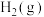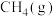名校
1 . 由 与
与 制备甲醇是当今研究的热点之一,也是我国科学家2021年发布的由
制备甲醇是当今研究的热点之一,也是我国科学家2021年发布的由 人工合成淀粉(节选途径见下图)中的重要反应之一,
人工合成淀粉(节选途径见下图)中的重要反应之一,
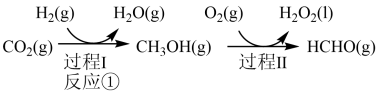
已知:
反应②:
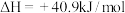
反应③:

(1)反应①的热化学方程式为___________ ;若过程Ⅱ中O得到4mol电子,则理论上可生成___________ molHCHO。
(2)反应①在有、无催化剂条件下的反应历程如下图所示,其中吸附在催化剂表面上的物种用*标注,TS为过渡态。
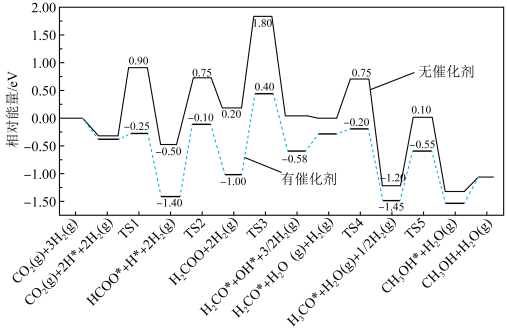
该反应历程中决速步骤的化学方程式为,催化剂使该步骤的活化能降低___________ eV。
(3)某研究小组采用上述催化剂,向密闭容器中通入3mol 和1mol
和1mol ,只发生反应①和反应②,在不同条件下达到平衡,在T=300℃下甲醇的体积分数
,只发生反应①和反应②,在不同条件下达到平衡,在T=300℃下甲醇的体积分数 (CHOH)随压强p的变化、在p-600kPa下
(CHOH)随压强p的变化、在p-600kPa下 (CHOH)随温度T的变化,如下图所示。
(CHOH)随温度T的变化,如下图所示。
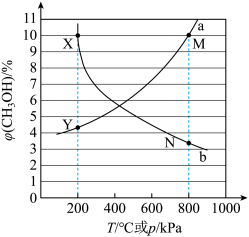
①X点对应的温度和压强为___________ ℃,___________ kPa。
②M点 的分压为
的分压为___________ kPa,此时容器中 为
为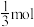 ,CO为
,CO为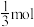 ,反应的压强平衡常数
,反应的压强平衡常数
___________ 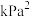 (压强平衡常数K是以分压代替浓度表示的平衡常数,分压=总压×物质的量分数)。
(压强平衡常数K是以分压代替浓度表示的平衡常数,分压=总压×物质的量分数)。
 与
与 制备甲醇是当今研究的热点之一,也是我国科学家2021年发布的由
制备甲醇是当今研究的热点之一,也是我国科学家2021年发布的由 人工合成淀粉(节选途径见下图)中的重要反应之一,
人工合成淀粉(节选途径见下图)中的重要反应之一,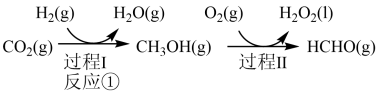
已知:
反应②:

反应③:


(1)反应①的热化学方程式为
(2)反应①在有、无催化剂条件下的反应历程如下图所示,其中吸附在催化剂表面上的物种用*标注,TS为过渡态。

该反应历程中决速步骤的化学方程式为,催化剂使该步骤的活化能降低
(3)某研究小组采用上述催化剂,向密闭容器中通入3mol
 和1mol
和1mol ,只发生反应①和反应②,在不同条件下达到平衡,在T=300℃下甲醇的体积分数
,只发生反应①和反应②,在不同条件下达到平衡,在T=300℃下甲醇的体积分数 (CHOH)随压强p的变化、在p-600kPa下
(CHOH)随压强p的变化、在p-600kPa下 (CHOH)随温度T的变化,如下图所示。
(CHOH)随温度T的变化,如下图所示。
①X点对应的温度和压强为
②M点
 的分压为
的分压为 为
为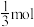 ,CO为
,CO为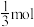 ,反应的压强平衡常数
,反应的压强平衡常数
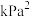 (压强平衡常数K是以分压代替浓度表示的平衡常数,分压=总压×物质的量分数)。
(压强平衡常数K是以分压代替浓度表示的平衡常数,分压=总压×物质的量分数)。
您最近一年使用:0次
解答题-原理综合题
|
适中(0.65)
|
解题方法
2 . 用学过的化学知识解决以下问题。
(1)一定条件下,在水溶液中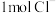 、
、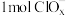 (
( 、2、3等)的相对能量(kJ)大小如图所示:
、2、3等)的相对能量(kJ)大小如图所示:
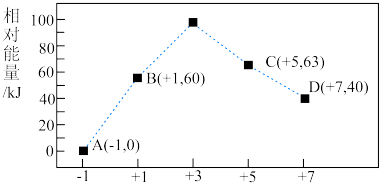
①C和D比较不稳定的离子是___________ (填离子符号)。
②在水溶液中发生反应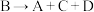 ,C和D的物质的量之比为2:1,写出该反应的热化学方程式:
,C和D的物质的量之比为2:1,写出该反应的热化学方程式:___________ (用离子符号表示)。
(2)已知:①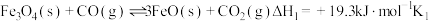
②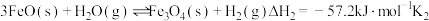
③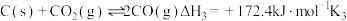
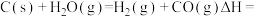
___________  ,
,
___________ 。
(3)已知断裂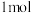 化学键所需的能量如下:
化学键所需的能量如下:
①COS的分子结构与 相似,COS的电子式为
相似,COS的电子式为___________ 。
②已知CO分子中有三对共用电子对, 还原COS发生的反应为
还原COS发生的反应为 ,该反应的
,该反应的
___________  。
。
(4)在标准状态下,由最稳定的单质生成单位物质的量的某纯物质的焓变称为该物质的标准摩尔生成焓( )。(已知:单质的标准生成焓有
)。(已知:单质的标准生成焓有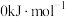 )。几种物质的标准摩尔生成焓如下:
)。几种物质的标准摩尔生成焓如下:
写出由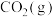 与
与 反应生成
反应生成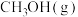 与的热化学方程式:
与的热化学方程式: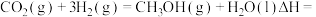
___________  。
。
(5)根据以下三个热化学方程式:
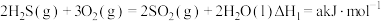
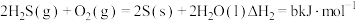
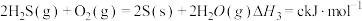
a、b、c最大的是___________ 。
(1)一定条件下,在水溶液中
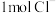 、
、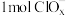 (
( 、2、3等)的相对能量(kJ)大小如图所示:
、2、3等)的相对能量(kJ)大小如图所示:
①C和D比较不稳定的离子是
②在水溶液中发生反应
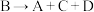 ,C和D的物质的量之比为2:1,写出该反应的热化学方程式:
,C和D的物质的量之比为2:1,写出该反应的热化学方程式:(2)已知:①
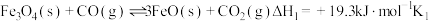
②
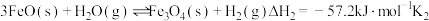
③
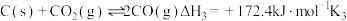
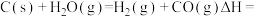
 ,
,
(3)已知断裂
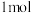 化学键所需的能量如下:
化学键所需的能量如下:| 化学键 | H-H | C=O | C=S | H-S |  |
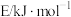 | 436 | 745 | 577 | 339 | 1072 |
 相似,COS的电子式为
相似,COS的电子式为②已知CO分子中有三对共用电子对,
 还原COS发生的反应为
还原COS发生的反应为 ,该反应的
,该反应的
 。
。(4)在标准状态下,由最稳定的单质生成单位物质的量的某纯物质的焓变称为该物质的标准摩尔生成焓(
 )。(已知:单质的标准生成焓有
)。(已知:单质的标准生成焓有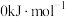 )。几种物质的标准摩尔生成焓如下:
)。几种物质的标准摩尔生成焓如下:| 物质 | 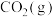 | 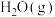 | 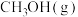 |
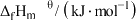 |  | 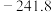 | 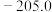 |
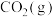 与
与 反应生成
反应生成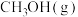 与的热化学方程式:
与的热化学方程式: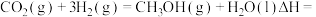
 。
。(5)根据以下三个热化学方程式:
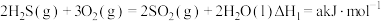
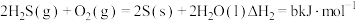
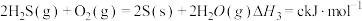
a、b、c最大的是
您最近一年使用:0次
名校
3 . 下列关于如图所示转化关系(X代表 ),说法
),说法正确 的是
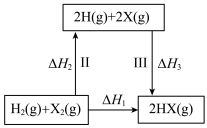
 ),说法
),说法
A.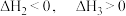 | B.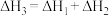 |
C.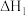 是该反应的活化能 是该反应的活化能 | D.若X分别是 ,则过程Ⅲ放出的热量依次减少 ,则过程Ⅲ放出的热量依次减少 |
您最近一年使用:0次
名校
解题方法
4 . 工业上可用 来生产甲醇等燃料,这一技术的应用对构建生态文明社会具有重要意义。
来生产甲醇等燃料,这一技术的应用对构建生态文明社会具有重要意义。
(1) 和
和 在催化剂作用下可发生以下两个反应:
在催化剂作用下可发生以下两个反应:
Ⅰ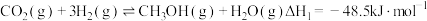
Ⅱ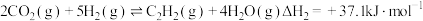
则反应 的
的
_______ 。
(2)在压强、 和
和 的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得
的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得 的平衡转化率和平衡时
的平衡转化率和平衡时 的选择性随温度的变化如图所示。
的选择性随温度的变化如图所示。

已知: 的选择性
的选择性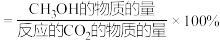
①曲线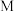 表示的是
表示的是_______ 。(填“ 的平衡转化率”或“
的平衡转化率”或“ 的选择性”)
的选择性”)
②温度高于280℃时,曲线N随温度升高而升高的原因是_______ 。
③要同时提高 的平衡转化率和
的平衡转化率和 的选择性,应选择的反应条件为
的选择性,应选择的反应条件为_______ (填标号)。
A.低温、低压 B.低温、高压 C.高温、高压 D.高温、低压
(3)在一定条件下,向体积为 的恒容密闭容器中,充入
的恒容密闭容器中,充入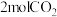 和
和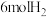 ,只反应Ⅰ、Ⅱ,tmin后达到平衡,转化率及和选择性如上图所示。
,只反应Ⅰ、Ⅱ,tmin后达到平衡,转化率及和选择性如上图所示。
①Q点时,生成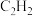 物质的量为
物质的量为_______ mol。
②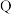 点时,从反应开始到平衡,
点时,从反应开始到平衡, 的反应速率
的反应速率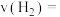
_______ mol·L-1·min-1。
③写出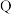 点时反应
点时反应 的化学平衡常数
的化学平衡常数
_______ (写计算式)。
 来生产甲醇等燃料,这一技术的应用对构建生态文明社会具有重要意义。
来生产甲醇等燃料,这一技术的应用对构建生态文明社会具有重要意义。(1)
 和
和 在催化剂作用下可发生以下两个反应:
在催化剂作用下可发生以下两个反应:Ⅰ
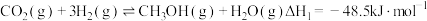
Ⅱ
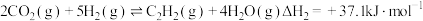
则反应
 的
的
(2)在压强、
 和
和 的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得
的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得 的平衡转化率和平衡时
的平衡转化率和平衡时 的选择性随温度的变化如图所示。
的选择性随温度的变化如图所示。
已知:
 的选择性
的选择性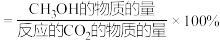
①曲线
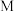 表示的是
表示的是 的平衡转化率”或“
的平衡转化率”或“ 的选择性”)
的选择性”)②温度高于280℃时,曲线N随温度升高而升高的原因是
③要同时提高
 的平衡转化率和
的平衡转化率和 的选择性,应选择的反应条件为
的选择性,应选择的反应条件为A.低温、低压 B.低温、高压 C.高温、高压 D.高温、低压
(3)在一定条件下,向体积为
 的恒容密闭容器中,充入
的恒容密闭容器中,充入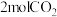 和
和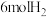 ,只反应Ⅰ、Ⅱ,tmin后达到平衡,转化率及和选择性如上图所示。
,只反应Ⅰ、Ⅱ,tmin后达到平衡,转化率及和选择性如上图所示。①Q点时,生成
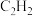 物质的量为
物质的量为②
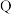 点时,从反应开始到平衡,
点时,从反应开始到平衡, 的反应速率
的反应速率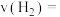
③写出
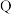 点时反应
点时反应 的化学平衡常数
的化学平衡常数
您最近一年使用:0次
名校
5 . Ⅰ.将氮氧化物进行无害化处理对治理大气污染、建设生态文明具有重要意义。
(1) 的氧化性强于
的氧化性强于 ,能更有效地氧化NO。已知:
,能更有效地氧化NO。已知:

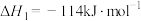

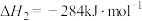
则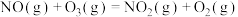
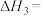
______  。
。
(2)在汽车排气管上安装催化转化器,能有效消除尾气对环境的污染:
①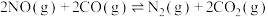
 ,反应能自发进行所需温度条件
,反应能自发进行所需温度条件_______ (填“高温”或“低温”)。
②已知:反应
 ,
,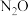 和CO在
和CO在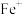 作用下发生的反应分两步进行:
作用下发生的反应分两步进行:
第一步;
第二步:
反应过程的能量变化如图所示:
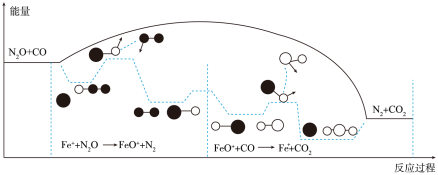
决定总反应速率的是________ (填“第一步”或“第二步)
(3)某温度下,在催化剂的作用下发生反应: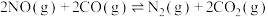 ,测得密闭容器中不同时间时CO和NO的浓度如表。
,测得密闭容器中不同时间时CO和NO的浓度如表。
①温度下,该反应的平衡常数为______ 。
②该恒温恒容条件下,下列能说明该反应达到化学平衡状态的是________ (填序号)。
a.体系的压强保持不变 b.
c.混合气体的密度不变 d.NO的转化率保持不变
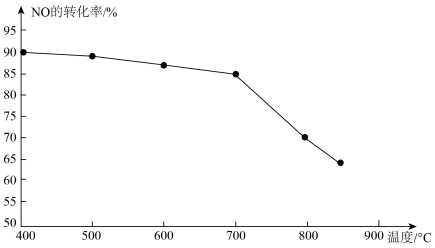
③恒容条件下,上述反应中若NO和CO的起始浓度一定,催化反应相同时间,测得NO的转化率随温度的变化如图所示,当温度超过700℃时,随温度的升高,NO的转化率下降明显,原因是________ 。
Ⅱ.由于亚硝酸钠和食盐性状相似,曾多次发生过将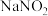 误当食盐食用的事件。欲测定某样品中
误当食盐食用的事件。欲测定某样品中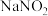 的含量,某同学设计如下实验:①称取样品ag,加水溶解,配制成100mL溶液。②取25.00mL溶液于锥形概中,用
的含量,某同学设计如下实验:①称取样品ag,加水溶解,配制成100mL溶液。②取25.00mL溶液于锥形概中,用 标准溶液(酸性)进行滴定,滴定结束后消耗
标准溶液(酸性)进行滴定,滴定结束后消耗 溶液VmL。
溶液VmL。
(4)在进行滴定操作时, 溶液盛装在
溶液盛装在_____ (填“酸式”或“碱式”)滴定管中。当滴入最后半滴 溶液,
溶液,_______ 时达到滴定终点。
(5)以下操作造成测定结果偏高的是________。
(6)滴定过程中发生反应的离子方程式是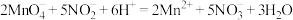 ,测得该样品中
,测得该样品中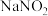 的质量分数为
的质量分数为______ 。(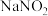 的摩尔质量
的摩尔质量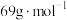 )
)
(1)
 的氧化性强于
的氧化性强于 ,能更有效地氧化NO。已知:
,能更有效地氧化NO。已知:
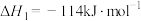

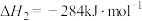
则
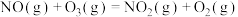
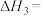
 。
。(2)在汽车排气管上安装催化转化器,能有效消除尾气对环境的污染:
①
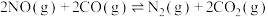
 ,反应能自发进行所需温度条件
,反应能自发进行所需温度条件②已知:反应

 ,
,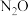 和CO在
和CO在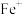 作用下发生的反应分两步进行:
作用下发生的反应分两步进行:第一步;

第二步:

反应过程的能量变化如图所示:

决定总反应速率的是
(3)某温度下,在催化剂的作用下发生反应:
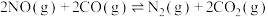 ,测得密闭容器中不同时间时CO和NO的浓度如表。
,测得密闭容器中不同时间时CO和NO的浓度如表。| 时间/s | 0 | 1 | 2 | 3 | 4 | 5 |
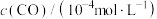 | 36.0 | 30.5 | 28.5 | 27.5 | 27.0 | 27.0 |
 | 10.0 | 4.5 | 2.5 | 1.5 | 1.0 | 1.0 |
②该恒温恒容条件下,下列能说明该反应达到化学平衡状态的是
a.体系的压强保持不变 b.

c.混合气体的密度不变 d.NO的转化率保持不变
③恒容条件下,上述反应中若NO和CO的起始浓度一定,催化反应相同时间,测得NO的转化率随温度的变化如图所示,当温度超过700℃时,随温度的升高,NO的转化率下降明显,原因是

Ⅱ.由于亚硝酸钠和食盐性状相似,曾多次发生过将
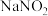 误当食盐食用的事件。欲测定某样品中
误当食盐食用的事件。欲测定某样品中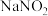 的含量,某同学设计如下实验:①称取样品ag,加水溶解,配制成100mL溶液。②取25.00mL溶液于锥形概中,用
的含量,某同学设计如下实验:①称取样品ag,加水溶解,配制成100mL溶液。②取25.00mL溶液于锥形概中,用 标准溶液(酸性)进行滴定,滴定结束后消耗
标准溶液(酸性)进行滴定,滴定结束后消耗 溶液VmL。
溶液VmL。(4)在进行滴定操作时,
 溶液盛装在
溶液盛装在 溶液,
溶液,(5)以下操作造成测定结果偏高的是________。
A.滴定管未用 标准溶液润洗 标准溶液润洗 |
| B.盛装标准溶液的滴定管,滴定前尖端有气泡,滴定后气泡消失 |
| C.盛装标准溶液的滴定管,滴定前仰视凹液面最低处,滴定后俯视读数 |
| D.若滴定过程中刚出现颜色变化就停止滴定 |
(6)滴定过程中发生反应的离子方程式是
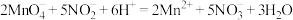 ,测得该样品中
,测得该样品中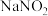 的质量分数为
的质量分数为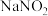 的摩尔质量
的摩尔质量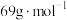 )
)
您最近一年使用:0次
6 . “绿水青山就是金山银山。”研究如何将NO2、NO、CO、SO2等大气污染物转化为能参与大气循环的物质,对建设美丽中国具有重要意义。
(1) 可在一定条件下转化为
可在一定条件下转化为 。已知:
。已知:

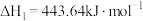
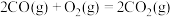
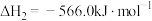
甲醇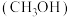 是一种重要的化工原料,也可以直接用作燃料。试写出
是一种重要的化工原料,也可以直接用作燃料。试写出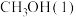 在氧气中完全燃烧生成
在氧气中完全燃烧生成 和
和 的热化学方程式:
的热化学方程式:___________ 。
(2)科研人员研发出一种由强碱溶液作电解质溶液的新型甲醇手机电池,充满电后手机可连续使用一个月,则放电时,甲醇在___________ (填“正”或“负”)极发生反应,其电极反应式为___________ 。
(3) 可在一定条件下转化为
可在一定条件下转化为 。向
。向 密闭容器中通入
密闭容器中通入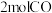 和
和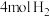 ,在适合的催化剂和
,在适合的催化剂和 温度下,发生反应
温度下,发生反应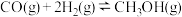
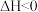 ,保持容器容积一直不变,
,保持容器容积一直不变, 时,反应达到平衡状态,
时,反应达到平衡状态,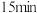 时改变温度为
时改变温度为 ,
, 时反应再次达到平衡状态,部分数据如下表:
时反应再次达到平衡状态,部分数据如下表:
① 时反应达到平衡,此时
时反应达到平衡,此时 的转化率为
的转化率为___________ ;化学平衡常数
___________
②
___________ (填“>”“<”或“=”) ;理由
;理由___________
③ 温度下,既能增大反应速率又能提高
温度下,既能增大反应速率又能提高 平䘖转化率的措施有
平䘖转化率的措施有___________ (填一种)。
④恒温恒容时,可以判断反应 达到平衡状态的有
达到平衡状态的有___________ (填标号)。
A.
B.容器内混合气体的压强不再改变
C. 和
和 的转化率的比值保持不变
的转化率的比值保持不变
D.容器内混合气体的平均相对分子质量不变
(1)
 可在一定条件下转化为
可在一定条件下转化为 。已知:
。已知:
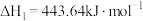
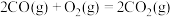
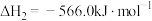
甲醇
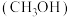 是一种重要的化工原料,也可以直接用作燃料。试写出
是一种重要的化工原料,也可以直接用作燃料。试写出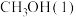 在氧气中完全燃烧生成
在氧气中完全燃烧生成 和
和 的热化学方程式:
的热化学方程式:(2)科研人员研发出一种由强碱溶液作电解质溶液的新型甲醇手机电池,充满电后手机可连续使用一个月,则放电时,甲醇在
(3)
 可在一定条件下转化为
可在一定条件下转化为 。向
。向 密闭容器中通入
密闭容器中通入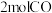 和
和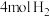 ,在适合的催化剂和
,在适合的催化剂和 温度下,发生反应
温度下,发生反应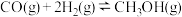
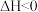 ,保持容器容积一直不变,
,保持容器容积一直不变, 时,反应达到平衡状态,
时,反应达到平衡状态,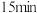 时改变温度为
时改变温度为 ,
, 时反应再次达到平衡状态,部分数据如下表:
时反应再次达到平衡状态,部分数据如下表:| 反应时间 |  | 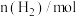 | 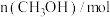 |
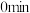 | 2 | 4 | 0 |
 | 1 | ||
 | 0.2 |
 时反应达到平衡,此时
时反应达到平衡,此时 的转化率为
的转化率为
②

 ;理由
;理由③
 温度下,既能增大反应速率又能提高
温度下,既能增大反应速率又能提高 平䘖转化率的措施有
平䘖转化率的措施有④恒温恒容时,可以判断反应
 达到平衡状态的有
达到平衡状态的有A.

B.容器内混合气体的压强不再改变
C.
 和
和 的转化率的比值保持不变
的转化率的比值保持不变 D.容器内混合气体的平均相对分子质量不变
您最近一年使用:0次
解题方法
7 . 工业上通过 与
与 共转化生成合成气,助力实现“碳达峰”。
共转化生成合成气,助力实现“碳达峰”。
其主反应ⅰ
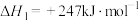 ;
;
副反应主要有:ⅱ
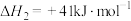 ;
;
ⅲ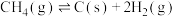
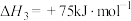 (积碳反应);
(积碳反应);
ⅳ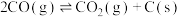
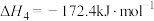 (积碳反应);
(积碳反应);
ⅴ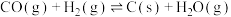
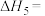 (积碳反应)
(积碳反应)
Ⅰ.若只考虑主反应
(1)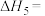
___________ 。
(2)该反应在一定条件下建立平衡后,下列说法正确的是___________。
(3)不同压强下,按照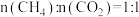 投料,实验测得
投料,实验测得 平衡转化率和
平衡转化率和 平衡产率随温度变化关系如下图所示,纵坐标可表示
平衡产率随温度变化关系如下图所示,纵坐标可表示 平衡转化率的为
平衡转化率的为___________ (填“甲”或“乙”),压强由大到小的顺序为___________ 。
(4)一定条件下,向真空密闭容器中通入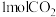 和
和 ,使总压为
,使总压为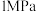 。反应达到平衡时,容器中气体压强为原来的
。反应达到平衡时,容器中气体压强为原来的 倍。已知容器中气体的分压
倍。已知容器中气体的分压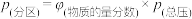 ,用气体的分压计算得到的平衡常数称为分压平衡常数
,用气体的分压计算得到的平衡常数称为分压平衡常数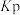 。则该反应的平衡常数
。则该反应的平衡常数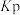 为
为___________  。
。
Ⅱ.若考虑副反应
在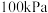 、CH4和
、CH4和 总物质的量为
总物质的量为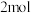 的条件下,温度及投料比[
的条件下,温度及投料比[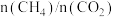 ]对或
]对或 平衡转化率的影响如图所示。
平衡转化率的影响如图所示。
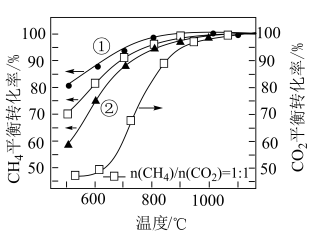
(5)根据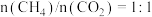 的曲线,推断表示
的曲线,推断表示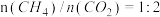 的
的 平衡转化率的曲线是
平衡转化率的曲线是___________ (填“①”或“②”)。
(6)在投料比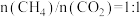 下
下 平衡转化率
平衡转化率 明显大于
明显大于 的原因为
的原因为___________ 。
(7)为了提高合成气的产量和消除积碳,可以往反应体系中加入适量的___________ 。
 与
与 共转化生成合成气,助力实现“碳达峰”。
共转化生成合成气,助力实现“碳达峰”。其主反应ⅰ

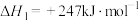 ;
;副反应主要有:ⅱ

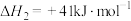 ;
;ⅲ
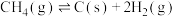
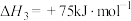 (积碳反应);
(积碳反应);ⅳ
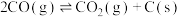
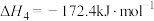 (积碳反应);
(积碳反应);ⅴ
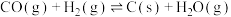
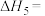 (积碳反应)
(积碳反应)Ⅰ.若只考虑主反应
(1)
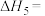
(2)该反应在一定条件下建立平衡后,下列说法正确的是___________。
| A.升高温度,反应平衡常数增大 |
| B.增大压强有利于反应平衡正向移动 |
C.增大 浓度,正、逆反应速率均增大 浓度,正、逆反应速率均增大 |
D.增大 浓度,可以提高 浓度,可以提高 的平衡转化率 的平衡转化率 |
(3)不同压强下,按照
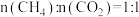 投料,实验测得
投料,实验测得 平衡转化率和
平衡转化率和 平衡产率随温度变化关系如下图所示,纵坐标可表示
平衡产率随温度变化关系如下图所示,纵坐标可表示 平衡转化率的为
平衡转化率的为
(4)一定条件下,向真空密闭容器中通入
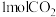 和
和 ,使总压为
,使总压为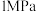 。反应达到平衡时,容器中气体压强为原来的
。反应达到平衡时,容器中气体压强为原来的 倍。已知容器中气体的分压
倍。已知容器中气体的分压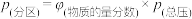 ,用气体的分压计算得到的平衡常数称为分压平衡常数
,用气体的分压计算得到的平衡常数称为分压平衡常数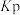 。则该反应的平衡常数
。则该反应的平衡常数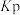 为
为 。
。Ⅱ.若考虑副反应
在
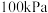 、CH4和
、CH4和 总物质的量为
总物质的量为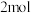 的条件下,温度及投料比[
的条件下,温度及投料比[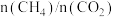 ]对或
]对或 平衡转化率的影响如图所示。
平衡转化率的影响如图所示。
(5)根据
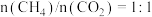 的曲线,推断表示
的曲线,推断表示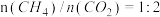 的
的 平衡转化率的曲线是
平衡转化率的曲线是(6)在投料比
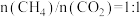 下
下 平衡转化率
平衡转化率 明显大于
明显大于 的原因为
的原因为(7)为了提高合成气的产量和消除积碳,可以往反应体系中加入适量的
您最近一年使用:0次
名校
8 . 铁及其化合物在生产、生活中有广泛的应用。
(1)中科院兰州化学物理研究所用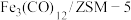 催化
催化 加氢合成低碳烯烃反应,反应过程如图,催化剂中添加助剂
加氢合成低碳烯烃反应,反应过程如图,催化剂中添加助剂 (也起催化作用)后可改变反应的选择性。
(也起催化作用)后可改变反应的选择性。
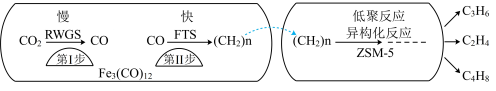
下列说法正确的是___________ 。
a.第1步所发生的反应为: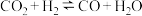
b.第1步反应的活化能低于第2步
c.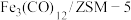 使
使 加氢合成低碳烯烃的
加氢合成低碳烯烃的 减小
减小
d.添加不同助剂后,各反应的平衡常数不变
(2)纳米铁是重要的储氢材料,可用反应 制得。在
制得。在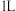 恒容密闭容器中加入足量铁粉和
恒容密闭容器中加入足量铁粉和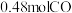 ,在
,在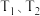 不同温度下进行反应,测得
不同温度下进行反应,测得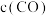 与温度、时间的关系如图所示。
与温度、时间的关系如图所示。
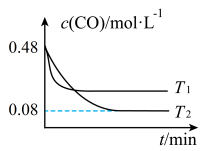
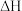
___________ 0(填“>”或“<”);
(3) 温度下,平衡时体系的压强为
温度下,平衡时体系的压强为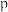 ,反应的标准平衡常数
,反应的标准平衡常数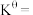
___________ (已知:标准平衡常数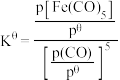 ,其中
,其中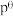 为标准压强
为标准压强 为各组分的平衡分压。)
为各组分的平衡分压。)
(4)高铁酸钾 被称为“绿色化学”净水剂,在酸性至弱碱性条件下不稳定。
被称为“绿色化学”净水剂,在酸性至弱碱性条件下不稳定。
①电解法可制得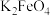 ,装置如图,阳极电极反应式为
,装置如图,阳极电极反应式为___________ 。

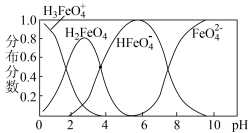
②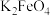 在水解过程中铁元素形成的微粒分布分数与
在水解过程中铁元素形成的微粒分布分数与 的关系如图所示,向
的关系如图所示,向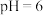 的溶液中加入
的溶液中加入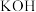 溶液,发生反应的离子方程式为
溶液,发生反应的离子方程式为___________ 。
(5)复合氧化物铁酸锰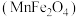 可用于热化学循环分解制氢气,原理如下:
可用于热化学循环分解制氢气,原理如下:
①
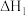
②

③
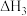
则: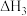 与
与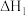 、
、 的关系为
的关系为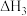
___________
(1)中科院兰州化学物理研究所用
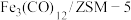 催化
催化 加氢合成低碳烯烃反应,反应过程如图,催化剂中添加助剂
加氢合成低碳烯烃反应,反应过程如图,催化剂中添加助剂 (也起催化作用)后可改变反应的选择性。
(也起催化作用)后可改变反应的选择性。
下列说法正确的是
a.第1步所发生的反应为:
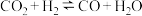
b.第1步反应的活化能低于第2步
c.
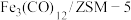 使
使 加氢合成低碳烯烃的
加氢合成低碳烯烃的 减小
减小d.添加不同助剂后,各反应的平衡常数不变
(2)纳米铁是重要的储氢材料,可用反应
 制得。在
制得。在 恒容密闭容器中加入足量铁粉和
恒容密闭容器中加入足量铁粉和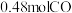 ,在
,在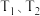 不同温度下进行反应,测得
不同温度下进行反应,测得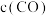 与温度、时间的关系如图所示。
与温度、时间的关系如图所示。
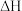
(3)
 温度下,平衡时体系的压强为
温度下,平衡时体系的压强为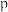 ,反应的标准平衡常数
,反应的标准平衡常数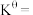
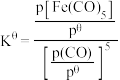 ,其中
,其中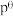 为标准压强
为标准压强 为各组分的平衡分压。)
为各组分的平衡分压。)(4)高铁酸钾
 被称为“绿色化学”净水剂,在酸性至弱碱性条件下不稳定。
被称为“绿色化学”净水剂,在酸性至弱碱性条件下不稳定。①电解法可制得
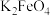 ,装置如图,阳极电极反应式为
,装置如图,阳极电极反应式为
②
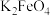 在水解过程中铁元素形成的微粒分布分数与
在水解过程中铁元素形成的微粒分布分数与 的关系如图所示,向
的关系如图所示,向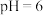 的溶液中加入
的溶液中加入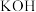 溶液,发生反应的离子方程式为
溶液,发生反应的离子方程式为
(5)复合氧化物铁酸锰
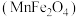 可用于热化学循环分解制氢气,原理如下:
可用于热化学循环分解制氢气,原理如下: ①

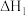
②


③

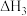
则:
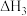 与
与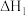 、
、 的关系为
的关系为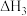
您最近一年使用:0次
解题方法
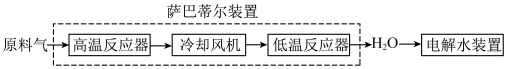
9 . 空间站内搭载萨巴蒂尔装置将 转化为
转化为 ,再通过电解水装置回收氧元素.其系统原理如图:
,再通过电解水装置回收氧元素.其系统原理如图:
萨巴蒂尔装置内发生反应为
反应Ⅰ: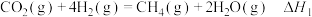
反应Ⅱ: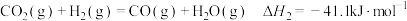
(1)已知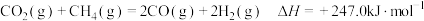 ,则
,则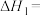
_________ .
(2)在“高温反应器”中以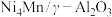 为催化剂,一段时间内不同温度
为催化剂,一段时间内不同温度 转化率如图.已知反应Ⅰ的速率方程:
转化率如图.已知反应Ⅰ的速率方程: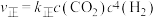 ,
, .
.
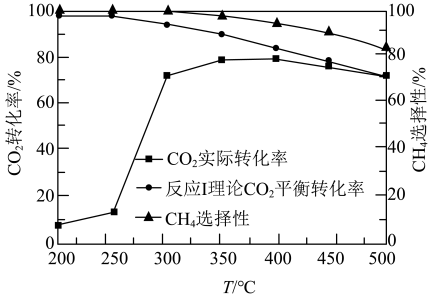
①250℃前, 实际转化率随温度变化不大的主要原因是
实际转化率随温度变化不大的主要原因是___________ .
②300℃时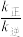
__________ 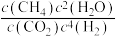 (填“>”“<”或“=”).
(填“>”“<”或“=”).
③达到平衡后,升高温度, 增大的倍数
增大的倍数_________ 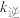 增大的倍数(填“>”“<”或“=”).
增大的倍数(填“>”“<”或“=”).
(3)在“低温反应器”中达平衡时,各组分物质的量分数随温度变化如图.已知原料气中含有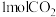 及
及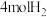 .
.
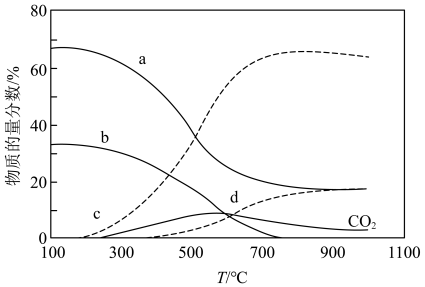
① 物质的量分数随温度变化的曲线为
物质的量分数随温度变化的曲线为____________ (填标号).
②300~500℃, 物质的量分数随温度升高而上升的原因是
物质的量分数随温度升高而上升的原因是___________ .
③某温度下达平衡时,总压为 ,剩余
,剩余 和
和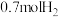 ,则反应Ⅰ的
,则反应Ⅰ的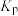 为
为___________ (列出计算式,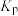 为以分压表示的平衡常数,分压=总压×物质的量分数);
为以分压表示的平衡常数,分压=总压×物质的量分数);
此时氧元素的回收率为____________ [回收率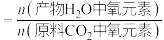 ].
].
(4)从平衡与速率角度分析萨巴蒂尔装置采用高温和低温反应器串联的优点是___________ .
 转化为
转化为 ,再通过电解水装置回收氧元素.其系统原理如图:
,再通过电解水装置回收氧元素.其系统原理如图:
萨巴蒂尔装置内发生反应为
反应Ⅰ:
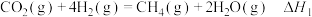
反应Ⅱ:
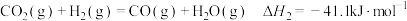
(1)已知
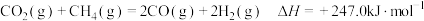 ,则
,则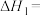
(2)在“高温反应器”中以
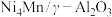 为催化剂,一段时间内不同温度
为催化剂,一段时间内不同温度 转化率如图.已知反应Ⅰ的速率方程:
转化率如图.已知反应Ⅰ的速率方程: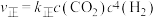 ,
, .
.
①250℃前,
 实际转化率随温度变化不大的主要原因是
实际转化率随温度变化不大的主要原因是②300℃时
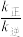
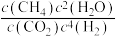 (填“>”“<”或“=”).
(填“>”“<”或“=”).③达到平衡后,升高温度,
 增大的倍数
增大的倍数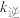 增大的倍数(填“>”“<”或“=”).
增大的倍数(填“>”“<”或“=”).(3)在“低温反应器”中达平衡时,各组分物质的量分数随温度变化如图.已知原料气中含有
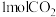 及
及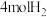 .
.
①
 物质的量分数随温度变化的曲线为
物质的量分数随温度变化的曲线为②300~500℃,
 物质的量分数随温度升高而上升的原因是
物质的量分数随温度升高而上升的原因是③某温度下达平衡时,总压为
 ,剩余
,剩余 和
和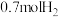 ,则反应Ⅰ的
,则反应Ⅰ的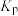 为
为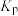 为以分压表示的平衡常数,分压=总压×物质的量分数);
为以分压表示的平衡常数,分压=总压×物质的量分数);此时氧元素的回收率为
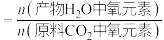 ].
].(4)从平衡与速率角度分析萨巴蒂尔装置采用高温和低温反应器串联的优点是
您最近一年使用:0次
解题方法
10 . 下列物质摩尔燃烧焓数据如下表:
下列表述正确的是
物质 | C(s,石墨) | C(s,金刚石) |
|
|
|
| -393.5 | -395.4 | -285.8 | -283.0 | -890.3 |
A.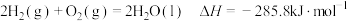 |
B.C(s,石墨)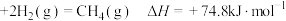 |
C.C(s,石墨)=C(s,金刚石)  |
D.C(s,石墨)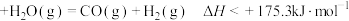 |
您最近一年使用:0次