解题方法
1 . Co、La元素及其化合物在工业及生活方面有重要应用。回答下列问题:
(1)基态Co的价电子排布图_______ ,核外电子的空间运动状态有_______ 种。
(2)一定条件下, 、
、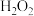 、
、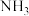 和
和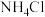 反应可制得
反应可制得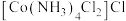 ,反应的方程式为
,反应的方程式为_______ ,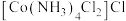 的配位数为
的配位数为_______ 。区别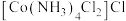 和
和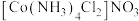 两种溶液的试剂是
两种溶液的试剂是_______ 。
(3)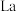 的八配位金属羰基化合物
的八配位金属羰基化合物 的结构如图所示,
的结构如图所示, 作配体,配位原子为C,而不是O,其原因是
作配体,配位原子为C,而不是O,其原因是_______ ,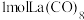 含
含_______  键。
键。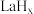 在储氢和超导等领域具有重要应用。高压下,
在储氢和超导等领域具有重要应用。高压下,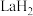 中的每个
中的每个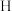 结合4个
结合4个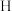 形成类似
形成类似 的结构独立存在,即得到晶体
的结构独立存在,即得到晶体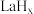 。则
。则
_______ 。晶体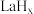 的密度为
的密度为_______ 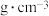 。(写出计算式即可)
。(写出计算式即可)
(1)基态Co的价电子排布图
(2)一定条件下,
 、
、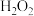 、
、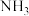 和
和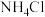 反应可制得
反应可制得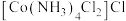 ,反应的方程式为
,反应的方程式为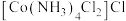 的配位数为
的配位数为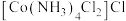 和
和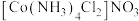 两种溶液的试剂是
两种溶液的试剂是(3)
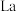 的八配位金属羰基化合物
的八配位金属羰基化合物 的结构如图所示,
的结构如图所示, 作配体,配位原子为C,而不是O,其原因是
作配体,配位原子为C,而不是O,其原因是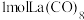 含
含 键。
键。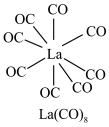
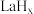 在储氢和超导等领域具有重要应用。高压下,
在储氢和超导等领域具有重要应用。高压下,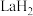 中的每个
中的每个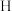 结合4个
结合4个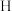 形成类似
形成类似 的结构独立存在,即得到晶体
的结构独立存在,即得到晶体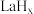 。则
。则
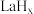 的密度为
的密度为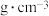 。(写出计算式即可)
。(写出计算式即可)
您最近一年使用:0次
解题方法
2 . 将盐酸分别滴加到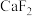 和
和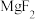 浊液中,体系中
浊液中,体系中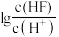 与
与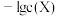 (
( 为
为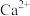 、
、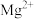 、
、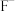 )的变化关系如图所示。已知
)的变化关系如图所示。已知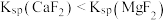 ,下列说法错误的是
,下列说法错误的是
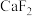 和
和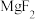 浊液中,体系中
浊液中,体系中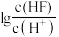 与
与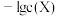 (
( 为
为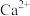 、
、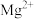 、
、 )的变化关系如图所示。已知
)的变化关系如图所示。已知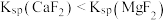 ,下列说法错误的是
,下列说法错误的是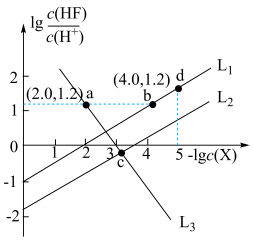
A. 表示 表示 与 与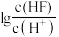 的变化曲线 的变化曲线 |
B.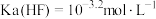 |
C.c点溶液中: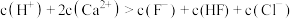 (不考虑水的电离因素) (不考虑水的电离因素) |
D.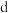 点坐标为 点坐标为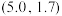 |
您最近一年使用:0次
3 . 1,6—烯炔类化合物在膦配位的铑催化剂的作用下,可进行不对称环化异构,生成五元杂环化合物,反应如下。下列说法正确的是

甲 乙
| A.甲和乙均不存在手性碳原子 | B.甲中所有碳原子不可能共平面 |
| C.可用新制银氨溶液鉴别甲和乙 | D.甲的同分异构体中苯环上有5个取代基的共有6种 |
您最近一年使用:0次
解题方法
4 . 甲烷干重整(DRM)是利用 和
和 在催化剂的作用下制备合成气,作为可持续替代燃料。DRM的主要反应为:
在催化剂的作用下制备合成气,作为可持续替代燃料。DRM的主要反应为:
Ⅰ.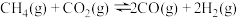
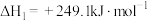
Ⅱ.
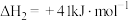
该过程中还伴随积碳反应的发生。
(1)一定压强下,由最稳定单质生成 化合物的焓变为该物质的摩尔生成焓。已知
化合物的焓变为该物质的摩尔生成焓。已知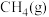 、
、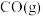 的摩尔生成焓分别为
的摩尔生成焓分别为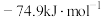 、
、 。则
。则 的摩尔生成焓为
的摩尔生成焓为_______  。
。
(2)甲烷和二氧化碳的起始物质的量均为 ,实验测得在
,实验测得在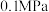 下,平衡时各组分的量随温度变化如图所示。
下,平衡时各组分的量随温度变化如图所示。_______ mol,反应Ⅱ的压强平衡常数
_______ 。
②为了消除积碳带来的影响,反应选择在_______ (填“高温”或“低温”)下进行。
(3)在不同压强下,按照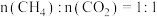 投料,假设只发生反应Ⅰ和Ⅱ。
投料,假设只发生反应Ⅰ和Ⅱ。 的平衡转化率随温度的变化关系如图所示。
的平衡转化率随温度的变化关系如图所示。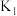 、
、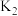 、
、 ,三者由大到小顺序为
,三者由大到小顺序为________ 。
②压强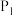 、
、 、
、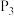 、
、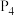 由小到大的顺序为
由小到大的顺序为________ 。
③ ,四条曲线几乎交于一点的原因是
,四条曲线几乎交于一点的原因是________ 。
 和
和 在催化剂的作用下制备合成气,作为可持续替代燃料。DRM的主要反应为:
在催化剂的作用下制备合成气,作为可持续替代燃料。DRM的主要反应为:Ⅰ.
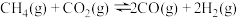
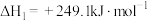
Ⅱ.

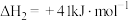
该过程中还伴随积碳反应的发生。
(1)一定压强下,由最稳定单质生成
 化合物的焓变为该物质的摩尔生成焓。已知
化合物的焓变为该物质的摩尔生成焓。已知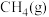 、
、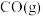 的摩尔生成焓分别为
的摩尔生成焓分别为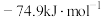 、
、 。则
。则 的摩尔生成焓为
的摩尔生成焓为 。
。(2)甲烷和二氧化碳的起始物质的量均为
 ,实验测得在
,实验测得在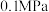 下,平衡时各组分的量随温度变化如图所示。
下,平衡时各组分的量随温度变化如图所示。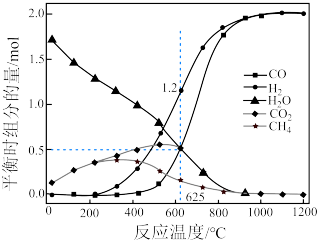

②为了消除积碳带来的影响,反应选择在
(3)在不同压强下,按照
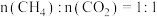 投料,假设只发生反应Ⅰ和Ⅱ。
投料,假设只发生反应Ⅰ和Ⅱ。 的平衡转化率随温度的变化关系如图所示。
的平衡转化率随温度的变化关系如图所示。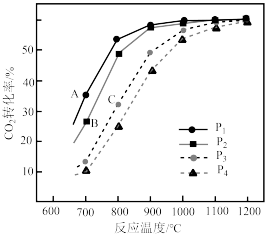
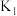 、
、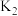 、
、 ,三者由大到小顺序为
,三者由大到小顺序为②压强
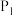 、
、 、
、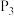 、
、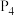 由小到大的顺序为
由小到大的顺序为③
 ,四条曲线几乎交于一点的原因是
,四条曲线几乎交于一点的原因是
您最近一年使用:0次
解题方法
5 . 锂离子电池正极材料(主要成分为 ,还含有少量铝箔、
,还含有少量铝箔、 等杂质),从废旧锂离子正极材料中回收氧化钴的工艺流程如图所示:
等杂质),从废旧锂离子正极材料中回收氧化钴的工艺流程如图所示: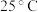 ,
, ,
, 。
。
回答下列问题:
(1)废旧电池拆解提取正极材料前,需将其浸入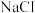 溶液中,其目的是
溶液中,其目的是________ 。
(2)滤液1的主要成分是________ (填化学式,下同)。滤液2的主要成分是________ 。
(3)“酸溶”时发生的主要反应的离子方程式________ ;不用盐酸替代硫酸的原因是________ 。
(4)不宜使用 沉钴的原因是
沉钴的原因是________ 。
(5)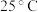 ,若“沉钴”开始时
,若“沉钴”开始时 ,为了获得纯净的
,为了获得纯净的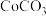 ,应控制溶液的
,应控制溶液的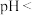
________ 。
(6)若最终得到产品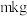 ,则可以重新制备
,则可以重新制备________ 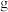 正极材料(含钴
正极材料(含钴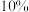 )。
)。
 ,还含有少量铝箔、
,还含有少量铝箔、 等杂质),从废旧锂离子正极材料中回收氧化钴的工艺流程如图所示:
等杂质),从废旧锂离子正极材料中回收氧化钴的工艺流程如图所示: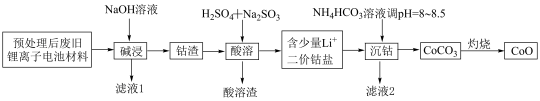
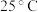 ,
, ,
, 。
。回答下列问题:
(1)废旧电池拆解提取正极材料前,需将其浸入
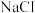 溶液中,其目的是
溶液中,其目的是(2)滤液1的主要成分是
(3)“酸溶”时发生的主要反应的离子方程式
(4)不宜使用
 沉钴的原因是
沉钴的原因是(5)
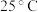 ,若“沉钴”开始时
,若“沉钴”开始时 ,为了获得纯净的
,为了获得纯净的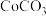 ,应控制溶液的
,应控制溶液的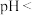
(6)若最终得到产品
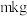 ,则可以重新制备
,则可以重新制备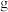 正极材料(含钴
正极材料(含钴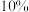 )。
)。
您最近一年使用:0次
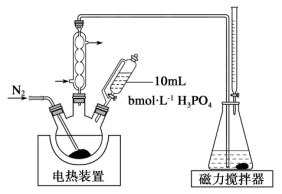
6 . 亚硫酸钠可用作人造纤维稳定剂、造纸木质素脱除剂等。某课题小组利用二氧化硫制备亚硫酸钠并测定其含量,装置如下所示。
(1)装置A中所装试剂为________ ,作用是________ 。
(2)实验时,关闭活塞 ,通入过量
,通入过量 ,再打开活塞
,再打开活塞 ,充分反应后即可制得亚硫酸钠。反应的离子方程式为
,充分反应后即可制得亚硫酸钠。反应的离子方程式为________ 。
(3)上述装置中存在的一处缺陷是________ 。
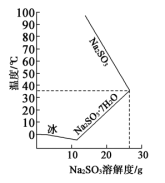
(4)亚硫酸钠的溶解度随温度的变化如上图所示,从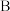 装置溶液中获得
装置溶液中获得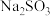 的方法是
的方法是________ 。
(5)测定上述产品中亚硫酸钠样品含量。其装置如图所示: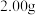 样品并加入水将其溶解,锥形瓶中加入
样品并加入水将其溶解,锥形瓶中加入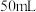 水、
水、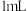 淀粉溶液,并预加
淀粉溶液,并预加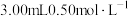 的碘标准溶液,搅拌。
的碘标准溶液,搅拌。
②持续通入氮气,再加入过量磷酸,加入并保持微沸。同时用碘标准液滴定,至终点时滴定消耗了 碘标准溶液。
碘标准溶液。
③做空白实验,消耗了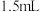 碘标准溶液。
碘标准溶液。
该样品中亚硫酸钠的含量为_______ (保留两位有效数字);
若先加入磷酸再通入氮气,会使测定结果_______ 。(填“偏高”“偏低”或“无影响”)

(1)装置A中所装试剂为
(2)实验时,关闭活塞
 ,通入过量
,通入过量 ,再打开活塞
,再打开活塞 ,充分反应后即可制得亚硫酸钠。反应的离子方程式为
,充分反应后即可制得亚硫酸钠。反应的离子方程式为(3)上述装置中存在的一处缺陷是
(4)亚硫酸钠的溶解度随温度的变化如上图所示,从
 装置溶液中获得
装置溶液中获得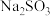 的方法是
的方法是(5)测定上述产品中亚硫酸钠样品含量。其装置如图所示:
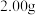 样品并加入水将其溶解,锥形瓶中加入
样品并加入水将其溶解,锥形瓶中加入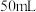 水、
水、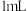 淀粉溶液,并预加
淀粉溶液,并预加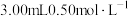 的碘标准溶液,搅拌。
的碘标准溶液,搅拌。②持续通入氮气,再加入过量磷酸,加入并保持微沸。同时用碘标准液滴定,至终点时滴定消耗了
 碘标准溶液。
碘标准溶液。③做空白实验,消耗了
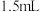 碘标准溶液。
碘标准溶液。该样品中亚硫酸钠的含量为
若先加入磷酸再通入氮气,会使测定结果
您最近一年使用:0次
解题方法
7 . 已知橙红色的配离子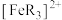 可被
可被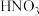 氧化成淡蓝色的配离子
氧化成淡蓝色的配离子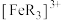 (
(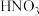 的还原产物是
的还原产物是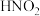 ),现用浓度分别为
),现用浓度分别为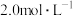 、
、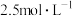 、
、 的
的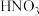 溶液进行实验,
溶液进行实验,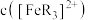 随时间
随时间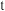 的变化曲线如图所示。下列有关说法错误的是
的变化曲线如图所示。下列有关说法错误的是
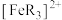 可被
可被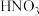 氧化成淡蓝色的配离子
氧化成淡蓝色的配离子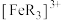 (
(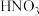 的还原产物是
的还原产物是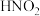 ),现用浓度分别为
),现用浓度分别为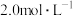 、
、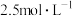 、
、 的
的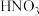 溶液进行实验,
溶液进行实验,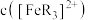 随时间
随时间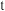 的变化曲线如图所示。下列有关说法错误的是
的变化曲线如图所示。下列有关说法错误的是
A.实验中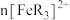 、 、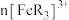 之和不变时,反应达到平衡状态 之和不变时,反应达到平衡状态 |
| B.三组实验中,反应速率都是前期速率增加,后期速率减小 |
C.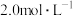 的硝酸进行实验,平衡时 的硝酸进行实验,平衡时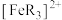 的平均消耗速率为 的平均消耗速率为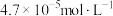 |
D.平衡后加水稀释, 增大 增大 |
您最近一年使用:0次
8 . 以废料铅膏(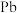 、
、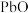 、
、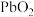 、
、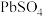 等)为原料,制备超细
等)为原料,制备超细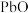 ,实现铅循环再利用的流程如下:
,实现铅循环再利用的流程如下: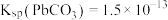 ,
,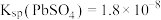 。②
。②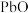 溶解在
溶解在 溶液中(杂质不溶于
溶液中(杂质不溶于 )的溶解度曲线如图所示:
)的溶解度曲线如图所示:
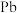 、
、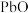 、
、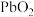 、
、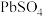 等)为原料,制备超细
等)为原料,制备超细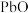 ,实现铅循环再利用的流程如下:
,实现铅循环再利用的流程如下: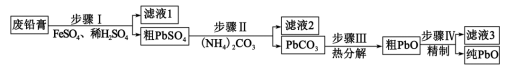
 ,
, 。②
。②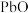 溶解在
溶解在 溶液中(杂质不溶于
溶液中(杂质不溶于 )的溶解度曲线如图所示:
)的溶解度曲线如图所示:
A.步骤Ⅰ发生的主要反应有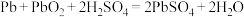 |
B.步骤Ⅱ,若滤出的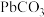 沉淀中混有 沉淀中混有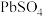 ,该滤液中 ,该滤液中 |
| C.过程中为提高产品产率,滤液3应循环使用 |
D.精制的操作是在 用碱溶解,然后趁热过滤、洗涤、干燥 用碱溶解,然后趁热过滤、洗涤、干燥 |
您最近一年使用:0次
9 .  (乙二胺四乙酸,简写为
(乙二胺四乙酸,简写为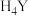 )滴定
)滴定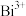 和
和 时发生反应:
时发生反应: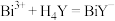 (无色)
(无色)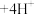
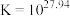 ;
;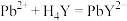 (无色)
(无色)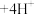
 ;二甲酚橙溶液呈黄色,在
;二甲酚橙溶液呈黄色,在 值
值 时能与
时能与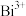 结合,
结合, 值
值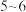 时与
时与 结合,形成的化合物均为紫色。用
结合,形成的化合物均为紫色。用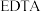 测定某
测定某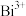 和
和 混合液中金属离子浓度,实验步骤为:
混合液中金属离子浓度,实验步骤为:
①取 混合液于锥形瓶中调
混合液于锥形瓶中调 值,滴加几滴二甲酚橙溶液,用浓度为
值,滴加几滴二甲酚橙溶液,用浓度为 标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。
标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。
②向锥形瓶中加入缓冲溶液调 至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。
至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。
③重复以上实验三次,数据一的均值为 ,数据二的均值为
,数据二的均值为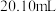 。
。
下列说法正确的是( )
 (乙二胺四乙酸,简写为
(乙二胺四乙酸,简写为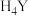 )滴定
)滴定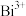 和
和 时发生反应:
时发生反应: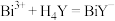 (无色)
(无色)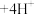
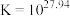 ;
;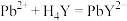 (无色)
(无色)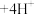
 ;二甲酚橙溶液呈黄色,在
;二甲酚橙溶液呈黄色,在 值
值 时能与
时能与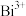 结合,
结合, 值
值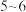 时与
时与 结合,形成的化合物均为紫色。用
结合,形成的化合物均为紫色。用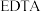 测定某
测定某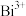 和
和 混合液中金属离子浓度,实验步骤为:
混合液中金属离子浓度,实验步骤为:①取
 混合液于锥形瓶中调
混合液于锥形瓶中调 值,滴加几滴二甲酚橙溶液,用浓度为
值,滴加几滴二甲酚橙溶液,用浓度为 标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。
标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。②向锥形瓶中加入缓冲溶液调
 至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。
至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。③重复以上实验三次,数据一的均值为
 ,数据二的均值为
,数据二的均值为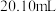 。
。下列说法正确的是( )
A.步骤② 值调好时溶液为黄色 值调好时溶液为黄色 | B.步骤①与步骤②突变颜色相同 |
C.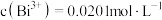 | D.若步骤①中多滴了EDTA,则测得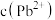 偏高 偏高 |
您最近一年使用:0次
10 .  (乙二胺四乙酸,简写为
(乙二胺四乙酸,简写为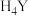 )滴定
)滴定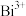 和
和 时发生反应:
时发生反应: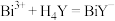 (无色)
(无色)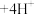
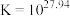 ;
;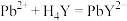 (无色)
(无色)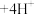
 ;二甲酚橙溶液呈黄色,在
;二甲酚橙溶液呈黄色,在 值
值 时能与
时能与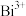 结合,
结合, 值
值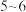 时与
时与 结合,形成的化合物均为紫色。用
结合,形成的化合物均为紫色。用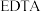 测定某
测定某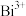 和
和 混合液中金属离子浓度,实验步骤为:
混合液中金属离子浓度,实验步骤为:
①取 混合液于锥形瓶中调
混合液于锥形瓶中调 值,滴加几滴二甲酚橙溶液,用浓度为
值,滴加几滴二甲酚橙溶液,用浓度为 标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。
标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。
②向锥形瓶中加入缓冲溶液调 至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。
至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。
③重复以上实验三次,数据一的均值为 ,数据二的均值为
,数据二的均值为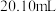 。
。
下列说法错误的是
 (乙二胺四乙酸,简写为
(乙二胺四乙酸,简写为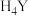 )滴定
)滴定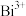 和
和 时发生反应:
时发生反应: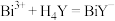 (无色)
(无色)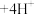
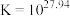 ;
;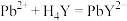 (无色)
(无色)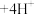
 ;二甲酚橙溶液呈黄色,在
;二甲酚橙溶液呈黄色,在 值
值 时能与
时能与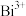 结合,
结合, 值
值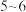 时与
时与 结合,形成的化合物均为紫色。用
结合,形成的化合物均为紫色。用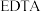 测定某
测定某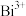 和
和 混合液中金属离子浓度,实验步骤为:
混合液中金属离子浓度,实验步骤为:①取
 混合液于锥形瓶中调
混合液于锥形瓶中调 值,滴加几滴二甲酚橙溶液,用浓度为
值,滴加几滴二甲酚橙溶液,用浓度为 标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。
标准EDTA溶液滴定至颜色突变且半分钟不恢复原色。此时消耗EDTA的体积记录为数据一。②向锥形瓶中加入缓冲溶液调
 至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。
至合适数值,再用EDTA滴定至颜色突变且半分钟不恢复原色。再次消耗EDTA体积记录为数据二。③重复以上实验三次,数据一的均值为
 ,数据二的均值为
,数据二的均值为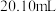 。
。下列说法错误的是
A.用滴定管量取 混合液 混合液 |
| B.配制EDTA标准液时可以用底部有少量蒸馏水的容量瓶 |
C.二甲酚橙与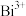 、 、 结合形成化合物能力比EDTA强 结合形成化合物能力比EDTA强 |
| D.滴定读数时,应单手持滴定管上端并保持自然垂直 |
您最近一年使用:0次



