
 ,下列说法正确的是
,下列说法正确的是| A.该反应在高温条件下可以自发进行 |
| B.向反应容器中加入催化剂,反应自发进行的临界温度会降低 |
| C.恒容条件下,通入He和Ar组成的混合气体,压强会增大,反应的速率会加快 |
D.恒容条件下,可通入过量的CO2气体来提高 转化率并增大生成物 转化率并增大生成物 的浓度 的浓度 |
2 . 阿根廷科学家MasVeronica等人利用Ni(Ⅱ)Al(Ⅲ)层状复合催化剂进行了乙醇水蒸气重整制氢的研究。其主要反应为
反应I.C2H5OH(g)+3H2O(g)=2CO2(g)+6H2(g) △H1=+173.3kJ/mol △S=+362.7J/(K•mol)
反应Ⅱ.C2H5OH(g)+H2O(g)=2CO(g)+4H2(g) △H2=+255.7kJ/mol
若在恒容密闭容器中发生上述反应。
(1)反应I在常温下
(2)对上述恒容密闭容器中的体系,下列说法正确的是
| A.增大H2O(g)的浓度,有利于提高C2H5OH平衡转化率 |
| B.一定温度下,增大C2H5OH的浓度可以提高C2H5OH的平衡转化率 |
| C.若气体的平均相对分子质量保持不变,说明反应体系已达平衡 |
| D.升高温度可以使反应I、Ⅱ的正反应速率加快,逆反应速率减小,平衡向正反应方向移动 |
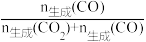 ×100%。
×100%。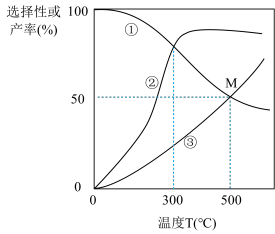
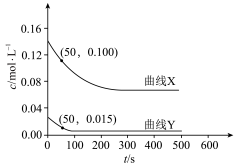
①图中曲线表示平衡时H2产率随温度变化的曲线是
②在密闭容器中,以投料比n(C2H5OH):n(H2O)=3:8通入混合气体,此时C2H5OH的转化率为66.67%,求交点M坐标为(500℃,50%)时反应Ⅱ的平衡常数Kp=
③一定能提高CO选择性的关键因素是
④请分析说明曲线②随温度变化的原因
(4)对乙醇水蒸气重整制氢反应I进行反应机理分析如下,“*”表示催化剂表面的活性中心,请补充完成基元反应iii。
i.C2H6O(g)+(*)→C2H6O*
ii.C2H6O*+(*)→CH4O*+CH2*
ⅲ.
iv.CH2*+2H2O(g)→CO2(g)+3H2(g)+(*)
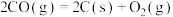 ,下列说法正确的是
,下列说法正确的是| A.低温下能自发进行 | B.高温下能自发进行 |
| C.任何温度下都不能自发进行 | D.任何温度下都能自发进行 |
反应I:
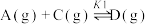

应Ⅱ:
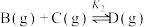

反应Ⅲ:

回答下列问题:
(1)①反应I、Ⅱ、Ⅲ的平衡常数
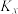 与温度T变化关系如图所示。据图判断,反应Ⅱ________自发进行;
与温度T变化关系如图所示。据图判断,反应Ⅱ________自发进行;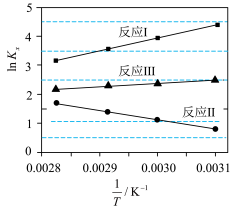
A.在任何温度下都不能 B.在任何温度下都能 C.在低温下能 D.在高温下能
②
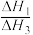 的数值范围是
的数值范围是A.<-1 B.-1~0 C.0~1 D.>1
(2)为研究上述反应体系的平衡关系,向VL某反应容器中加入a mol D(g),控制温度为353K,保持容积不变,测得D(g)的平衡转化率为b。该温度下已知反应Ⅲ的平衡常数
 ,则353K时平衡时,B(g)与D(g)物质的量浓度之比
,则353K时平衡时,B(g)与D(g)物质的量浓度之比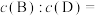
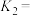
(3)为研究反应体系的动力学行为,向另一容器中加入一定量A(g)、B(g)、C(g)。控制温度为353K,A(g)、B(g)物质的量浓度c随反应时间t的变化如图2所示。代表B(g)变化曲线为
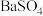 制得BaS,进而生产各种含钡化合物。温度对反应后组分的影响如图。
制得BaS,进而生产各种含钡化合物。温度对反应后组分的影响如图。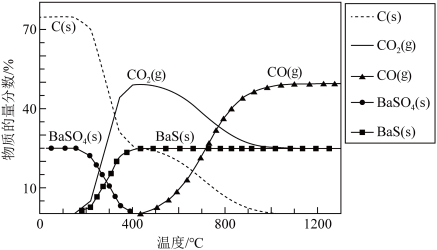
已知:碳热还原
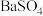 过程中可能发生下列反应。
过程中可能发生下列反应。ⅰ.


ⅱ.

 kJ/mol
kJ/molⅲ.

 kJ/mol
kJ/mol下列关于碳热还原
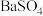 过程的说法正确的是
过程的说法正确的是A. kJ/mol kJ/mol |
| B.反应ⅱ在高温条件下自发进行 |
C.温度升高,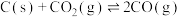 的平衡常数K减小 的平衡常数K减小 |
D.反应过程中,生成的 和CO的物质的量之和始终等于投入C的物质的量 和CO的物质的量之和始终等于投入C的物质的量 |
I.
(1)闪电时空气中的
 和
和 会发生反应:
会发生反应: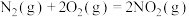
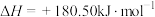 ,
,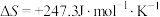 ,若不考虑温度对该反应焓变的影响,则下列说法中正确的是___________。
,若不考虑温度对该反应焓变的影响,则下列说法中正确的是___________。| A.在1000℃时,此反应能自发进行 |
| B.在1000℃时,此反应不能自发进行 |
| C.该反应能自发进行的最低温度约为730℃ |
D.该反应能自发进行的最高温度约为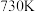 |
II.已知
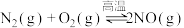 ,该反应在2404℃时的平衡常数
,该反应在2404℃时的平衡常数 。
。(2)该温度下,向
 密闭容器中充入
密闭容器中充入 与
与 各
各 ,平衡时
,平衡时 的转化率是
的转化率是(3)该温度下,某时刻测得容器内
 、
、 、
、 的浓度分别为
的浓度分别为 、
、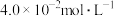 、
、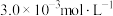 ,此时反应
,此时反应(4)将
 、
、 的混合气体充入恒温恒容密闭容器中,下图所示变化趋势正确的是
的混合气体充入恒温恒容密闭容器中,下图所示变化趋势正确的是A.
 B.
B. C.
C.
(5)向恒温恒容的密闭容器中充入等物质的量的
 与
与 ,达到平衡状态后再向其中充入一定量
,达到平衡状态后再向其中充入一定量 ,重新达到化学平衡状态。与原平衡状态相比,此时平衡混合气中
,重新达到化学平衡状态。与原平衡状态相比,此时平衡混合气中 的体积分数
的体积分数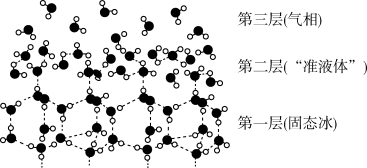
| A.第一层固态冰中,水分子间通过氢键形成空间网状结构,密度比液态水小 |
| B.第二层“准液体”水分子之间形成的氢键较第一层多 |
| C.当高于一定温度时,“准液体”中的水分子与下层冰连接的氢键断裂,产生“流动性水分子”,使冰面变滑 |
D.冰熔化成水的过程,是  |
A. 只能在高温下自发进行,则该反应的 只能在高温下自发进行,则该反应的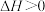 |
B.不同状态的镁中失去最外层一个电子所需能量: |
C.常温下 溶液中加入少量 溶液中加入少量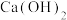 固体,溶液中 固体,溶液中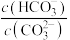 值减小 值减小 |
D.在一容积可变的密闭容器中反应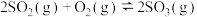 达平衡后,保持温度不变,缩小体积,平衡正向移动, 达平衡后,保持温度不变,缩小体积,平衡正向移动, 的值增大 的值增大 |
| A.该反应在任何条件下都能自发进行 |
B.反应的平衡常数可表示为K=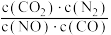 |
| C.使用高效的催化剂可以降低反应的焓变 |
D.其它条件不变,增大 的值,NO的转化率下降 的值,NO的转化率下降 |
| A.2N2(g)+O2(g)=2N2O(g) ΔH=+163 kJ·mol-1 |
| B.2Ag(s)+Cl2(g)= 2AgCl(s) ΔH=-127 kJ·mol-1 |
| C.2HgO(s)=2Hg(l)+O2(g) ΔH=+91 kJ·mol-1 |
| D.2H2O2(l)=O2(g)+2H2O(l) ΔH=-98 kJ·mol-1 |



