名校
1 . “氯碱工业”电解饱和食盐水为基础制取氯气等产品,氯气是实验室和工业上的常用气体。请回答:
(1)下列说法不正确的是___________。
(2)在一定温度下,氯气溶于水的过程及其平衡常数为:
 ;
;
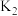 ,其中p为
,其中p为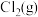 的平衡压强,
的平衡压强, 为Cl2在水溶液中的平衡浓度。
为Cl2在水溶液中的平衡浓度。
① 的焓变
的焓变
___________ 0。(填“>”、“=”或“<”)
②平衡常数K2的表达式为K2=___________ 。氯气在水中的溶解度(以物质的量浓度表示)为c,则c=___________ 。(用平衡压强p和上述平衡常数表示,忽略HClO的电离)
(3)工业上常采用“加碳氯化”的方法以高钛渣(主成分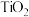 )为原料生产
)为原料生产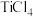 ,相应化学方程式为:
,相应化学方程式为:
Ⅰ.
 ,
,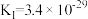
Ⅱ.
 ,
,
结合数据说明氯化过程中加碳的理由___________ 。
(4)在一定温度下,以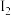 为催化剂,氯苯和
为催化剂,氯苯和 在
在 中发生平行反应,分别生成邻二氯苯和对二氯苯,两产物浓度之比与反应时间无关。反应物起始浓度均为0.5mol/L,反应30min测得氯苯15%转化为邻二氯苯,25%转化为对二氯苯。保持其他条件不变,若要提高产物中邻二氯苯的比例,可采用的最佳措施是
中发生平行反应,分别生成邻二氯苯和对二氯苯,两产物浓度之比与反应时间无关。反应物起始浓度均为0.5mol/L,反应30min测得氯苯15%转化为邻二氯苯,25%转化为对二氯苯。保持其他条件不变,若要提高产物中邻二氯苯的比例,可采用的最佳措施是___________ 。
(1)下列说法不正确的是___________。
| A.可采用碱石灰干燥氯气 |
| B.可通过排饱和食盐水法收集氯气 |
| C.常温下,可通过加压使氯气液化而储存于钢瓶中 |
| D.工业上,常用氢气和氯气反应生成的氯化氢溶于水制取盐酸 |

 ;
;
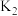 ,其中p为
,其中p为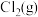 的平衡压强,
的平衡压强, 为Cl2在水溶液中的平衡浓度。
为Cl2在水溶液中的平衡浓度。①
 的焓变
的焓变
②平衡常数K2的表达式为K2=
(3)工业上常采用“加碳氯化”的方法以高钛渣(主成分
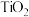 )为原料生产
)为原料生产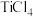 ,相应化学方程式为:
,相应化学方程式为:Ⅰ.

 ,
,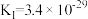
Ⅱ.

 ,
,
结合数据说明氯化过程中加碳的理由
(4)在一定温度下,以
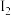 为催化剂,氯苯和
为催化剂,氯苯和 在
在 中发生平行反应,分别生成邻二氯苯和对二氯苯,两产物浓度之比与反应时间无关。反应物起始浓度均为0.5mol/L,反应30min测得氯苯15%转化为邻二氯苯,25%转化为对二氯苯。保持其他条件不变,若要提高产物中邻二氯苯的比例,可采用的最佳措施是
中发生平行反应,分别生成邻二氯苯和对二氯苯,两产物浓度之比与反应时间无关。反应物起始浓度均为0.5mol/L,反应30min测得氯苯15%转化为邻二氯苯,25%转化为对二氯苯。保持其他条件不变,若要提高产物中邻二氯苯的比例,可采用的最佳措施是
您最近一年使用:0次
名校
2 . 二甲醚(CH3OCH3)被称为“21世纪的清洁燃料”。以CO2、H2为原料制备二甲醚涉及的主要反应如下:
Ⅰ.
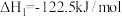
Ⅱ.
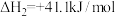
回答下列问题:
(1)反应 的
的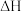 =
=___________ 。
(2)在压强、 和
和 的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得
的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得 平衡转化率和平衡时
平衡转化率和平衡时 的选择性随温度的变化如图所示。
的选择性随温度的变化如图所示。
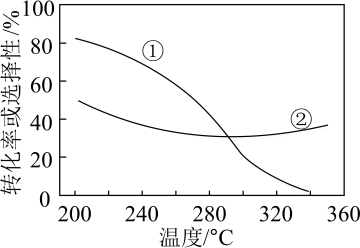
已知: 的选择性=
的选择性=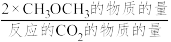 ×100%,其中表示平衡时
×100%,其中表示平衡时 的选择性的是曲线
的选择性的是曲线___________ (填“①”或“②”);温度高于300℃时,曲线②随温度升高而升高的原因是___________ ;为同时提高 的平衡转化率和平衡时
的平衡转化率和平衡时 的选择性,应选择的反应条件为
的选择性,应选择的反应条件为___________ (填标号)。
a.低温、低压b.高温、高压
c.高温、低压d.低温、高压
(3)也可以利用甲醇脱水制备二甲醚: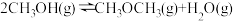 。已知:
。已知: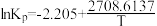 ,其中Kp为以分压表示的平衡常数,T为热力学温度。
,其中Kp为以分压表示的平衡常数,T为热力学温度。
①为提高 的平衡产率,可以采取的措施有
的平衡产率,可以采取的措施有___________ (任写一条)。
②473K时,在密闭容器中加入一定量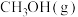 ,采用合适的催化剂进行反应,达到平衡时体系中
,采用合适的催化剂进行反应,达到平衡时体系中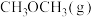 的物质的量分数为
的物质的量分数为___________ (填标号)。
a.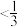 b.
b.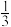 c.
c.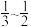 d.
d.
(4)对于反应
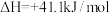 ,反应速率v=v正-v逆=k正p(CO2)·p(H2)-k逆p(CO)·p(H2O),其中k正、k逆分别为正、逆反应速率常数,p为气体的分压(分压=总压×物质的量分数)。
,反应速率v=v正-v逆=k正p(CO2)·p(H2)-k逆p(CO)·p(H2O),其中k正、k逆分别为正、逆反应速率常数,p为气体的分压(分压=总压×物质的量分数)。
①降低温度,k正:k逆___________ (填“增大”、“减小”或“不变”);
②在T K、101kPa下,按照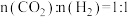 投料,
投料, 转化率为50%时,v正:v逆=4:5,用气体分压表示的平衡常数Kp=
转化率为50%时,v正:v逆=4:5,用气体分压表示的平衡常数Kp=___________ 。
Ⅰ.

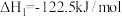
Ⅱ.

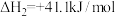
回答下列问题:
(1)反应
 的
的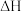 =
=(2)在压强、
 和
和 的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得
的起始投料一定的条件下,发生反应Ⅰ、Ⅱ,实验测得 平衡转化率和平衡时
平衡转化率和平衡时 的选择性随温度的变化如图所示。
的选择性随温度的变化如图所示。
已知:
 的选择性=
的选择性=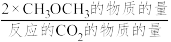 ×100%,其中表示平衡时
×100%,其中表示平衡时 的选择性的是曲线
的选择性的是曲线 的平衡转化率和平衡时
的平衡转化率和平衡时 的选择性,应选择的反应条件为
的选择性,应选择的反应条件为a.低温、低压b.高温、高压
c.高温、低压d.低温、高压
(3)也可以利用甲醇脱水制备二甲醚:
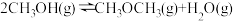 。已知:
。已知: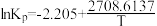 ,其中Kp为以分压表示的平衡常数,T为热力学温度。
,其中Kp为以分压表示的平衡常数,T为热力学温度。①为提高
 的平衡产率,可以采取的措施有
的平衡产率,可以采取的措施有②473K时,在密闭容器中加入一定量
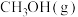 ,采用合适的催化剂进行反应,达到平衡时体系中
,采用合适的催化剂进行反应,达到平衡时体系中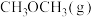 的物质的量分数为
的物质的量分数为a.
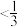 b.
b.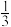 c.
c.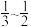 d.
d.
(4)对于反应

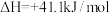 ,反应速率v=v正-v逆=k正p(CO2)·p(H2)-k逆p(CO)·p(H2O),其中k正、k逆分别为正、逆反应速率常数,p为气体的分压(分压=总压×物质的量分数)。
,反应速率v=v正-v逆=k正p(CO2)·p(H2)-k逆p(CO)·p(H2O),其中k正、k逆分别为正、逆反应速率常数,p为气体的分压(分压=总压×物质的量分数)。①降低温度,k正:k逆
②在T K、101kPa下,按照
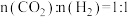 投料,
投料, 转化率为50%时,v正:v逆=4:5,用气体分压表示的平衡常数Kp=
转化率为50%时,v正:v逆=4:5,用气体分压表示的平衡常数Kp=
您最近一年使用:0次
2021-11-13更新
|
507次组卷
|
2卷引用:湖北省华中师范大学第一附属中学2021-2022学年高二上学期期中检测化学试题
3 . I. 丁烯是一种重要的化工原料,可由丁烷催化脱氢制备。回答下列问题:正丁烷(C4H10)脱氢制
1-丁烯(C4H8)的热化学方程式如下:
①C4H10(g)=C4H8(g)+H2(g) ΔH1
已知:②C4H10(g)+ O2(g)=C4H8(g)+H2O(g) ΔH2=-119 kJ·mol-1
O2(g)=C4H8(g)+H2O(g) ΔH2=-119 kJ·mol-1
③H2(g)+ O2(g)=H2O(g) ΔH3=-242 kJ·mol-1
O2(g)=H2O(g) ΔH3=-242 kJ·mol-1
(1)反应①的ΔH1为________ kJ·mol-1。
(2)如图是反应①平衡转化率与反应温度及压强的关系图,x_____ (填“大于”或“小于”)0.1;
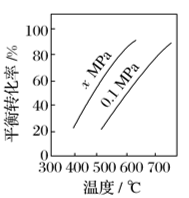
(3)欲使丁烯的平衡产率提高,应采取的措施是___(填标号)。
II.向一容积不变的密闭容器中充入一定量A和B,发生如下反应:xA(g)+2B(s) yC(g);△H<0。在一定条件下,容器中A、C的物质的量浓度随时间变化的曲线如下图。请回答下列问题:
yC(g);△H<0。在一定条件下,容器中A、C的物质的量浓度随时间变化的曲线如下图。请回答下列问题:
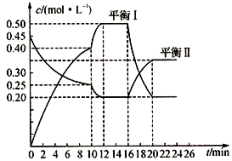
(4)用A的浓度变化表示该反应0~10min内的平均反应速率v(A)=_______ ;
(5)根据图示可确定x:y=______________ ;
(6)0~l0min容器内压强______________ (填“变大”,“不变”或“变小”)
(7)推测第l0min引起曲线变化的反应条件可能是___________ (填序号);第16 min引起曲线变化的反应条件可能是______________ (填序号);
①减压;②增大A的浓度;③增大C的量;④升温;⑤降温;⑥加催化剂
1-丁烯(C4H8)的热化学方程式如下:
①C4H10(g)=C4H8(g)+H2(g) ΔH1
已知:②C4H10(g)+
 O2(g)=C4H8(g)+H2O(g) ΔH2=-119 kJ·mol-1
O2(g)=C4H8(g)+H2O(g) ΔH2=-119 kJ·mol-1③H2(g)+
 O2(g)=H2O(g) ΔH3=-242 kJ·mol-1
O2(g)=H2O(g) ΔH3=-242 kJ·mol-1(1)反应①的ΔH1为
(2)如图是反应①平衡转化率与反应温度及压强的关系图,x

(3)欲使丁烯的平衡产率提高,应采取的措施是___(填标号)。
| A.升高温度 | B.降低温度 | C.增大压强 | D.降低压强 |
II.向一容积不变的密闭容器中充入一定量A和B,发生如下反应:xA(g)+2B(s)
 yC(g);△H<0。在一定条件下,容器中A、C的物质的量浓度随时间变化的曲线如下图。请回答下列问题:
yC(g);△H<0。在一定条件下,容器中A、C的物质的量浓度随时间变化的曲线如下图。请回答下列问题:
(4)用A的浓度变化表示该反应0~10min内的平均反应速率v(A)=
(5)根据图示可确定x:y=
(6)0~l0min容器内压强
(7)推测第l0min引起曲线变化的反应条件可能是
①减压;②增大A的浓度;③增大C的量;④升温;⑤降温;⑥加催化剂
您最近一年使用:0次
解题方法
4 . 雾霾严重影响人们的生活,汽车尾气的排放是造成雾霾天气的重要原因之一、已知汽车尾气排放时容易发生以下反应:
①

②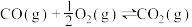
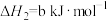
③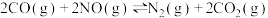
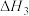
请回答下列问题:
(1)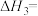
___________  (用含a、b的代数式表示)。
(用含a、b的代数式表示)。
(2)能说明反应③已达平衡状态的标志是___________(填字母)。
(3)在一定温度下,向体积为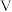
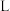 的恒容密闭容器中充入一定量的
的恒容密闭容器中充入一定量的 和
和 ,发生反应③。在
,发生反应③。在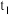 时刻达到平衡,此时
时刻达到平衡,此时 ,
,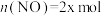 ,
, ,则
,则 的平衡转化率为
的平衡转化率为___________ (用含x、y的代数式表示);再向容器中充入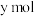
 和
和
 ,则此时
,则此时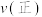
___________ 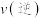 (填“
(填“ ”、“
”、“ ”或“
”或“ ”)。
”)。
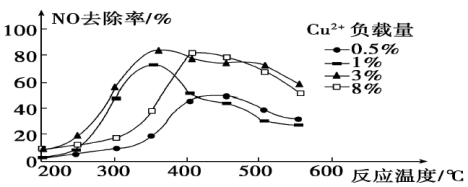
(4)烟气中也含有氮氧化物, 可用于烟气脱硝。为研究温度、催化剂中
可用于烟气脱硝。为研究温度、催化剂中 负载量对
负载量对 去除率的影响,控制其它条件一定,实验结果如图所示。为达到最高的
去除率的影响,控制其它条件一定,实验结果如图所示。为达到最高的 去除率,应选择的反应温度约为
去除率,应选择的反应温度约为___________ , 负载量为
负载量为___________ 。
①


②
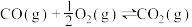
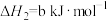
③
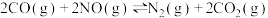
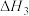
请回答下列问题:
(1)
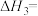
 (用含a、b的代数式表示)。
(用含a、b的代数式表示)。(2)能说明反应③已达平衡状态的标志是___________(填字母)。
A.单位时间内生成  的同时消耗了 的同时消耗了  |
| B.在恒温恒容的容器中,混合气体的密度保持不变 |
| C.在绝热恒容的容器中,反应的平衡常数不再变化 |
D.在恒温恒压的容器中, 的体积分数保持不变 的体积分数保持不变 |
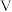
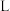 的恒容密闭容器中充入一定量的
的恒容密闭容器中充入一定量的 和
和 ,发生反应③。在
,发生反应③。在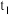 时刻达到平衡,此时
时刻达到平衡,此时 ,
,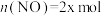 ,
, ,则
,则 的平衡转化率为
的平衡转化率为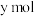
 和
和
 ,则此时
,则此时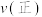
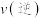 (填“
(填“ ”、“
”、“ ”或“
”或“ ”)。
”)。(4)烟气中也含有氮氧化物,
 可用于烟气脱硝。为研究温度、催化剂中
可用于烟气脱硝。为研究温度、催化剂中 负载量对
负载量对 去除率的影响,控制其它条件一定,实验结果如图所示。为达到最高的
去除率的影响,控制其它条件一定,实验结果如图所示。为达到最高的 去除率,应选择的反应温度约为
去除率,应选择的反应温度约为 负载量为
负载量为
您最近一年使用:0次
5 . 甲醚又称二甲醚,被誉为“21世纪的清洁燃料”。由合成气( 、
、 )制备二甲醚的反应原理如下:
)制备二甲醚的反应原理如下:
①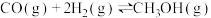

②

回答下列问题:
(1)若由合成气( 、
、 )制备
)制备
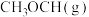 ,且生成
,且生成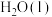 ,整个过程中放出的热量为
,整个过程中放出的热量为 ,则
,则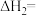
______  。【已知:
。【已知: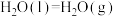
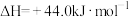 】
】
(2)有人模拟该制备原理,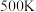 时,在
时,在 的密闭容器中充入
的密闭容器中充入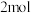
 和
和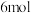
 ,
,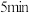 达到平衡,平衡时
达到平衡,平衡时 的转化率为60%,
的转化率为60%, ,用
,用 表示反应①的速率是
表示反应①的速率是______ 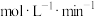 ,可逆反应②的平衡常数
,可逆反应②的平衡常数
______ 。若在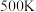 时,测得容器中
时,测得容器中 ,此时反应②的
,此时反应②的
______  (填“
(填“ ”“
”“ ”或“
”或“ ”)。
”)。
(3)在体积一定的密闭容器中发生反应②,如果该反应的平衡常数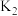 值变小,下列说法正确的是___________。
值变小,下列说法正确的是___________。
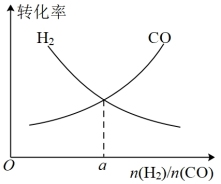
(4)一定条件下在恒温恒容的密闭容器中,按不同投料比充入 和
和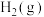 进行反应①,平衡时
进行反应①,平衡时 和
和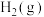 的转化率如图所示,则
的转化率如图所示,则
___________ (填数值)。
(5)甲醇液相脱水法制二甲醚的原理是: ,
, ,与合成气制备二甲醚比较,该工艺的优点是反应温度低,转化率高,其缺点是
,与合成气制备二甲醚比较,该工艺的优点是反应温度低,转化率高,其缺点是___________ 。
 、
、 )制备二甲醚的反应原理如下:
)制备二甲醚的反应原理如下:①
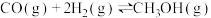

②


回答下列问题:
(1)若由合成气(
 、
、 )制备
)制备
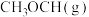 ,且生成
,且生成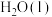 ,整个过程中放出的热量为
,整个过程中放出的热量为 ,则
,则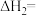
 。【已知:
。【已知: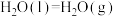
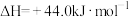 】
】(2)有人模拟该制备原理,
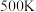 时,在
时,在 的密闭容器中充入
的密闭容器中充入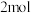
 和
和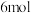
 ,
,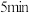 达到平衡,平衡时
达到平衡,平衡时 的转化率为60%,
的转化率为60%, ,用
,用 表示反应①的速率是
表示反应①的速率是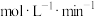 ,可逆反应②的平衡常数
,可逆反应②的平衡常数
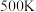 时,测得容器中
时,测得容器中 ,此时反应②的
,此时反应②的
 (填“
(填“ ”“
”“ ”或“
”或“ ”)。
”)。(3)在体积一定的密闭容器中发生反应②,如果该反应的平衡常数
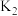 值变小,下列说法正确的是___________。
值变小,下列说法正确的是___________。| A.在平衡移动过程中逆反应速率先增大后减小 |
B.容器中 的体积分数增大 的体积分数增大 |
| C.容器中混合气体的平均相对分子质量减小 |
| D.达到新平衡后体系的压强增大 |
 和
和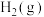 进行反应①,平衡时
进行反应①,平衡时 和
和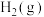 的转化率如图所示,则
的转化率如图所示,则

(5)甲醇液相脱水法制二甲醚的原理是:
 ,
, ,与合成气制备二甲醚比较,该工艺的优点是反应温度低,转化率高,其缺点是
,与合成气制备二甲醚比较,该工艺的优点是反应温度低,转化率高,其缺点是
您最近一年使用:0次
解题方法
6 . 氢能是理想的清洁能源,资源丰富。以太阳能为热源分解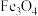 ,经由热化学铁氧化合物循环分解水制
,经由热化学铁氧化合物循环分解水制 的过程如下:
的过程如下:
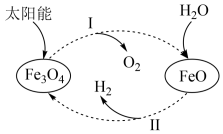
(1)过程Ⅰ:

①过程Ⅰ需要将 不断分离出去,目的是
不断分离出去,目的是______ 。
②平衡常数K随温度变化的关系是______ 。
③一定温度下,在容积可变的密闭容器中,该反应已达平衡,下列说法不正确的是______ 。
A.升高温度,容器内气体密度变大
B.容器内气体密度和相对分子质量都不再改变
C.向容器中通入 ,
,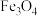 转化率不变
转化率不变
D.缩小容器的容积,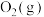 的浓度变大
的浓度变大
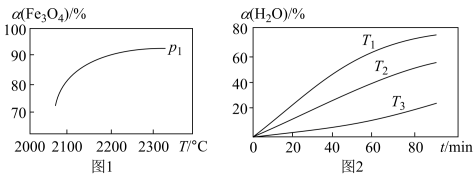
④在压强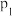 下,
下,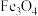 的平衡转化率随温度变化的曲线如图1所示。若将压强由
的平衡转化率随温度变化的曲线如图1所示。若将压强由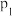 增大到
增大到 ,在图1中画出
,在图1中画出 的
的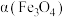 ~
~ 曲线示意图
曲线示意图______ 。
(2)已知 的燃烧热是
的燃烧热是 ,则液态水通过过程Ⅱ转化的热化学方程式为
,则液态水通过过程Ⅱ转化的热化学方程式为___________ 。
(3)其他条件不变时,过程Ⅱ在不同温度下,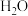 的转化率随时间的变化
的转化率随时间的变化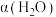 ~
~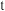 曲线如图2所示。温度
曲线如图2所示。温度 、
、 、
、 由大到小的关系是
由大到小的关系是______ 。
(4)科研人员研制出透氧膜( ),它允许电子、
),它允许电子、 同时透过,可实现水连续分解制
同时透过,可实现水连续分解制 。工作时,
。工作时, 、
、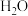 分别在透氧膜的两侧反应。工作原理示意图如下:
分别在透氧膜的两侧反应。工作原理示意图如下:
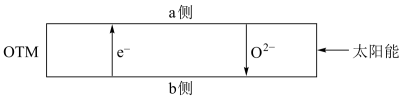
 在
在___________ 侧反应(填“ ”或“
”或“ ”),另一侧的电极反应式是
”),另一侧的电极反应式是___________ 。
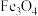 ,经由热化学铁氧化合物循环分解水制
,经由热化学铁氧化合物循环分解水制 的过程如下:
的过程如下:
(1)过程Ⅰ:


①过程Ⅰ需要将
 不断分离出去,目的是
不断分离出去,目的是②平衡常数K随温度变化的关系是
③一定温度下,在容积可变的密闭容器中,该反应已达平衡,下列说法不正确的是
A.升高温度,容器内气体密度变大
B.容器内气体密度和相对分子质量都不再改变
C.向容器中通入
 ,
,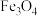 转化率不变
转化率不变D.缩小容器的容积,
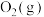 的浓度变大
的浓度变大④在压强
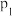 下,
下,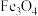 的平衡转化率随温度变化的曲线如图1所示。若将压强由
的平衡转化率随温度变化的曲线如图1所示。若将压强由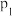 增大到
增大到 ,在图1中画出
,在图1中画出 的
的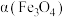 ~
~ 曲线示意图
曲线示意图
(2)已知
 的燃烧热是
的燃烧热是 ,则液态水通过过程Ⅱ转化的热化学方程式为
,则液态水通过过程Ⅱ转化的热化学方程式为(3)其他条件不变时,过程Ⅱ在不同温度下,
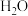 的转化率随时间的变化
的转化率随时间的变化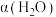 ~
~ 曲线如图2所示。温度
曲线如图2所示。温度 、
、 、
、 由大到小的关系是
由大到小的关系是(4)科研人员研制出透氧膜(
 ),它允许电子、
),它允许电子、 同时透过,可实现水连续分解制
同时透过,可实现水连续分解制 。工作时,
。工作时, 、
、 分别在透氧膜的两侧反应。工作原理示意图如下:
分别在透氧膜的两侧反应。工作原理示意图如下:
 在
在 ”或“
”或“ ”),另一侧的电极反应式是
”),另一侧的电极反应式是
您最近一年使用:0次
7 . 电催化合成氨方法被认为是人工固氮最有前途的方法之一,具有反应条件温和、利用可再生能源驱动、直接以水为氢源等优点,近年来引起科研工作者广泛的研究兴趣。
(1)已知H2的标准摩尔燃烧热为a kJ/mol,NH3的标准摩尔燃烧热为b kJ/mol,请计算反应N2(g)+3H2(g) 2NH3(g)的△H=
2NH3(g)的△H=___________ 。
(2)向一恒定温度的密闭容器中充入平均相对分子质量为15的N2和H2的混合气体,保持30 MPa条件下反应:
①下列哪些选项可以判断反应已达到平衡:___________ 。
A.3v(H2)正=2v(NH3)逆 B.N2的体积分数不再变化
C.N2和H2的浓度比为定值 D.混合气体的密度不再变化
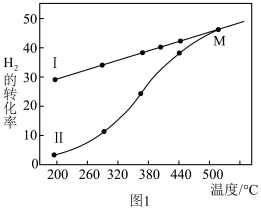
②上述混合气体中氢气的转化率如图1所示,图中曲线Ⅰ为氢气的平衡转化率与温度的关系,曲线Ⅱ表示不同温度下经过相同反应时间后的氢气转化率,请说明随着温度的升高,曲线Ⅱ向曲线Ⅰ逼近,于M点重合,其原因是___________ 。
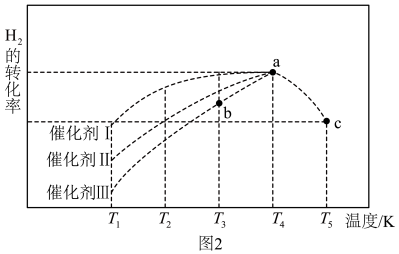
③上述条件下,在不同催化剂作用下,相同时间内H2的转化率随温度的变化如图2所示,催化剂效果最佳的是催化剂___________ (填“Ⅰ”“Ⅱ”“Ⅲ”)。b点v正___________ v逆(填“>”“<”或“=”)。图2中测得a点混合气体平均相对分子质量为18.75,a点对应温度下反应的平衡常数Kp=___________ 。(保留两位有效数字,Kp为以分压表示的平衡常数,分压=总压×物质的量分数)
(3)东南大学王金兰课题组提出合成氨的“表面氢化机理”如图,在较低的电压下实现氮气的还原合成氨。已知:第一步: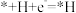 (快)(吸附在催化剂表面的物种用*表示)
(快)(吸附在催化剂表面的物种用*表示)
第二步: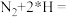 中间体(吸附在催化剂表面)(慢)
中间体(吸附在催化剂表面)(慢)
第三步:___________(快)
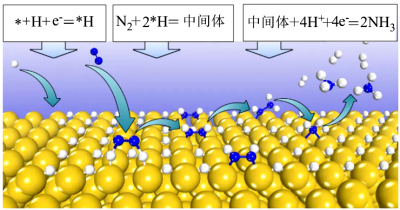
第三步的方程式为___________ ,上述三步中的决速步为第二步,原因是___________ 。该法在合成氨的过程中能量的转化形式是___________ ,较传统工业合成氨法,具有能耗小、环境友好的优点。
(1)已知H2的标准摩尔燃烧热为a kJ/mol,NH3的标准摩尔燃烧热为b kJ/mol,请计算反应N2(g)+3H2(g)
 2NH3(g)的△H=
2NH3(g)的△H=(2)向一恒定温度的密闭容器中充入平均相对分子质量为15的N2和H2的混合气体,保持30 MPa条件下反应:
①下列哪些选项可以判断反应已达到平衡:
A.3v(H2)正=2v(NH3)逆 B.N2的体积分数不再变化
C.N2和H2的浓度比为定值 D.混合气体的密度不再变化
②上述混合气体中氢气的转化率如图1所示,图中曲线Ⅰ为氢气的平衡转化率与温度的关系,曲线Ⅱ表示不同温度下经过相同反应时间后的氢气转化率,请说明随着温度的升高,曲线Ⅱ向曲线Ⅰ逼近,于M点重合,其原因是

③上述条件下,在不同催化剂作用下,相同时间内H2的转化率随温度的变化如图2所示,催化剂效果最佳的是催化剂

(3)东南大学王金兰课题组提出合成氨的“表面氢化机理”如图,在较低的电压下实现氮气的还原合成氨。已知:第一步:
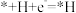 (快)(吸附在催化剂表面的物种用*表示)
(快)(吸附在催化剂表面的物种用*表示)第二步:
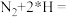 中间体(吸附在催化剂表面)(慢)
中间体(吸附在催化剂表面)(慢)第三步:___________(快)
第三步的方程式为

您最近一年使用:0次
名校
解题方法
8 . 为了去除合成氨原料气中的 以降低能耗及减轻设备的酸蚀,工业上常采用醇胺溶液进行吸收。N一甲基二乙醇胺(用
以降低能耗及减轻设备的酸蚀,工业上常采用醇胺溶液进行吸收。N一甲基二乙醇胺(用 表示)水溶液具有吸收能力强、对设备腐蚀小、可再生等特点被广泛应用。
表示)水溶液具有吸收能力强、对设备腐蚀小、可再生等特点被广泛应用。
 吸收
吸收 的反应可以表示为:
的反应可以表示为:
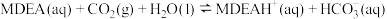
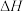
该反应分以下两步进行:
①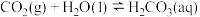
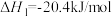 (慢反应)
(慢反应)
②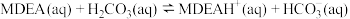
 (快反应)
(快反应)
(1)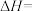
____ 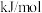 ,该反应在
,该反应在____ (填“高温”“低温”或“任意温度”)条件下自发进行。
(2) 的结构简式为
的结构简式为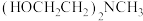 ,以氮原子为中心的
,以氮原子为中心的 模型是
模型是_____ 。
(3)能够正确表示 吸收
吸收 反应能量变化的是______(填标号)。
反应能量变化的是______(填标号)。
(4)其它条件不变时,加入
____ (填“能”或“不能”)增大单位时间内 的吸收率。
的吸收率。
(5)已知 中的氮具有一元碱(类似于
中的氮具有一元碱(类似于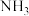 )的性质,
)的性质, ,已知
,已知 的
的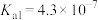 ,
,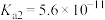 ,则
,则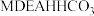 溶液中
溶液中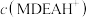
____ 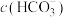 (填“
(填“ ”“
”“ ”或“
”或“ ”)。
”)。
(6)标准平衡常数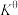 可以表示平衡时各物质的浓度关系:如反应
可以表示平衡时各物质的浓度关系:如反应 的
的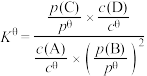 ,其中
,其中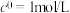 ,
, 为标准大气压,
为标准大气压,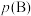 、
、 分别为气体的分压,c为物质的量浓度。在T℃时的刚性密闭容器中,用
分别为气体的分压,c为物质的量浓度。在T℃时的刚性密闭容器中,用 的
的 溶液吸收总压为
溶液吸收总压为 的合成氨原料气(含体积分数分别为
的合成氨原料气(含体积分数分别为 的
的 、
、 的
的 、
、 的
的 ),充分吸收后,
),充分吸收后, 浓度降低为
浓度降低为 ,二氧化碳的吸收率为60%,忽略反应过程中溶液的体积变化,则反应的标准平衡常数
,二氧化碳的吸收率为60%,忽略反应过程中溶液的体积变化,则反应的标准平衡常数
_______ 。
 以降低能耗及减轻设备的酸蚀,工业上常采用醇胺溶液进行吸收。N一甲基二乙醇胺(用
以降低能耗及减轻设备的酸蚀,工业上常采用醇胺溶液进行吸收。N一甲基二乙醇胺(用 表示)水溶液具有吸收能力强、对设备腐蚀小、可再生等特点被广泛应用。
表示)水溶液具有吸收能力强、对设备腐蚀小、可再生等特点被广泛应用。 吸收
吸收 的反应可以表示为:
的反应可以表示为: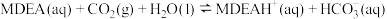
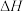
该反应分以下两步进行:
①
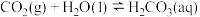
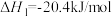 (慢反应)
(慢反应)②
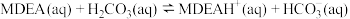
 (快反应)
(快反应)(1)
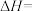
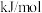 ,该反应在
,该反应在(2)
 的结构简式为
的结构简式为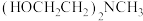 ,以氮原子为中心的
,以氮原子为中心的 模型是
模型是(3)能够正确表示
 吸收
吸收 反应能量变化的是______(填标号)。
反应能量变化的是______(填标号)。A.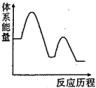 | B.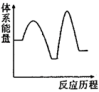 | C.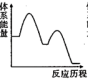 | D.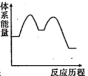 |

 的吸收率。
的吸收率。(5)已知
 中的氮具有一元碱(类似于
中的氮具有一元碱(类似于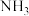 )的性质,
)的性质, ,已知
,已知 的
的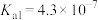 ,
,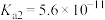 ,则
,则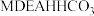 溶液中
溶液中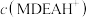
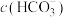 (填“
(填“ ”“
”“ ”或“
”或“ ”)。
”)。(6)标准平衡常数
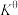 可以表示平衡时各物质的浓度关系:如反应
可以表示平衡时各物质的浓度关系:如反应 的
的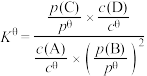 ,其中
,其中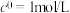 ,
, 为标准大气压,
为标准大气压,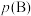 、
、 分别为气体的分压,c为物质的量浓度。在T℃时的刚性密闭容器中,用
分别为气体的分压,c为物质的量浓度。在T℃时的刚性密闭容器中,用 的
的 溶液吸收总压为
溶液吸收总压为 的合成氨原料气(含体积分数分别为
的合成氨原料气(含体积分数分别为 的
的 、
、 的
的 、
、 的
的 ),充分吸收后,
),充分吸收后, 浓度降低为
浓度降低为 ,二氧化碳的吸收率为60%,忽略反应过程中溶液的体积变化,则反应的标准平衡常数
,二氧化碳的吸收率为60%,忽略反应过程中溶液的体积变化,则反应的标准平衡常数
您最近一年使用:0次
2021-09-11更新
|
476次组卷
|
2卷引用:湖北省武汉市部分学校2021-2022学年高三上学期起点质量检测化学试题
9 . 含碳物质的价值型转化,有利于“减碳”和可持续发展。回答下列问题:
(1)科学家用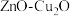 做催化剂,可将
做催化剂,可将 和
和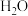 转化为甲烷。已知有关化学反应的能量变化如图所示,则该转化反应的热化学方程式为
转化为甲烷。已知有关化学反应的能量变化如图所示,则该转化反应的热化学方程式为___________ 。

(2)用惰性电极电解 溶液,可将空气中的
溶液,可将空气中的 转化为甲酸根(
转化为甲酸根(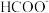 ),然后进一步可制得化工原料甲酸。
),然后进一步可制得化工原料甲酸。 发生反应的电极反应式为
发生反应的电极反应式为___________ ,若电解过程中转移1mol电子,阳极生成氧气的体积(标准状况)为___________ L。
(3)乙苯催化脱氢制取苯乙烯的反应为: (g)+CO2(g)
(g)+CO2(g)
 (g)+H2O(g)+CO(g),其反应历程如下:
(g)+H2O(g)+CO(g),其反应历程如下:
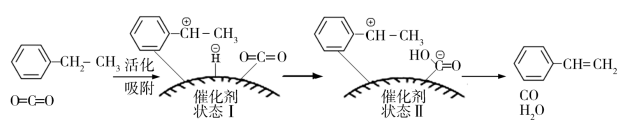
①由原料到“状态Ⅰ”___________ (填“放出”或“吸收”)能量。
②一定温度下,向恒容密闭容器中充入2mol乙苯和 ,起始压强为
,起始压强为 ,平衡时容器内气体总物质的量为5mol,乙苯的转化率为
,平衡时容器内气体总物质的量为5mol,乙苯的转化率为___________ ,用平衡分压代替平衡浓度表示的化学平衡常数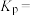
___________ 。
③乙苯平衡转化率与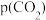 的关系如图所示,乙苯平衡转化率随着
的关系如图所示,乙苯平衡转化率随着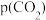 变化的原因是
变化的原因是___________ 。

(1)科学家用
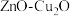 做催化剂,可将
做催化剂,可将 和
和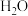 转化为甲烷。已知有关化学反应的能量变化如图所示,则该转化反应的热化学方程式为
转化为甲烷。已知有关化学反应的能量变化如图所示,则该转化反应的热化学方程式为
(2)用惰性电极电解
 溶液,可将空气中的
溶液,可将空气中的 转化为甲酸根(
转化为甲酸根(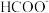 ),然后进一步可制得化工原料甲酸。
),然后进一步可制得化工原料甲酸。 发生反应的电极反应式为
发生反应的电极反应式为(3)乙苯催化脱氢制取苯乙烯的反应为:
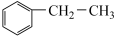 (g)+CO2(g)
(g)+CO2(g)
 (g)+H2O(g)+CO(g),其反应历程如下:
(g)+H2O(g)+CO(g),其反应历程如下:
①由原料到“状态Ⅰ”
②一定温度下,向恒容密闭容器中充入2mol乙苯和
 ,起始压强为
,起始压强为 ,平衡时容器内气体总物质的量为5mol,乙苯的转化率为
,平衡时容器内气体总物质的量为5mol,乙苯的转化率为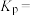
③乙苯平衡转化率与
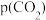 的关系如图所示,乙苯平衡转化率随着
的关系如图所示,乙苯平衡转化率随着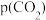 变化的原因是
变化的原因是
您最近一年使用:0次
10 . (1)标准摩尔生成焓是指由稳态单质生成1mol该化合物的焓变,25℃时几种物质的标准生成焓如下:
ⅰ.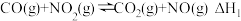
ⅱ.
①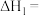
___________ ;CO还原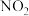 生成两种无污染的气态物质的热化学方程式为
生成两种无污染的气态物质的热化学方程式为___________ 。
②运用化学反应原理分析,反应过程中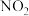 的转化率随温度升高先增大后减小的原因是
的转化率随温度升高先增大后减小的原因是___________ 。
(2)在催化剂Rh表面CO还原NO基元反应机理路径如图a表示,两个N(s)结合生成的 称为
称为 ,
,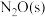 解离生成的
解离生成的 称为
称为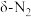 ,基元反应速率随温度的变化关系如图b所示。
,基元反应速率随温度的变化关系如图b所示。
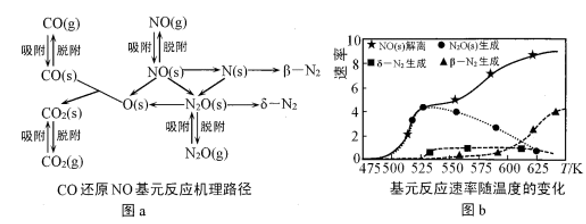
下列说法正确的是___________ (填字母)。
A.在催化剂表面NO的吸附并解离是反应发生的先决条件
B.在催化剂表面 的及时脱附有利于反应的进行
的及时脱附有利于反应的进行
C.当温度超过625K时,生成的 主要是由CO还原
主要是由CO还原 生成的
生成的
D.当温度低于530K时,发生的主要反应为
E.寻找低温下高效的催化剂控制汽车尾气的污染是未来研究的方向
(3)已知对于反应ⅱ: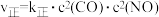 ,
, (
(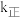 、
、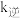 为速率常数,只与温度有关)。
为速率常数,只与温度有关)。
①既要加快NO的消除速率,又要消除更多的NO,可采取措施有___________ (写出其中一条即可)。
②达到平衡后,只升高温度,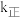 增大的倍数
增大的倍数___________ (填“>”、“<”或“=”)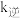 增大的倍数。若在1L恒容密闭容器中充入1molCO和1moLNO发生上述反应,在一定温度下反应达到平衡时,CO的转化率为40%,则
增大的倍数。若在1L恒容密闭容器中充入1molCO和1moLNO发生上述反应,在一定温度下反应达到平衡时,CO的转化率为40%,则
___________ (计算结果用分数表示)。
| 物质 | 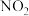 | CO |  | NO |
| 标准生成焓(kJ/mol) | 33.1 | -110.5 | -183.6 | 90.3 |
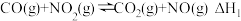
ⅱ.

①
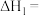
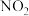 生成两种无污染的气态物质的热化学方程式为
生成两种无污染的气态物质的热化学方程式为②运用化学反应原理分析,反应过程中
 的转化率随温度升高先增大后减小的原因是
的转化率随温度升高先增大后减小的原因是(2)在催化剂Rh表面CO还原NO基元反应机理路径如图a表示,两个N(s)结合生成的
 称为
称为 ,
,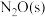 解离生成的
解离生成的 称为
称为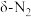 ,基元反应速率随温度的变化关系如图b所示。
,基元反应速率随温度的变化关系如图b所示。
下列说法正确的是
A.在催化剂表面NO的吸附并解离是反应发生的先决条件
B.在催化剂表面
 的及时脱附有利于反应的进行
的及时脱附有利于反应的进行C.当温度超过625K时,生成的
 主要是由CO还原
主要是由CO还原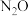 生成的
生成的D.当温度低于530K时,发生的主要反应为

E.寻找低温下高效的催化剂控制汽车尾气的污染是未来研究的方向
(3)已知对于反应ⅱ:
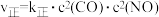 ,
, (
(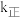 、
、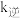 为速率常数,只与温度有关)。
为速率常数,只与温度有关)。①既要加快NO的消除速率,又要消除更多的NO,可采取措施有
②达到平衡后,只升高温度,
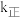 增大的倍数
增大的倍数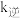 增大的倍数。若在1L恒容密闭容器中充入1molCO和1moLNO发生上述反应,在一定温度下反应达到平衡时,CO的转化率为40%,则
增大的倍数。若在1L恒容密闭容器中充入1molCO和1moLNO发生上述反应,在一定温度下反应达到平衡时,CO的转化率为40%,则
您最近一年使用:0次



