,其工作原理如图所示。__________________ 。
②设两极区溶液体积均为1 L,溶液的密度为1.2 g/cm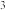 ,忽略溶液体积和密度的变化。当A极产生11.2 L(标准状况)气体时,B极区NaOH溶液的质量分数为40%,计算初始时NaOH溶液的浓度:
,忽略溶液体积和密度的变化。当A极产生11.2 L(标准状况)气体时,B极区NaOH溶液的质量分数为40%,计算初始时NaOH溶液的浓度:______ (用质量分数表示,结果保留三位有效数字)。
Ⅱ.我国的祝融号火星车已于2021年成功登陆火星,火星表面大气的主要成分是 ,占总量的95.32%。科学家们设想通过还原反应将其转化为推进剂以实现星际旅行的愿望,二氧化碳加氢
,占总量的95.32%。科学家们设想通过还原反应将其转化为推进剂以实现星际旅行的愿望,二氧化碳加氢
甲烷化(Sabatier反应)可表示为:
反应1: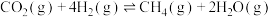
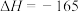 kJ/mol
kJ/mol
反应2:
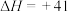 kJ/mol
kJ/mol
将5 mol 和20 mol
和20 mol  通入某恒压密闭容器中将
通入某恒压密闭容器中将 甲烷化,平衡时体系中各含碳元素物质的物质的量n(X)与温度T的关系如图所示。
甲烷化,平衡时体系中各含碳元素物质的物质的量n(X)与温度T的关系如图所示。______ (填化学式),曲线Z所代表的物质在1100K以上物质的量减小的原因是____________ 。
(5)当反应1和反应2均达到化学平衡状态时,若维持温度不变,向容器内通入惰性气体He,则反应2平衡______ (填“正向移动”“逆向移动”或“不移动”)。
(6)800K时,若平衡时容器内总压为p,则反应2的压强平衡常数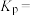
______ (结果保留两位有效数字,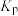 为以分压表示的平衡常数,分压=总压×物质的量分数)。
为以分压表示的平衡常数,分压=总压×物质的量分数)。
Ⅰ.肼(
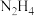 )常温下呈液态,具有弱碱性和强还原性,可与
)常温下呈液态,具有弱碱性和强还原性,可与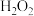 等氧化剂用作火箭燃料。
等氧化剂用作火箭燃料。(1)肼溶于水形成二元弱碱,碱性比
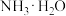 弱,在水中的电离与
弱,在水中的电离与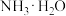 相似,写出肼在水中电离方程式
相似,写出肼在水中电离方程式(2)在标准压强和指定温度(298.15K)下,由元素最稳定的单质生成1mol化合物时的反应热称为该化合物的标准摩尔生成焓。已知
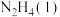 ,
,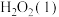 ,
, 的标准摩尔生成焓分别为50.63kJ/mol,-187.78kJ/mol,-241.82kJ/mol。,写出火箭燃料
的标准摩尔生成焓分别为50.63kJ/mol,-187.78kJ/mol,-241.82kJ/mol。,写出火箭燃料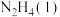 和氧化剂
和氧化剂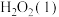 反应生成
反应生成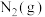 和
和 的热化学方程式:
的热化学方程式:(3)肼(
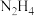 )-双氧水燃料电池由于绿色环保且具有较高的能量密度而被广泛使用,其工作原理如图所示。
)-双氧水燃料电池由于绿色环保且具有较高的能量密度而被广泛使用,其工作原理如图所示。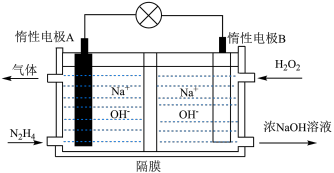
②设两极区溶液体积均为1 L,溶液的密度为1.2 g/cm
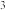 ,忽略溶液体积和密度的变化。当A极产生11.2 L(标准状况)气体时,B极区NaOH溶液的质量分数为40%,计算初始时NaOH溶液的浓度:
,忽略溶液体积和密度的变化。当A极产生11.2 L(标准状况)气体时,B极区NaOH溶液的质量分数为40%,计算初始时NaOH溶液的浓度:Ⅱ.我国的祝融号火星车已于2021年成功登陆火星,火星表面大气的主要成分是
 ,占总量的95.32%。科学家们设想通过还原反应将其转化为推进剂以实现星际旅行的愿望,二氧化碳加氢
,占总量的95.32%。科学家们设想通过还原反应将其转化为推进剂以实现星际旅行的愿望,二氧化碳加氢甲烷化(Sabatier反应)可表示为:
反应1:
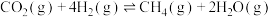
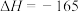 kJ/mol
kJ/mol反应2:

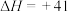 kJ/mol
kJ/mol将5 mol
 和20 mol
和20 mol  通入某恒压密闭容器中将
通入某恒压密闭容器中将 甲烷化,平衡时体系中各含碳元素物质的物质的量n(X)与温度T的关系如图所示。
甲烷化,平衡时体系中各含碳元素物质的物质的量n(X)与温度T的关系如图所示。
(5)当反应1和反应2均达到化学平衡状态时,若维持温度不变,向容器内通入惰性气体He,则反应2平衡
(6)800K时,若平衡时容器内总压为p,则反应2的压强平衡常数
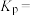
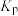 为以分压表示的平衡常数,分压=总压×物质的量分数)。
为以分压表示的平衡常数,分压=总压×物质的量分数)。
您最近一年使用:0次
2024-06-07更新
|
45次组卷
|
2卷引用:2024届新高考教学教研联盟高三下学期第一次联考化学试卷
3 . 673K时,向一恒容密闭容器中充入H2(g),并加入足量I2(s),发生如下反应。
①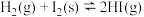
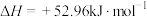
②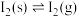
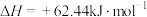
该温度下,反应②可以非常迅速地达到平衡,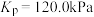 该容器内的总压p随反应时间t的变化如表所示。
该容器内的总压p随反应时间t的变化如表所示。
回答下列问题:
(1)反应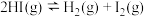
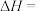
_______ 
(2)达到平衡后,若将体系降温至500K,体系中固体碘质量_______ (填“增大”、“减小”或“不变”),主要原因为_______ 。
(3)针对氢气与碘蒸气的反应提出了如下机理。
第一步: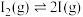 快速平衡
快速平衡
第二步: 慢步骤
慢步骤
据此分析,第一步为_______ (填“吸热”或“放热”)过程,主要原因为_______ 。总反应速率主要由_______ (填“第一步”或“第二步”)反应速率决定。
(4)研究表明,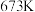 时,
时, 的反应速率
的反应速率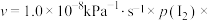
 ,
, 时,体系中的
时,体系中的
_______  ,
,
_______ 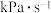 (计算结果均保留两位有效数字)。
(计算结果均保留两位有效数字)。
①
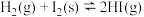
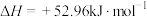
②
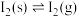
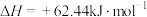
该温度下,反应②可以非常迅速地达到平衡,
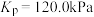 该容器内的总压p随反应时间t的变化如表所示。
该容器内的总压p随反应时间t的变化如表所示。t/s | 0 | 20000 | 40000 | 80000 | 90000 | ∞ |
p/kPa | 170.7 | 171.9 | 173.1 | 175.3 | 175.9 | 176.0 |
(1)反应
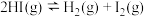
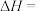

(2)达到平衡后,若将体系降温至500K,体系中固体碘质量
(3)针对氢气与碘蒸气的反应提出了如下机理。
第一步:
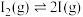 快速平衡
快速平衡第二步:
 慢步骤
慢步骤据此分析,第一步为
(4)研究表明,
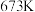 时,
时, 的反应速率
的反应速率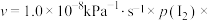
 ,
, 时,体系中的
时,体系中的
 ,
,
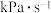 (计算结果均保留两位有效数字)。
(计算结果均保留两位有效数字)。
您最近一年使用:0次
4 . 以二氧化碳为碳源合成有机物,既能为有机合成提供廉价的原料,又有利于降低碳排放,实现“碳中和”。
回答下列问题:
(1) 催化加氢制备乙酸的过程中涉及如下反应:
催化加氢制备乙酸的过程中涉及如下反应:
Ⅰ.
 ;
;
Ⅱ.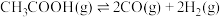
 ;
;
Ⅲ.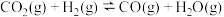
 。
。
则
______ ;已知反应Ⅲ中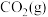 、
、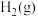 的总键能分别为
的总键能分别为
 、
、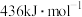 ,
, 键的键能为
键的键能为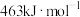 ,则
,则 中
中 键的键能为
键的键能为______  。
。
(2)已知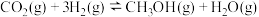
 。向某密闭容器中充入
。向某密闭容器中充入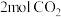 ,
,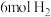 ,控制适当的温度、压强使二者发生反应生成
,控制适当的温度、压强使二者发生反应生成 ,测得
,测得 的平衡转化率如图所示:
的平衡转化率如图所示: 表示
表示______ (填“温度”或“压强”),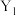
______ 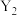 (填“>”或“<”)。
(填“>”或“<”)。
②若 点对应的压强为
点对应的压强为 ,反应从开始到平衡所用时间为
,反应从开始到平衡所用时间为 ,则
,则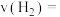
______  ;平衡常数
;平衡常数
______ 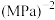 (保留两位有效数字)。
(保留两位有效数字)。
③该反应的速率常数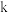 与反应温度
与反应温度 的关系为
的关系为 (其中
(其中 为活化能,
为活化能, 、
、 均为常数),则能使速率常数增大的措施是
均为常数),则能使速率常数增大的措施是______ (填选项字母)。
A.升高温度 B.压缩容器
C.增大 D.使用催化剂
D.使用催化剂
④上述容器中还存在两个副反应:
i.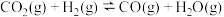
 ;
;
ii.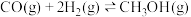

研究表明,在温度较低时, 的平衡转化率开始时随着温度的升高而减小,当温度高于
的平衡转化率开始时随着温度的升高而减小,当温度高于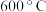 时,
时, 的平衡转化率却随着温度的升高而增大,其原因是
的平衡转化率却随着温度的升高而增大,其原因是______ 。
回答下列问题:
(1)
 催化加氢制备乙酸的过程中涉及如下反应:
催化加氢制备乙酸的过程中涉及如下反应:Ⅰ.

 ;
;Ⅱ.
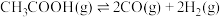
 ;
;Ⅲ.
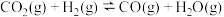
 。
。则

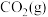 、
、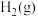 的总键能分别为
的总键能分别为
 、
、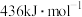 ,
, 键的键能为
键的键能为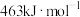 ,则
,则 中
中 键的键能为
键的键能为 。
。(2)已知
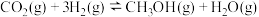
 。向某密闭容器中充入
。向某密闭容器中充入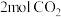 ,
,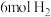 ,控制适当的温度、压强使二者发生反应生成
,控制适当的温度、压强使二者发生反应生成 ,测得
,测得 的平衡转化率如图所示:
的平衡转化率如图所示: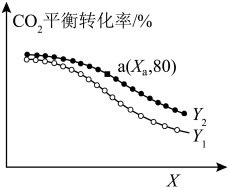
 表示
表示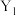
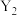 (填“>”或“<”)。
(填“>”或“<”)。②若
 点对应的压强为
点对应的压强为 ,反应从开始到平衡所用时间为
,反应从开始到平衡所用时间为 ,则
,则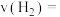
 ;平衡常数
;平衡常数
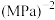 (保留两位有效数字)。
(保留两位有效数字)。③该反应的速率常数
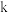 与反应温度
与反应温度 的关系为
的关系为 (其中
(其中 为活化能,
为活化能, 、
、 均为常数),则能使速率常数增大的措施是
均为常数),则能使速率常数增大的措施是A.升高温度 B.压缩容器
C.增大
 D.使用催化剂
D.使用催化剂④上述容器中还存在两个副反应:
i.
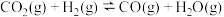
 ;
;ii.
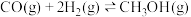

研究表明,在温度较低时,
 的平衡转化率开始时随着温度的升高而减小,当温度高于
的平衡转化率开始时随着温度的升高而减小,当温度高于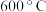 时,
时, 的平衡转化率却随着温度的升高而增大,其原因是
的平衡转化率却随着温度的升高而增大,其原因是
您最近一年使用:0次
2024高三下·全国·专题练习
5 . 氢能作为清洁能源是题佳碳中和能源载体,为应对气候变化全球掀起了氢能发展热潮,制备氢气有多种途径。
I.甲醇产生氢气的反应为: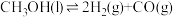

(1)已知相关物质的标准熵数值如表:
该反应能自发进行的最低温度为_______ K。(精确至 )
)
Ⅱ.乙酸制氢过程发生如下反应:
反应I: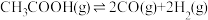
反应Ⅱ: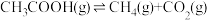
(2)已知反应: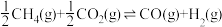
①由图1可得,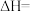
_______  。
。_______ 。
A.混合气体密度不变时,反应达到平衡 B.加入 ,可提高
,可提高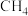 的转化率
的转化率
C.加入催化剂,可提高 的平衡产率 D.充入Ar,对
的平衡产率 D.充入Ar,对 的产率不产生影响
的产率不产生影响
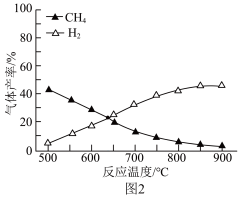
(3)在容积相同的密闭容器中,加入等量乙酸蒸气(只发生反应I和Ⅱ),在相同时间测得温度与气体产率的关系如图2所示。约650℃之前,氢气产率低于甲烷的可能原因是_______ 。(答出一点即可)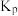 为
为_______ 。(用平衡分压代替平衡浓度计算)
Ⅲ.热分解也可制氢,其原理是: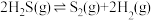 。
。
(5)不同温度和压强下,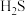 的平衡转化率变化如图。生成物
的平衡转化率变化如图。生成物 的状态为
的状态为_______ (填“气态”或“非气态”),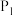
_______ 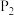 (填“>”或“<”)。
(填“>”或“<”)。
I.甲醇产生氢气的反应为:
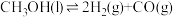

(1)已知相关物质的标准熵数值如表:
| 化学式 |  | 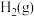 | 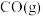 |
标准熵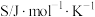 |  | 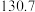 | 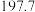 |
 )
)Ⅱ.乙酸制氢过程发生如下反应:
反应I:
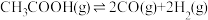
反应Ⅱ:
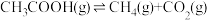
(2)已知反应:
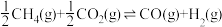
①由图1可得,
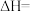
 。
。
A.混合气体密度不变时,反应达到平衡 B.加入
 ,可提高
,可提高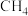 的转化率
的转化率C.加入催化剂,可提高
 的平衡产率 D.充入Ar,对
的平衡产率 D.充入Ar,对 的产率不产生影响
的产率不产生影响(3)在容积相同的密闭容器中,加入等量乙酸蒸气(只发生反应I和Ⅱ),在相同时间测得温度与气体产率的关系如图2所示。约650℃之前,氢气产率低于甲烷的可能原因是
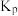 为
为Ⅲ.热分解也可制氢,其原理是:
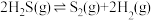 。
。(5)不同温度和压强下,
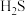 的平衡转化率变化如图。生成物
的平衡转化率变化如图。生成物 的状态为
的状态为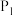
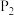 (填“>”或“<”)。
(填“>”或“<”)。
您最近一年使用:0次
6 . 回答下列问题:
(1)已知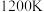 时在某恒容密闭容器中加入足量的
时在某恒容密闭容器中加入足量的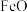 和
和 发生如下几个反应:
发生如下几个反应:

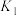
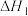

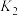

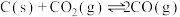
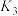

①写出基态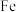 原子的价层电子排布图:
原子的价层电子排布图:_______ 。
②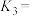
_______ (用含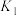 、
、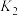 代数式表示)。
代数式表示)。
③若上述反应达到平衡后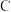 的转化率为
的转化率为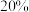 ,经检测体系中含有
,经检测体系中含有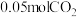 ,则得到
,则得到_______  金属铁。若再加入一定量的
金属铁。若再加入一定量的 ,再次达到平衡后,
,再次达到平衡后,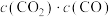 的值将
的值将_______ (填标号)。
A.增大 B.减小 C.不变 D.无法确定
(2)通过下列反应可实现 的再转化,合成
的再转化,合成 、
、 :
:
①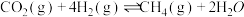

②
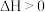
在密闭容器中通入一定量的 和
和 ,分别在压强为
,分别在压强为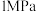 和
和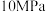 下进行反应。实验中对平衡体系的三种物质(
下进行反应。实验中对平衡体系的三种物质( 、
、 、
、 )进行分析,其中温度对
)进行分析,其中温度对 和
和 在三种物质中的体积分数影响如图所示。
在三种物质中的体积分数影响如图所示。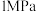 时
时 平衡体积分数随温度变化关系的曲线是
平衡体积分数随温度变化关系的曲线是_______ (填“a”“b”“o”或“d”中的一种)。简述理由:_______ 。
(3)有一个经验规则可以用于估算基元反应的活化能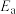 :
:
(BE:反应中断掉的化学键的键能之和)
①反应中没有化学键断裂的反应;②自由基与分子的放热反应;③反应中化学键不完全断裂(如协同反应)的反应;④反应中化学键完全断裂的反应。
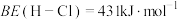
根据上述经验规则,估算氢气与氯气生成氯化氢的反应历程中第ⅱ步基元反应的活化能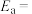
_______ ,并与实验值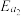 比较。
比较。
ⅰ.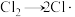
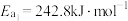
ⅱ.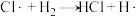
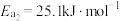
ⅲ.
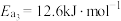
ⅳ.

 ;
; ;
;
上述反应历程中决定整个反应的速率是第_______ (填标号)步反应。
(1)已知
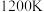 时在某恒容密闭容器中加入足量的
时在某恒容密闭容器中加入足量的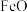 和
和 发生如下几个反应:
发生如下几个反应:
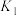
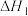

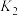

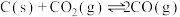
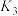

①写出基态
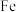 原子的价层电子排布图:
原子的价层电子排布图:②
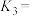
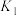 、
、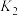 代数式表示)。
代数式表示)。③若上述反应达到平衡后
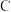 的转化率为
的转化率为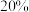 ,经检测体系中含有
,经检测体系中含有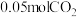 ,则得到
,则得到 金属铁。若再加入一定量的
金属铁。若再加入一定量的 ,再次达到平衡后,
,再次达到平衡后,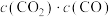 的值将
的值将A.增大 B.减小 C.不变 D.无法确定
(2)通过下列反应可实现
 的再转化,合成
的再转化,合成 、
、 :
:①
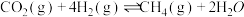

②

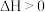
在密闭容器中通入一定量的
 和
和 ,分别在压强为
,分别在压强为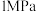 和
和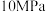 下进行反应。实验中对平衡体系的三种物质(
下进行反应。实验中对平衡体系的三种物质( 、
、 、
、 )进行分析,其中温度对
)进行分析,其中温度对 和
和 在三种物质中的体积分数影响如图所示。
在三种物质中的体积分数影响如图所示。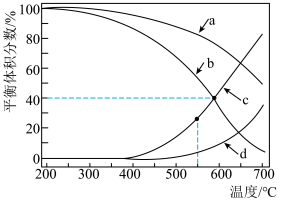
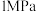 时
时 平衡体积分数随温度变化关系的曲线是
平衡体积分数随温度变化关系的曲线是(3)有一个经验规则可以用于估算基元反应的活化能
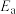 :
:反应类型 | ① | ② | ③ | ④ |
| 0 | 0.05 | 0.3 | 1 |
①反应中没有化学键断裂的反应;②自由基与分子的放热反应;③反应中化学键不完全断裂(如协同反应)的反应;④反应中化学键完全断裂的反应。
根据上述经验规则,估算氢气与氯气生成氯化氢的反应历程中第ⅱ步基元反应的活化能
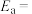
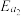 比较。
比较。ⅰ.
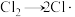
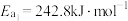
ⅱ.
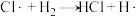
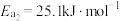
ⅲ.

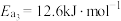
ⅳ.


 ;
; ;
;
上述反应历程中决定整个反应的速率是第
您最近一年使用:0次
解题方法
7 . 二甲醚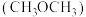 既是一种有机燃料,又是一种重要的有机化工原料。利用
既是一种有机燃料,又是一种重要的有机化工原料。利用 催化氢化制备二甲醚的反应原理如下:
催化氢化制备二甲醚的反应原理如下:
 .
.

 .
.

 .
.

回答下列问题:
(1)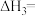
___________ 。
(2)向初始温度为T℃的某恒容绝热容器中投入2mol 只发生反应
只发生反应 ,平衡时
,平衡时 的转化率为
的转化率为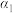 。
。
①下列叙述能说明反应 已经达到平衡的是
已经达到平衡的是___________ (填选项字母)。
a.混合气体密度不再发生变化
b.容器内压强不再发生变化
c. 的消耗速率等于
的消耗速率等于 的消耗速率
的消耗速率
d. 的体积分数不再发生变化
的体积分数不再发生变化
②相同条件下,若向该容器中加入 和
和 各1mol,平衡时
各1mol,平衡时 的转化率为
的转化率为 。则
。则
___________ 1(填“>”“=”或“<”)。
③在催化剂条件下反应 的反应过程如图甲所示,“*”表示吸附在催化剂上。
的反应过程如图甲所示,“*”表示吸附在催化剂上。
该催化过程的决速步骤为___________ (填“第一步”或“第二步”),判断的理由是___________ 。
(3)向压强恒定为p kPa的某密闭容器中按 投入
投入 和
和 ,发生
,发生 催化氢化制备二甲醚系列反应
催化氢化制备二甲醚系列反应 和
和 。
。 平衡转化率
平衡转化率 随温度的变化情况如图乙。
随温度的变化情况如图乙。
①图中能表示 随温度变化的曲线是
随温度变化的曲线是___________ (填“ ”或“
”或“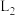 ”),原因为
”),原因为___________ 。
②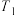 K时,
K时,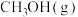 的平衡体积分数为10%。则平衡时
的平衡体积分数为10%。则平衡时 的体积分数为
的体积分数为___________ (保留3位有效数字);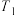 K时反应
K时反应 的压强平衡常数
的压强平衡常数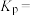
___________  (用含p的代数式表示,列出计算式即可)。
(用含p的代数式表示,列出计算式即可)。
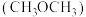 既是一种有机燃料,又是一种重要的有机化工原料。利用
既是一种有机燃料,又是一种重要的有机化工原料。利用 催化氢化制备二甲醚的反应原理如下:
催化氢化制备二甲醚的反应原理如下: .
.

 .
.

 .
.

回答下列问题:
(1)
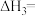
(2)向初始温度为T℃的某恒容绝热容器中投入2mol
 只发生反应
只发生反应 ,平衡时
,平衡时 的转化率为
的转化率为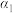 。
。①下列叙述能说明反应
 已经达到平衡的是
已经达到平衡的是a.混合气体密度不再发生变化
b.容器内压强不再发生变化
c.
 的消耗速率等于
的消耗速率等于 的消耗速率
的消耗速率d.
 的体积分数不再发生变化
的体积分数不再发生变化②相同条件下,若向该容器中加入
 和
和 各1mol,平衡时
各1mol,平衡时 的转化率为
的转化率为 。则
。则
③在催化剂条件下反应
 的反应过程如图甲所示,“*”表示吸附在催化剂上。
的反应过程如图甲所示,“*”表示吸附在催化剂上。
该催化过程的决速步骤为
(3)向压强恒定为p kPa的某密闭容器中按
 投入
投入 和
和 ,发生
,发生 催化氢化制备二甲醚系列反应
催化氢化制备二甲醚系列反应 和
和 。
。 平衡转化率
平衡转化率 随温度的变化情况如图乙。
随温度的变化情况如图乙。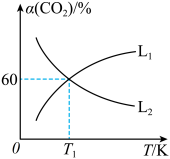
①图中能表示
 随温度变化的曲线是
随温度变化的曲线是 ”或“
”或“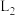 ”),原因为
”),原因为②
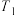 K时,
K时,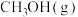 的平衡体积分数为10%。则平衡时
的平衡体积分数为10%。则平衡时 的体积分数为
的体积分数为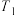 K时反应
K时反应 的压强平衡常数
的压强平衡常数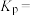
 (用含p的代数式表示,列出计算式即可)。
(用含p的代数式表示,列出计算式即可)。
您最近一年使用:0次
2024-05-19更新
|
243次组卷
|
4卷引用:2024届陕西省安康市汉滨区高三下学期联考模拟预测(四)理综试题-高中化学
2024届陕西省安康市汉滨区高三下学期联考模拟预测(四)理综试题-高中化学(已下线)押题卷01(14+4题型)-2024高考化学考点必杀300题(新高考通用)河南省青桐鸣联考2023-2024学年高二下学期3月月考化学试题(已下线)压轴题13?化学反应原理综合题(5大题型+方法总结+压轴题速练)-2024年高考化学压轴题专项训练(新高考通用)
8 . 含硫化合物在能源、材料及环境等工业领域均有广泛的应用。
Ⅰ.工业废气 分解可制取
分解可制取 。
。
(1)已知热化学方程式:
ⅰ.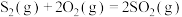

ⅱ.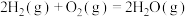

ⅲ.

 热分解反应
热分解反应 的
的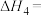
___________ (用含 、
、 、
、 的式子表示)。
的式子表示)。
(2)一定温度下,2mol 在体积为VL的恒容密闭容器中发生上述分解反应,ts时
在体积为VL的恒容密闭容器中发生上述分解反应,ts时 的产率为40%,则0~ts内
的产率为40%,则0~ts内 的平均分解速率为
的平均分解速率为___________ 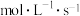 ;
; 充分分解达到平衡时,容器中
充分分解达到平衡时,容器中 和
和 的分压相等,则该温度下的平衡常数
的分压相等,则该温度下的平衡常数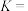
___________ 。
Ⅱ.KSCN可用于镀铬工业。常温下,用KSCN配制电镀液,溶液中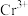 与
与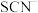 发生第一、二步络合的反应如下:
发生第一、二步络合的反应如下:
ⅳ.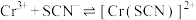
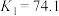
ⅴ.

(3)根据以上络合反应,下列说法正确的有___________(填字母)。
(4)常温下,某研究小组配制了起始浓度 、
、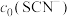 不同的系列溶液,测得平衡时
不同的系列溶液,测得平衡时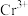 、
、 、
、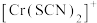 的浓度
的浓度 (含铬微粒)随
(含铬微粒)随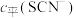 的变化曲线如图所示,平衡后其他含铬微粒
的变化曲线如图所示,平衡后其他含铬微粒 (3≤x≤6,图中未画出)总浓度为amol/L。
(3≤x≤6,图中未画出)总浓度为amol/L。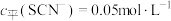 时,图中含铬微粒按浓度由大到小的顺序为
时,图中含铬微粒按浓度由大到小的顺序为___________ ;A点时,溶液中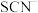 的平衡浓度为
的平衡浓度为___________ (列出计算式即可)。
②在某电镀工艺中,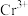 的浓度需要在
的浓度需要在 以上,结合计算判断C点所对应的溶液能否用于该电镀工艺
以上,结合计算判断C点所对应的溶液能否用于该电镀工艺________ (写出计算过程)。
Ⅰ.工业废气
 分解可制取
分解可制取 。
。(1)已知热化学方程式:
ⅰ.
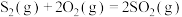

ⅱ.
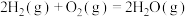

ⅲ.


 热分解反应
热分解反应 的
的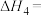
 、
、 、
、 的式子表示)。
的式子表示)。(2)一定温度下,2mol
 在体积为VL的恒容密闭容器中发生上述分解反应,ts时
在体积为VL的恒容密闭容器中发生上述分解反应,ts时 的产率为40%,则0~ts内
的产率为40%,则0~ts内 的平均分解速率为
的平均分解速率为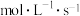 ;
; 充分分解达到平衡时,容器中
充分分解达到平衡时,容器中 和
和 的分压相等,则该温度下的平衡常数
的分压相等,则该温度下的平衡常数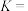
Ⅱ.KSCN可用于镀铬工业。常温下,用KSCN配制电镀液,溶液中
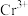 与
与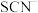 发生第一、二步络合的反应如下:
发生第一、二步络合的反应如下:ⅳ.
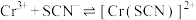
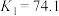
ⅴ.


(3)根据以上络合反应,下列说法正确的有___________(填字母)。
| A.加水稀释后,溶液中离子的总数减少 |
B.加入少量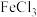 固体,溶液中含铬微粒总数不变 固体,溶液中含铬微粒总数不变 |
C.反应 的平衡常数 的平衡常数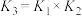 |
D.溶液中 减小, 减小,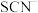 与 与 浓度的比值减小 浓度的比值减小 |
(4)常温下,某研究小组配制了起始浓度
 、
、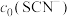 不同的系列溶液,测得平衡时
不同的系列溶液,测得平衡时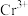 、
、 、
、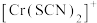 的浓度
的浓度 (含铬微粒)随
(含铬微粒)随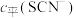 的变化曲线如图所示,平衡后其他含铬微粒
的变化曲线如图所示,平衡后其他含铬微粒 (3≤x≤6,图中未画出)总浓度为amol/L。
(3≤x≤6,图中未画出)总浓度为amol/L。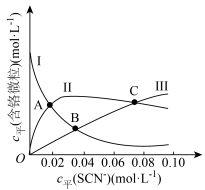
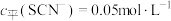 时,图中含铬微粒按浓度由大到小的顺序为
时,图中含铬微粒按浓度由大到小的顺序为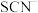 的平衡浓度为
的平衡浓度为②在某电镀工艺中,
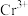 的浓度需要在
的浓度需要在 以上,结合计算判断C点所对应的溶液能否用于该电镀工艺
以上,结合计算判断C点所对应的溶液能否用于该电镀工艺
您最近一年使用:0次
名校
9 . 某温度下,利用CO2生产甲醇主要涉及以下两个反应。
反应I._______
反应Ⅱ.CO2(g)+H2(g)⇌CO(g)+H2O(g) ΔH2=+41.2kJ·mol-1
回答下列问题:
(1)已知CO(g)+2H2(g) CH3OH(g) ΔH3=–90.6kJ·mol-1。反应I的转化原理如图所示。该反应的热化学方程式为
CH3OH(g) ΔH3=–90.6kJ·mol-1。反应I的转化原理如图所示。该反应的热化学方程式为_______ ,在_______ 条件下反应I可自发发生。_______ 移动(填“正向”、“逆向”或“不”)。
(3)将1molCO2(g)和3molH2(g)充入密闭容器发生反应I和Ⅱ,并达到平衡状态。相同温度下,在不同压强下测得CO2的平衡转化率、CH3OH(g)的选择性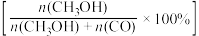 和CO的选择性
和CO的选择性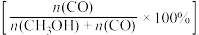 随压强变化曲线如图所示。图中表示CO2的平衡转化率的曲线是
随压强变化曲线如图所示。图中表示CO2的平衡转化率的曲线是_______ (填“m”、“n”或“p”),A点时H2的转化率为_______ ,CH3OH的平衡分压为_______ MPa(保留两位有效数字)。 ,式中x(CO)、x(H2O)、x(CO2)、x(H2)分别表示各组分的物质的量分数。Kp为平衡常数,k为反应的速率常数,温度升高时k值增大。在气体组成和催化剂一定的情况下,反应速率随温度变化的曲线如图所示。温度升高时,CO催化变换反应的Kp
,式中x(CO)、x(H2O)、x(CO2)、x(H2)分别表示各组分的物质的量分数。Kp为平衡常数,k为反应的速率常数,温度升高时k值增大。在气体组成和催化剂一定的情况下,反应速率随温度变化的曲线如图所示。温度升高时,CO催化变换反应的Kp_______ (填“增大”、“减小”或“不变”)。根据速率方程分析,T>Tm时v逐渐减小的原因是_______ 。
反应I._______
反应Ⅱ.CO2(g)+H2(g)⇌CO(g)+H2O(g) ΔH2=+41.2kJ·mol-1
回答下列问题:
(1)已知CO(g)+2H2(g)
 CH3OH(g) ΔH3=–90.6kJ·mol-1。反应I的转化原理如图所示。该反应的热化学方程式为
CH3OH(g) ΔH3=–90.6kJ·mol-1。反应I的转化原理如图所示。该反应的热化学方程式为
(3)将1molCO2(g)和3molH2(g)充入密闭容器发生反应I和Ⅱ,并达到平衡状态。相同温度下,在不同压强下测得CO2的平衡转化率、CH3OH(g)的选择性
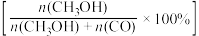 和CO的选择性
和CO的选择性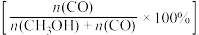 随压强变化曲线如图所示。图中表示CO2的平衡转化率的曲线是
随压强变化曲线如图所示。图中表示CO2的平衡转化率的曲线是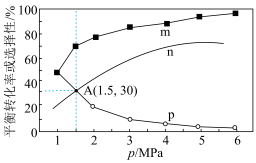
 ,式中x(CO)、x(H2O)、x(CO2)、x(H2)分别表示各组分的物质的量分数。Kp为平衡常数,k为反应的速率常数,温度升高时k值增大。在气体组成和催化剂一定的情况下,反应速率随温度变化的曲线如图所示。温度升高时,CO催化变换反应的Kp
,式中x(CO)、x(H2O)、x(CO2)、x(H2)分别表示各组分的物质的量分数。Kp为平衡常数,k为反应的速率常数,温度升高时k值增大。在气体组成和催化剂一定的情况下,反应速率随温度变化的曲线如图所示。温度升高时,CO催化变换反应的Kp
您最近一年使用:0次
10 . 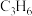 (丙烯)是重要基本有机原料。
(丙烯)是重要基本有机原料。 (丙烷)催化脱氢制
(丙烷)催化脱氢制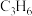 过程如下:
过程如下:
Ⅰ.主反应: ;
;
Ⅱ.副反应: 。
。
回答下列问题:
(1)已知断裂 下列化学键所需能量如下表:
下列化学键所需能量如下表:
上述主反应的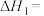
___________  。
。
(2)一定条件下, 催化脱氢制
催化脱氢制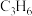 发生上述Ⅰ、Ⅱ反应,
发生上述Ⅰ、Ⅱ反应, 和
和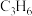 的平衡体积分数与温度、压强的关系如图1所示(图中压强分别为
的平衡体积分数与温度、压强的关系如图1所示(图中压强分别为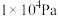 和
和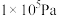 )。
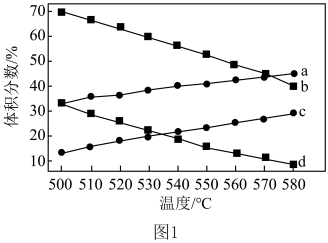
)。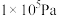 时,图中表示
时,图中表示 和
和 积分数变化的曲线分别为
积分数变化的曲线分别为___________ 、___________ 。
②提高 催化脱氢制
催化脱氢制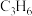 的反应平衡转化率的方法是
的反应平衡转化率的方法是___________ (任写一种)。
(3)在压力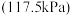 恒定,以
恒定,以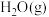 作为稀释气,不同水烃比
作为稀释气,不同水烃比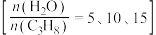 时,
时, 催化脱氢制备
催化脱氢制备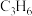 反应(上述反应Ⅰ、Ⅱ)平衡转化率随温度的变化曲线如图2所示。
反应(上述反应Ⅰ、Ⅱ)平衡转化率随温度的变化曲线如图2所示。 平衡转化率相同时,水烃比越高,对应的反应温度越
平衡转化率相同时,水烃比越高,对应的反应温度越___________ (填“高”或“低”)。
②相同温度下,水烃比远大于15:1时,丙烷的消耗速率明显下降,可能的原因是:
ⅰ.丙烷的浓度过低;
ⅱ.___________ 。
③M点对应条件下,若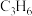 的选择性为
的选择性为 ,则反应Ⅰ的分压平衡常数
,则反应Ⅰ的分压平衡常数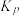 为
为___________ kPa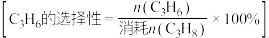 。
。
(4)利用 的弱氧化性,科研人员开发了
的弱氧化性,科研人员开发了 氧化脱氢制
氧化脱氢制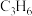 的新工艺。该工艺可采用铬的氧化物为催化剂,其反应机理如图3所示。
的新工艺。该工艺可采用铬的氧化物为催化剂,其反应机理如图3所示。___________ 。
②该工艺可以有效消除催化剂表面的积炭,维持催化剂活性,原因是___________ 。
(5)研究表明, 氧化脱氢制
氧化脱氢制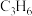 的催化剂中含有多种元素,如Cr、Zr、Mg、Fe、Ni等。由Mg、Fe、Ni组成的
的催化剂中含有多种元素,如Cr、Zr、Mg、Fe、Ni等。由Mg、Fe、Ni组成的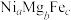 的立方晶胞结构如图4所示。已知晶胞的边长为
的立方晶胞结构如图4所示。已知晶胞的边长为 ,设
,设 为阿伏加德罗常数的值。该晶体密度是
为阿伏加德罗常数的值。该晶体密度是___________ 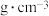 (用含
(用含 、
、 的代数式表示)。
的代数式表示)。
 (丙烯)是重要基本有机原料。
(丙烯)是重要基本有机原料。 (丙烷)催化脱氢制
(丙烷)催化脱氢制 过程如下:
过程如下:Ⅰ.主反应:
 ;
;Ⅱ.副反应:
 。
。回答下列问题:
(1)已知断裂
 下列化学键所需能量如下表:
下列化学键所需能量如下表:| 化学键 |  |  |  |
能量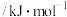 | 436.0 | 413.4 | 344.7 |
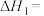
 。
。(2)一定条件下,
 催化脱氢制
催化脱氢制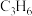 发生上述Ⅰ、Ⅱ反应,
发生上述Ⅰ、Ⅱ反应, 和
和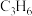 的平衡体积分数与温度、压强的关系如图1所示(图中压强分别为
的平衡体积分数与温度、压强的关系如图1所示(图中压强分别为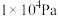 和
和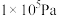 )。
)。
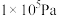 时,图中表示
时,图中表示 和
和 积分数变化的曲线分别为
积分数变化的曲线分别为②提高
 催化脱氢制
催化脱氢制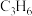 的反应平衡转化率的方法是
的反应平衡转化率的方法是(3)在压力
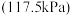 恒定,以
恒定,以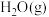 作为稀释气,不同水烃比
作为稀释气,不同水烃比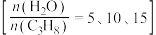 时,
时, 催化脱氢制备
催化脱氢制备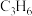 反应(上述反应Ⅰ、Ⅱ)平衡转化率随温度的变化曲线如图2所示。
反应(上述反应Ⅰ、Ⅱ)平衡转化率随温度的变化曲线如图2所示。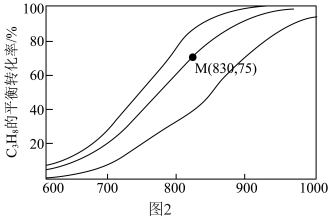
 平衡转化率相同时,水烃比越高,对应的反应温度越
平衡转化率相同时,水烃比越高,对应的反应温度越②相同温度下,水烃比远大于15:1时,丙烷的消耗速率明显下降,可能的原因是:
ⅰ.丙烷的浓度过低;
ⅱ.
③M点对应条件下,若
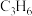 的选择性为
的选择性为 ,则反应Ⅰ的分压平衡常数
,则反应Ⅰ的分压平衡常数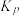 为
为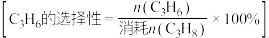 。
。(4)利用
 的弱氧化性,科研人员开发了
的弱氧化性,科研人员开发了 氧化脱氢制
氧化脱氢制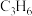 的新工艺。该工艺可采用铬的氧化物为催化剂,其反应机理如图3所示。
的新工艺。该工艺可采用铬的氧化物为催化剂,其反应机理如图3所示。
②该工艺可以有效消除催化剂表面的积炭,维持催化剂活性,原因是
(5)研究表明,
 氧化脱氢制
氧化脱氢制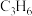 的催化剂中含有多种元素,如Cr、Zr、Mg、Fe、Ni等。由Mg、Fe、Ni组成的
的催化剂中含有多种元素,如Cr、Zr、Mg、Fe、Ni等。由Mg、Fe、Ni组成的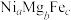 的立方晶胞结构如图4所示。已知晶胞的边长为
的立方晶胞结构如图4所示。已知晶胞的边长为 ,设
,设 为阿伏加德罗常数的值。该晶体密度是
为阿伏加德罗常数的值。该晶体密度是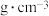 (用含
(用含 、
、 的代数式表示)。
的代数式表示)。
您最近一年使用:0次



